Experimental protocol PIPE
|
|
|
- Lesley Casey
- 5 years ago
- Views:
Transcription
1 Experimental protocol PIPE May 5, 2016 Abstract PIPE is a uorescence perturbation technique that works by measuring the expansion of a laser induced perturbation of photo convertible fused protein in the cytosol of cells. By measuring the expansion of the Gaussian distribution to nd the mean square displacement (MSD) as a function of time, which is a measure of the spatial extent of random motion in the case of diusion, PIPE calculates the diusion coecient of a cytosolic protein in question. The diusion coecient can then compared to a standard which could be a similar protein of the same size or even the same protein under dierent condition to evaluate changes in the protein structure or interactions. Here we describe how Dendra2, a green (ex: 488) to red (ex: 651) photoconvertible protein fused to a cytosolic protein can be used to measure the diusion coecient of dierent proteins. 1 Transfection We preformed our experiment in COS-7 cells which are easily handled and transfected but other types of cells can also be used, however as the perturbation expands it reaches the cell membrane which changes the Gaussian distribution of the diusing protein, therefore it is best to choose a cell line with medium to large cell volume. 1. Three days prior to performing the experiment determine the cells conuency. When cells are about percent conuent, dilute the cells 1:4 in culture medium and grow them in a 4-well 35mm glass bottom plate until cells reach 50% conuency. 2. When cells reach 50% conuency, which should take about one day, prewarm PEI (Polyethylenimine 1 µg/µl) and the transfer vector DNA to room temperature and prepare a DNA:PEI mix in 1:2.5 ratio in 50 µl of culture medium (without serum). Incubate the mix for 30 minutes at room temperature. 3. Wash the cell with PBS. and add the DNA:PEI mix to 450 µl of culture medium and add the medium with the DNA:PEI mix to the prewashed cells. 1
2 4. Incubate the cells for 2-3 hours at a temperature suitable for cell growth. 5. Remove the DNA/PEI mix and wash twice with PBS. Add 500µl of culture medium. 6. The cells start expressing the protein fused to Dendra2 within a few hours of transfection and should be ready to image within hours of transfection 2 Microscopy PIPE can be performed on any commercial confocal microscope system that has a linear intensity response to uorescent signal and can perform photoconversion experiments. We performed our experiments on a NIKON A1r system in resonant mode which is a fast acquisition mode. However, other settings can be used as long as the guidelines below are met: Important notes: Prior to beginning the experiment it is recommended that a sample with an isotropic uorescence, for example an auto-uorescent plastic slide, be used to check the spatial evenness of illumination. In other words illuminating a uniform sample could be used to make sure the uniformity of the imaged area. By acquiring a series of frames and averaging these frames, data collected could be corrected for non uniform illumination. Before starting the experiment the photo-converting laser pulse power and pulse duration should be adjusted such that the amplitude of the photo activated region is maximized without causing damage to the cell. 1. Photo-conversion setup (a) Set the 405nm activation laser power to 100% (on our system we use a 50mW laser) (b) Set the activating laser time interval. We recommend using 100ms as a starting point and gradually increasing the time interval in case the resulting photo-converted uorescent intensity is too low. Data analysis and accuracy depend on identifying the photo-activation pulse as it expands. Therefore a stronger pulse will be visible further away from the initial pulse position. However, longer time intervals allow for more time for the initial pulse to expand away from the photoconversion point, therefore users should check as at least that the initial uorescence after the pulse is clearly visible to the eye. (c) Set the total experiment duration for about 30 seconds and the time interval for acquisition before the photo activation session to about 4 seconds. The frames acquired prior to photo-activation could be used as a reference later on to measure background levels. Note that depending on the rate of diusion the total acquisition time and frame 2
3 rate will need to be adjusted. For slow moving proteins, such as proteins that aggregate or have many binding partners, longer acquisition time to capture the pulse expansion with longer time intervals between individual frames to reduce bleaching. Faster smaller proteins that freely diuse will require shorter acquisition time with a high frame rate. (d) Select the photo-activation position to the center of the imaged area. 2. Image acquisition setup (a) Set the image scanning mode to bi-directional and the frame rate be 30 frames per second. Some commercial systems that do not allow such high frame rate, can decrease the ROI size in order to gain an increase in frame rate. Also depending on the protein in question, lower frame rates could be used. (b) Using a 60x objective pick a few cells (about 5-10 cells should be enough) and adjust the gain and laser power using the 488nm laser, make sure the green image is not saturated. These values might require adjustment during the experiment to allow cells with dierent protein concentration to be imaged. (c) Move the cells in the center of the ROI and zoom in until most of the cell edges are not in the ROI. Make sure that photo-activation pulse is aimed at the desired cellular compartment while making sure to avoid, as much as possible, large organelle that are sequestered from the cytoplasm, such as the nucleus. (d) Adjust the the gain to the same setting as the 488 channel the and laser power of the red channel (561nm) to be three times larger than that of the green channel. These values should be adjusted and checked while acquiring data to make sure that: i. The initial pulse after photo-activation is not saturated. Detectors or camera have limited capabilities to translate the intensity of an area in the imaged object to a digital signal (aka digital levels). This range known as the dynamic range sets the number of values that span the dierent uorescence intensities in the imaged cell. Smaller values or larger values that do t in this range are assigned the smaller or larger accordingly in the dynamic range resulting in a cuto. For example, if the initial pulse intensity is too strong the result will be that the center of the Gaussian pulse, which has the higher values, will be cuto, attening the peak. ii. That the pulse is visible for a maximum number of frames after photo activation (there are no minimum or maximum requirement, rather this note should be regarded as a recommendation). This can achieved by making sure that the initial pulse values 3
4 are at the top of the dynamic range while keeping that no pixels are saturated. Another ecient way to do this is to increase the laser in the expanse of the gain which increases the span of values which could be represented by the dynamic range. (e) Usually uorescence perturbation techniques are very sensitive to bleaching and require that experiments be performed with low laser power. However, PIPE was shown to be less sensitive to bleaching, therefore the red channel laser power can be increased in the expense of reducing the gain. Although increasing the gain increases the image intensity is also increases noise by the same ratio, therefore it is better to increase the laser power as this improves the signal to noise ratio. Important note: high laser power can also be damaging to cells, therefore increasing the laser power should be done moderately. (f) Always make sure to check the shape of the raw data in the graphical reports of the computational analysis (see the separate user manual for that part). If you see that the initial peak is saturated, execute the measurement again with decreased laser power or gain. If the signal vanishes quickly, increase the laser power or gain, or choose a brighter cell. 4
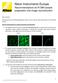
5 5 Figure 1: (A) A time series showing the expansion of a photo-converted Dendra 2 in the cytosol of a COS-7 cell following a 100ms pulse. Image size is 36µm x 36µm. (B) Image of cell expressing cytosolic Dendra2 demonstrating ROI selection (white square) as well as the photo-activation point (white arrow).
1 Co Localization and Working flow with the lsm700
 1 Co Localization and Working flow with the lsm700 Samples -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ BrDU with alexa 488. -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ no BrDU (but with
1 Co Localization and Working flow with the lsm700 Samples -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ BrDU with alexa 488. -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ no BrDU (but with
Nasmyth Ultraview Vox User Protocol
 Nasmyth Ultraview Vox User Protocol Switch on all wall sockets labelled Nasmyth, switch camera on (power supply located on table behind monitor), switch on laser switch in laser rack, switch computer on
Nasmyth Ultraview Vox User Protocol Switch on all wall sockets labelled Nasmyth, switch camera on (power supply located on table behind monitor), switch on laser switch in laser rack, switch computer on
Multifluorescence The Crosstalk Problem and Its Solution
 Multifluorescence The Crosstalk Problem and Its Solution If a specimen is labeled with more than one fluorochrome, each image channel should only show the emission signal of one of them. If, in a specimen
Multifluorescence The Crosstalk Problem and Its Solution If a specimen is labeled with more than one fluorochrome, each image channel should only show the emission signal of one of them. If, in a specimen
Comparing FCS and FRAP as methodologies for calculating diffusion
 Bi/BE 227 Winter 2018 Assignment #4 Comparing FCS and FRAP as methodologies for calculating diffusion Schedule: Jan 29: Assignment Jan 29-Feb 14: Work on assignment Feb 14: Student PowerPoint presentations.
Bi/BE 227 Winter 2018 Assignment #4 Comparing FCS and FRAP as methodologies for calculating diffusion Schedule: Jan 29: Assignment Jan 29-Feb 14: Work on assignment Feb 14: Student PowerPoint presentations.
Confocal imaging on the Leica TCS SP8. 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software:
 Confocal imaging on the Leica TCS SP8 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software: 4) Do not touch the microscope while the software is initializing. Choose your options: Turn
Confocal imaging on the Leica TCS SP8 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software: 4) Do not touch the microscope while the software is initializing. Choose your options: Turn
LSM 780 Confocal Microscope Standard Operation Protocol
 LSM 780 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Sign on log sheet according to Actual start time 2. Check Compressed Air supply for the air table 3. Switch
LSM 780 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Sign on log sheet according to Actual start time 2. Check Compressed Air supply for the air table 3. Switch
Spatial intensity distribution analysis Matlab user guide
 Spatial intensity distribution analysis Matlab user guide August 2011 Guide on how to use the SpIDA graphical user interface. This little tutorial provides a step by step tutorial explaining how to get
Spatial intensity distribution analysis Matlab user guide August 2011 Guide on how to use the SpIDA graphical user interface. This little tutorial provides a step by step tutorial explaining how to get
Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev. Microscopy course, Michmoret Dec 2005
 Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev Why use confocal microscopy? Principles of the laser scanning confocal microscope. Image resolution. Manipulating the
Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev Why use confocal microscopy? Principles of the laser scanning confocal microscope. Image resolution. Manipulating the
SUPPLEMENTARY METHODS. PIN2-Dendra2 transgenic line creation
 SUPPLEMENTARY METHODS PIN2-Dendra2 transgenic line creation The PIN2-Dendra2 construct was described previously (Jásik et al., 2013). Shortly, for generation of the expressing vector encompassing the PIN2-Dendra2
SUPPLEMENTARY METHODS PIN2-Dendra2 transgenic line creation The PIN2-Dendra2 construct was described previously (Jásik et al., 2013). Shortly, for generation of the expressing vector encompassing the PIN2-Dendra2
Practical work no. 3: Confocal Live Cell Microscopy
 Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Opterra. Multipoint Scanning Confocal Microscope. Innovation with Integrity. Cell-Friendly, High-Speed, High-Resolution Imaging
 Opterra Multipoint Scanning Confocal Microscope Cell-Friendly, High-Speed, High-Resolution Imaging Innovation with Integrity Fluorescence Microscopy Opterra Multipoint Scanning Confocal Microscope Superior
Opterra Multipoint Scanning Confocal Microscope Cell-Friendly, High-Speed, High-Resolution Imaging Innovation with Integrity Fluorescence Microscopy Opterra Multipoint Scanning Confocal Microscope Superior
Supplementary Figure S1: Schematic view of the confocal laser scanning STED microscope used for STED-RICS. For a detailed description of our
 Supplementary Figure S1: Schematic view of the confocal laser scanning STED microscope used for STED-RICS. For a detailed description of our home-built STED microscope used for the STED-RICS experiments,
Supplementary Figure S1: Schematic view of the confocal laser scanning STED microscope used for STED-RICS. For a detailed description of our home-built STED microscope used for the STED-RICS experiments,
Opterra II Multipoint Scanning Confocal Microscope. Innovation with Integrity
 Opterra II Multipoint Scanning Confocal Microscope Enabling 4D Live-Cell Fluorescence Imaging through Speed, Sensitivity, Viability and Simplicity Innovation with Integrity Fluorescence Microscopy The
Opterra II Multipoint Scanning Confocal Microscope Enabling 4D Live-Cell Fluorescence Imaging through Speed, Sensitivity, Viability and Simplicity Innovation with Integrity Fluorescence Microscopy The
LSM 710 Confocal Microscope Standard Operation Protocol
 LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
Nikon AZ100. Laser Scanning Macro Confocal Microscope. Jordan Briscoe Adam Fries Kyle Marchuk Kaitlin Corbin. May 2017.
 Nikon AZ100 Laser Scanning Macro Confocal Microscope Jordan Briscoe Adam Fries Kyle Marchuk Kaitlin Corbin May 2017 Contents 1 Introduction 2 2 Hardware - Startup 2 3 Software/Operation 4 3.1 Multidimensional
Nikon AZ100 Laser Scanning Macro Confocal Microscope Jordan Briscoe Adam Fries Kyle Marchuk Kaitlin Corbin May 2017 Contents 1 Introduction 2 2 Hardware - Startup 2 3 Software/Operation 4 3.1 Multidimensional
Zeiss LSM 780 Protocol
 Zeiss LSM 780 Protocol 1) System Startup F Please note the sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time.
Zeiss LSM 780 Protocol 1) System Startup F Please note the sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time.
Training Guide for Leica SP8 Confocal/Multiphoton Microscope
 Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
SPINNING DISK CSU-X1 USER MANUAL
 SPINNING DISK CSU-X1 USER MANUAL Starting the temperature controller... 2 Starting the CO2 controller... 3 Start the spinning disk... 4 Sample observation with the oculars... 5 Spatial sampling, Pixel
SPINNING DISK CSU-X1 USER MANUAL Starting the temperature controller... 2 Starting the CO2 controller... 3 Start the spinning disk... 4 Sample observation with the oculars... 5 Spatial sampling, Pixel
Nikon Instruments Europe
 Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
Leica SP8 Resonant Confocal. Quick-Start Guide
 Leica SP8 Resonant Confocal Quick-Start Guide Contents Start-up Preparing for Imaging Part 1 On the scope Part 2 Software interface Part 3 Heat & CO2 incubation Part 4 Other hardware options Shut-down
Leica SP8 Resonant Confocal Quick-Start Guide Contents Start-up Preparing for Imaging Part 1 On the scope Part 2 Software interface Part 3 Heat & CO2 incubation Part 4 Other hardware options Shut-down
Nikon. King s College London. Imaging Centre. N-SIM guide NIKON IMAGING KING S COLLEGE LONDON
 N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
Imaging Beyond the Basics: Optimizing Settings on the Leica SP8 Confocal
 Imaging Beyond the Basics: Optimizing Settings on the Leica SP8 Confocal Todays Goal: Introduce some additional functionalities of the Leica SP8 confocal HyD vs. PMT detectors Dye Assistant Scanning By
Imaging Beyond the Basics: Optimizing Settings on the Leica SP8 Confocal Todays Goal: Introduce some additional functionalities of the Leica SP8 confocal HyD vs. PMT detectors Dye Assistant Scanning By
3D Optical Motion Analysis of Micro Systems. Heinrich Steger, Polytec GmbH, Waldbronn
 3D Optical Motion Analysis of Micro Systems Heinrich Steger, Polytec GmbH, Waldbronn SEMICON Europe 2012 Outline Needs and Challenges of measuring Micro Structure and MEMS Tools and Applications for optical
3D Optical Motion Analysis of Micro Systems Heinrich Steger, Polytec GmbH, Waldbronn SEMICON Europe 2012 Outline Needs and Challenges of measuring Micro Structure and MEMS Tools and Applications for optical
3D light microscopy techniques
 3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 2D imaging 3D imaging Resolution
3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 2D imaging 3D imaging Resolution
Sizing of nano structures below the diffraction limit using laser scanning microscopy
 Sizing of nano structures below the diffraction limit using laser scanning microscopy JAN BERGSTRAND Master s Thesis Supervisor: Stefan Wennmalm Examiner: Jerker Widengren trita? Abstract The resolution
Sizing of nano structures below the diffraction limit using laser scanning microscopy JAN BERGSTRAND Master s Thesis Supervisor: Stefan Wennmalm Examiner: Jerker Widengren trita? Abstract The resolution
Renishaw InVia Raman microscope
 Laser Spectroscopy Labs Renishaw InVia Raman microscope Operation instructions 1. Turn On the power switch, system power switch is located towards the back of the system on the right hand side. Wait ~10
Laser Spectroscopy Labs Renishaw InVia Raman microscope Operation instructions 1. Turn On the power switch, system power switch is located towards the back of the system on the right hand side. Wait ~10
Supplemental Method Information Zeiss LSM710
 Supplemental Method Information Zeiss LSM710 1 Under the Light Path window set up the confocal for imaging a green dye (Alexa488-EGFP). For example, set up the light path as shown here using the 488 nm
Supplemental Method Information Zeiss LSM710 1 Under the Light Path window set up the confocal for imaging a green dye (Alexa488-EGFP). For example, set up the light path as shown here using the 488 nm
Zeiss 780 Training Notes
 Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 Supplemental correlative nanomanipulation-fluorescence traces probing nascent RNA and fluorescent Mfd during TCR initiation. Supplemental correlative nanomanipulation-fluorescence
Supplementary Figure 1 Supplemental correlative nanomanipulation-fluorescence traces probing nascent RNA and fluorescent Mfd during TCR initiation. Supplemental correlative nanomanipulation-fluorescence
Confocal Microscopy. Kristin Jensen
 Confocal Microscopy Kristin Jensen 17.11.05 References Cell Biological Applications of Confocal Microscopy, Brian Matsumoto, chapter 1 Studying protein dynamics in living cells,, Jennifer Lippincott-Schwartz
Confocal Microscopy Kristin Jensen 17.11.05 References Cell Biological Applications of Confocal Microscopy, Brian Matsumoto, chapter 1 Studying protein dynamics in living cells,, Jennifer Lippincott-Schwartz
1 Set up the confocal light path for imaging a green dye (Alexa488-EGFP). For example, the
 1 Set up the confocal light path for imaging a green dye (Alexa488-EGFP). For example, the light path as shown here using the 488 nm LASER (Laser Unit 1) reflecting off of the 405/488 nm Dichroic mirror
1 Set up the confocal light path for imaging a green dye (Alexa488-EGFP). For example, the light path as shown here using the 488 nm LASER (Laser Unit 1) reflecting off of the 405/488 nm Dichroic mirror
3D light microscopy techniques
 3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
Figure S1 Figure S1. Wild type IgG and FcRn colocalize in APPL+ TCs.
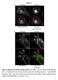 Figure S1 Figure S1. Wild type IgG and FcRn colocalize in APPL+ TCs. HMEC-1 cells were cotransfected with GFP-FcRn and β2m. Transfected cells were incubated with 200 μg/ml Alexa 647-wild type IgG (A) or
Figure S1 Figure S1. Wild type IgG and FcRn colocalize in APPL+ TCs. HMEC-1 cells were cotransfected with GFP-FcRn and β2m. Transfected cells were incubated with 200 μg/ml Alexa 647-wild type IgG (A) or
Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope
 Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Verify that main power switches on the
Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Verify that main power switches on the
Zeiss LSM 880 Protocol
 Zeiss LSM 880 Protocol 1) System Startup Please note put sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time.
Zeiss LSM 880 Protocol 1) System Startup Please note put sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time.
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement
 Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
Bi/BE 227 Winter Assignment #3. Adding the third dimension: 3D Confocal Imaging
 Bi/BE 227 Winter 2016 Assignment #3 Adding the third dimension: 3D Confocal Imaging Schedule: Jan 20: Assignment Jan 20-Feb 8: Work on assignment Feb 10: Student PowerPoint presentations. Goals for this
Bi/BE 227 Winter 2016 Assignment #3 Adding the third dimension: 3D Confocal Imaging Schedule: Jan 20: Assignment Jan 20-Feb 8: Work on assignment Feb 10: Student PowerPoint presentations. Goals for this
Leica TCS SP8 Quick Start Guide
 Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, Fluorescent Light for the microscope stand. 2. Turn on the Scanner Power (1) on the front
Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, Fluorescent Light for the microscope stand. 2. Turn on the Scanner Power (1) on the front
Nature Neuroscience: doi: /nn Supplementary Figure 1. Optimized Bessel foci for in vivo volume imaging.
 Supplementary Figure 1 Optimized Bessel foci for in vivo volume imaging. (a) Images taken by scanning Bessel foci of various NAs, lateral and axial FWHMs: (Left panels) in vivo volume images of YFP + neurites
Supplementary Figure 1 Optimized Bessel foci for in vivo volume imaging. (a) Images taken by scanning Bessel foci of various NAs, lateral and axial FWHMs: (Left panels) in vivo volume images of YFP + neurites
Diskovery Spinning Disk Guide
 Diskovery Spinning Disk Guide qbi.microscopy@uq.edu.au Getting started The microscope and its peripherals (Fig. 1a) should always be turned on, but if they are not, turn them on in the following way: 1.
Diskovery Spinning Disk Guide qbi.microscopy@uq.edu.au Getting started The microscope and its peripherals (Fig. 1a) should always be turned on, but if they are not, turn them on in the following way: 1.
WITec Alpha 300R Quick Operation Summary October 2018
 WITec Alpha 300R Quick Operation Summary October 2018 This document is frequently updated if you feel information should be added, please indicate that to the facility manager (currently Philip Carubia,
WITec Alpha 300R Quick Operation Summary October 2018 This document is frequently updated if you feel information should be added, please indicate that to the facility manager (currently Philip Carubia,
Point Spread Function. Confocal Laser Scanning Microscopy. Confocal Aperture. Optical aberrations. Alternative Scanning Microscopy
 Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
Leica TCS SP8 Quick Start Guide
 Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, EL6000 fluorescent light source for the microscope stand. 2. Turn on the Scanner Power
Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, EL6000 fluorescent light source for the microscope stand. 2. Turn on the Scanner Power
MIF ZEISS VIOLET CONFOCAL ZEN 2009 PROTOCOL
 MIF ZEISS VIOLET CONFOCAL ZEN 2009 PROTOCOL START-UP On the Switchbox, turn both black switches to the ON position. Wait for the microscope to boot up completely (watch the screen on the side of the microscope).
MIF ZEISS VIOLET CONFOCAL ZEN 2009 PROTOCOL START-UP On the Switchbox, turn both black switches to the ON position. Wait for the microscope to boot up completely (watch the screen on the side of the microscope).
Nikon A1R. Multi-Photon & Laser Scanning Confocal Microscope. Kyle Marchuk Adam Fries Jordan Briscoe Kaitlin Corbin. April 2017.
 Nikon A1R Multi-Photon & Laser Scanning Confocal Microscope Kyle Marchuk Adam Fries Jordan Briscoe Kaitlin Corbin April 2017 Contents 1 Introduction 2 2 Start-Up 2 3 Imaging 4 3.1 Sample Alignment...........................................
Nikon A1R Multi-Photon & Laser Scanning Confocal Microscope Kyle Marchuk Adam Fries Jordan Briscoe Kaitlin Corbin April 2017 Contents 1 Introduction 2 2 Start-Up 2 3 Imaging 4 3.1 Sample Alignment...........................................
Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each
 Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each lens with a 1 Airy unit pinhole setting. Many water lenses
Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each lens with a 1 Airy unit pinhole setting. Many water lenses
Imaging Retreat - UMASS Customized real-time confocal and 2-photon imaging
 Imaging Retreat - UMASS 2012 Customized real-time confocal and 2-photon imaging Mike Sanderson Department of Microbiology and Physiological Systems University of Massachusetts Medical School Thanks for
Imaging Retreat - UMASS 2012 Customized real-time confocal and 2-photon imaging Mike Sanderson Department of Microbiology and Physiological Systems University of Massachusetts Medical School Thanks for
(N)MR Imaging. Lab Course Script. FMP PhD Autumn School. Location: C81, MRI Lab B0.03 (basement) Instructor: Leif Schröder. Date: November 3rd, 2010
 (N)MR Imaging Lab Course Script FMP PhD Autumn School Location: C81, MRI Lab B0.03 (basement) Instructor: Leif Schröder Date: November 3rd, 2010 1 Purpose: Understanding the basic principles of MR imaging
(N)MR Imaging Lab Course Script FMP PhD Autumn School Location: C81, MRI Lab B0.03 (basement) Instructor: Leif Schröder Date: November 3rd, 2010 1 Purpose: Understanding the basic principles of MR imaging
Zeiss 880 Training Notes Zen 2.3
 Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
Bi Imaging. Multicolor Imaging: The Important Question of Co-Localization. Anna Smallcombe Bio-Rad Laboratories, Hemel Hempstead, UK
 Multicolor Imaging: The Important Question of Co-Localization Anna Smallcombe Bio-Rad Laboratories, Hemel Hempstead, UK The use of specific fluorescent probes, combined with confocal or multiphoton microscopy
Multicolor Imaging: The Important Question of Co-Localization Anna Smallcombe Bio-Rad Laboratories, Hemel Hempstead, UK The use of specific fluorescent probes, combined with confocal or multiphoton microscopy
Swept-Field User Guide
 Swept-Field User Guide Note: for more details see the Prairie user manual at http://www.prairietechnologies.com/resources/software/prairieview.html Please report any problems to Julie Last (jalast@wisc.edu)
Swept-Field User Guide Note: for more details see the Prairie user manual at http://www.prairietechnologies.com/resources/software/prairieview.html Please report any problems to Julie Last (jalast@wisc.edu)
Laboratory 2: Microscopy and Observation of Cells authors: Dr. Ruth Dahlquist-Willard & Michael Kunz
 Laboratory 2: Microscopy and Observation of Cells authors: Dr. Ruth Dahlquist-Willard & Michael Kunz Corresponding Readings: Campbell Ch. 4 BIOL-100L Safety Information: We will be using laboratory glassware
Laboratory 2: Microscopy and Observation of Cells authors: Dr. Ruth Dahlquist-Willard & Michael Kunz Corresponding Readings: Campbell Ch. 4 BIOL-100L Safety Information: We will be using laboratory glassware
EUV microscopy - a user s perspective Dimitri Scholz EUV,
 EUV microscopy - a user s perspective Dimitri Scholz EUV, 09.11.2011 Imaging technologies: available at UCD now and in the next future Begin ab ovo - Simple approaches direct to the goal - Standard methods
EUV microscopy - a user s perspective Dimitri Scholz EUV, 09.11.2011 Imaging technologies: available at UCD now and in the next future Begin ab ovo - Simple approaches direct to the goal - Standard methods
Point Spread Function Estimation Tool, Alpha Version. A Plugin for ImageJ
 Tutorial Point Spread Function Estimation Tool, Alpha Version A Plugin for ImageJ Benedikt Baumgartner Jo Helmuth jo.helmuth@inf.ethz.ch MOSAIC Lab, ETH Zurich www.mosaic.ethz.ch This tutorial explains
Tutorial Point Spread Function Estimation Tool, Alpha Version A Plugin for ImageJ Benedikt Baumgartner Jo Helmuth jo.helmuth@inf.ethz.ch MOSAIC Lab, ETH Zurich www.mosaic.ethz.ch This tutorial explains
Nikon A1Rsi Confocal Start-Up Sequence
 1. Turn the key on the Nikon LUN-V Laser Launch. Nikon A1Rsi Confocal Start-Up Sequence 2. Press the button the left side of the A1Rsi Controller unit. 3. Turn on the power strip underneath the microscope.
1. Turn the key on the Nikon LUN-V Laser Launch. Nikon A1Rsi Confocal Start-Up Sequence 2. Press the button the left side of the A1Rsi Controller unit. 3. Turn on the power strip underneath the microscope.
Fast Laser Raman Microscope RAMAN
 Fast Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Fast Raman Imaging A New Generation of Raman Microscope RAMAN-11 developed by Nanophoton was created by combining confocal laser microscope technology
Fast Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Fast Raman Imaging A New Generation of Raman Microscope RAMAN-11 developed by Nanophoton was created by combining confocal laser microscope technology
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Sikirzhytski et al., http://www.jcb.org/cgi/content/full/jcb.201401090/dc1 Figure S1. Behavior and organization of K-fibers in PtK 2
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Sikirzhytski et al., http://www.jcb.org/cgi/content/full/jcb.201401090/dc1 Figure S1. Behavior and organization of K-fibers in PtK 2
Title: Nikon A1R Confocal User Manual
 Title: Nikon A1R Confocal User Manual Date of first issue: 23/10/2015 Date of review: Version: Admin For assistance or to report an issue Office: CG.07 or CG.05 Email: Igmm-imaginghelpdesk@igmm.ed.ac.uk
Title: Nikon A1R Confocal User Manual Date of first issue: 23/10/2015 Date of review: Version: Admin For assistance or to report an issue Office: CG.07 or CG.05 Email: Igmm-imaginghelpdesk@igmm.ed.ac.uk
Lab Report 3: Speckle Interferometry LIN PEI-YING, BAIG JOVERIA
 Lab Report 3: Speckle Interferometry LIN PEI-YING, BAIG JOVERIA Abstract: Speckle interferometry (SI) has become a complete technique over the past couple of years and is widely used in many branches of
Lab Report 3: Speckle Interferometry LIN PEI-YING, BAIG JOVERIA Abstract: Speckle interferometry (SI) has become a complete technique over the past couple of years and is widely used in many branches of
Last updated: May 2014 Y.DeGraaf
 FLINDERS MICROSCOPY BIOMEDICAL SERVICES AVAILABLE MICROSCOPES AND SPECIFICATIONS & INFORMATION REGARDING TRAINING FOR NEW USERS Last updated: May 2014 Y.DeGraaf If you have new staff or students (Honours/Masters
FLINDERS MICROSCOPY BIOMEDICAL SERVICES AVAILABLE MICROSCOPES AND SPECIFICATIONS & INFORMATION REGARDING TRAINING FOR NEW USERS Last updated: May 2014 Y.DeGraaf If you have new staff or students (Honours/Masters
Chapter 2 The Study of Microbial Structure: Microscopy and Specimen Preparation
 Chapter 2 The Study of Microbial Structure: Microscopy and Specimen Preparation 1 Lenses and the Bending of Light light is refracted (bent) when passing from one medium to another refractive index a measure
Chapter 2 The Study of Microbial Structure: Microscopy and Specimen Preparation 1 Lenses and the Bending of Light light is refracted (bent) when passing from one medium to another refractive index a measure
Things to check before start-up.
 Byeong Cha Page 1 11/24/2009 Manual for Leica SP2 Confocal Microscope Enter you name, the date, the time, and the account number in the user log book. Things to check before start-up. Make sure that your
Byeong Cha Page 1 11/24/2009 Manual for Leica SP2 Confocal Microscope Enter you name, the date, the time, and the account number in the user log book. Things to check before start-up. Make sure that your
Leica SP8 TCS Users Manual
 Leica SP8 TCS Users Manual Follow the procedure for start up and log on as posted in the lab. Please log on with your account only and do not share your password with anyone. We track and confirm usage
Leica SP8 TCS Users Manual Follow the procedure for start up and log on as posted in the lab. Please log on with your account only and do not share your password with anyone. We track and confirm usage
Overview. About other software. Administrator password. 58. UltraVIEW VoX Getting Started Guide
 Operation 58. UltraVIEW VoX Getting Started Guide Overview This chapter outlines the basic methods used to operate the UltraVIEW VoX system. About other software Volocity places great demands on the computer
Operation 58. UltraVIEW VoX Getting Started Guide Overview This chapter outlines the basic methods used to operate the UltraVIEW VoX system. About other software Volocity places great demands on the computer
The Zeiss AiryScan System, Confocal Four.
 The Zeiss AiryScan System, Confocal Four. Overview. The Zeiss AiryScan module is a segmented, radially stacked GaASP detector and collector system designed to subsample the airy disk of a point emission
The Zeiss AiryScan System, Confocal Four. Overview. The Zeiss AiryScan module is a segmented, radially stacked GaASP detector and collector system designed to subsample the airy disk of a point emission
Improving the Collection Efficiency of Raman Scattering
 PERFORMANCE Unparalleled signal-to-noise ratio with diffraction-limited spectral and imaging resolution Deep-cooled CCD with excelon sensor technology Aberration-free optical design for uniform high resolution
PERFORMANCE Unparalleled signal-to-noise ratio with diffraction-limited spectral and imaging resolution Deep-cooled CCD with excelon sensor technology Aberration-free optical design for uniform high resolution
Using Autofocus in NIS-Elements
 Using Autofocus in NIS-Elements Overview This technical note provides an overview of the available autofocus routines in NIS-Elements, and describes the necessary steps for using the autofocus functions.
Using Autofocus in NIS-Elements Overview This technical note provides an overview of the available autofocus routines in NIS-Elements, and describes the necessary steps for using the autofocus functions.
Prime Scientific CMOS Camera Processing Tools for Super-Resolution Microscopy
 Technical Note Prime Scientific CMOS Camera Processing Tools for Super-Resolution Microscopy Prime Scientific CMOS cameras provide the highest levels of sensitivity which make them ideal for low-light
Technical Note Prime Scientific CMOS Camera Processing Tools for Super-Resolution Microscopy Prime Scientific CMOS cameras provide the highest levels of sensitivity which make them ideal for low-light
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Two-photon (2P) photoconversion is negligible for many popular photoconvertible proteins. a,b, After strong (50 mw average power) illumination, the red channel data (a) shows photoconversion
Supplementary Figure 1 Two-photon (2P) photoconversion is negligible for many popular photoconvertible proteins. a,b, After strong (50 mw average power) illumination, the red channel data (a) shows photoconversion
Thorough Small Angle X-ray Scattering analysis of the instability of liquid micro-jets in air
 Supplementary Information Thorough Small Angle X-ray Scattering analysis of the instability of liquid micro-jets in air Benedetta Marmiroli a *, Fernando Cacho-Nerin a, Barbara Sartori a, Javier Pérez
Supplementary Information Thorough Small Angle X-ray Scattering analysis of the instability of liquid micro-jets in air Benedetta Marmiroli a *, Fernando Cacho-Nerin a, Barbara Sartori a, Javier Pérez
Fast Laser Raman Microscope RAMAN
 Fast Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Fast Raman Imaging A New Generation of Raman Microscope RAMAN-11 developed by Nanophoton was created by combining confocal laser microscope technology
Fast Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Fast Raman Imaging A New Generation of Raman Microscope RAMAN-11 developed by Nanophoton was created by combining confocal laser microscope technology
3. are adherent cells (ie. cells in suspension are too far away from the coverslip)
 Before you begin, make sure your sample... 1. is seeded on #1.5 coverglass (thickness = 0.17) 2. is an aqueous solution (ie. fixed samples mounted on a slide will not work - not enough difference in refractive
Before you begin, make sure your sample... 1. is seeded on #1.5 coverglass (thickness = 0.17) 2. is an aqueous solution (ie. fixed samples mounted on a slide will not work - not enough difference in refractive
RENISHAW INVIA RAMAN SPECTROMETER
 STANDARD OPERATING PROCEDURE: RENISHAW INVIA RAMAN SPECTROMETER Purpose of this Instrument: The Renishaw invia Raman Spectrometer is an instrument used to analyze the Raman scattered light from samples
STANDARD OPERATING PROCEDURE: RENISHAW INVIA RAMAN SPECTROMETER Purpose of this Instrument: The Renishaw invia Raman Spectrometer is an instrument used to analyze the Raman scattered light from samples
Information & Instructions
 KEY FEATURES 1. USB 3.0 For the Fastest Transfer Rates Up to 10X faster than regular USB 2.0 connections (also USB 2.0 compatible) 2. High Resolution 4.2 MegaPixels resolution gives accurate profile measurements
KEY FEATURES 1. USB 3.0 For the Fastest Transfer Rates Up to 10X faster than regular USB 2.0 connections (also USB 2.0 compatible) 2. High Resolution 4.2 MegaPixels resolution gives accurate profile measurements
Training Guide for Carl Zeiss LSM 510 META Confocal Microscope
 Training Guide for Carl Zeiss LSM 510 META Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON Components and System/PC switches
Training Guide for Carl Zeiss LSM 510 META Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON Components and System/PC switches
Laser Speckle Reducer LSR-3000 Series
 Datasheet: LSR-3000 Series Update: 06.08.2012 Copyright 2012 Optotune Laser Speckle Reducer LSR-3000 Series Speckle noise from a laser-based system is reduced by dynamically diffusing the laser beam. A
Datasheet: LSR-3000 Series Update: 06.08.2012 Copyright 2012 Optotune Laser Speckle Reducer LSR-3000 Series Speckle noise from a laser-based system is reduced by dynamically diffusing the laser beam. A
contents TABLE OF The SECOM platform Applications - sections Applications - whole cells Features Integrated workflow Automated overlay
 S E C O M TABLE OF contents The SECOM platform 4 Applications - sections 5 Applications - whole cells 8 Features 9 Integrated workflow 12 Automated overlay ODEMIS - integrated software Specifications 13
S E C O M TABLE OF contents The SECOM platform 4 Applications - sections 5 Applications - whole cells 8 Features 9 Integrated workflow 12 Automated overlay ODEMIS - integrated software Specifications 13
Contents STARTUP MICROSCOPE CONTROLS CAMERA CONTROLS SOFTWARE CONTROLS EXPOSURE AND CONTRAST MONOCHROME IMAGE HANDLING
 Operations Guide Contents STARTUP MICROSCOPE CONTROLS CAMERA CONTROLS SOFTWARE CONTROLS EXPOSURE AND CONTRAST MONOCHROME IMAGE HANDLING Nikon Eclipse 90i Operations Guide STARTUP Startup Powering Up Fluorescence
Operations Guide Contents STARTUP MICROSCOPE CONTROLS CAMERA CONTROLS SOFTWARE CONTROLS EXPOSURE AND CONTRAST MONOCHROME IMAGE HANDLING Nikon Eclipse 90i Operations Guide STARTUP Startup Powering Up Fluorescence
WHITE PAPER FAST PROTEIN INTERACTION BINDING CURVES WITH INO S F-HS CONFOCAL MICROSCOPE
 WHITE PAPER FAST PROTEIN INTERACTION BINDING CURVES WITH INO S F-HS CONFOCAL MICROSCOPE Christian Tardif, Jean-Pierre Bouchard Pascal Gallant, Sebastien Roy, Ozzy Mermut September 2017 Introduction Protein-protein
WHITE PAPER FAST PROTEIN INTERACTION BINDING CURVES WITH INO S F-HS CONFOCAL MICROSCOPE Christian Tardif, Jean-Pierre Bouchard Pascal Gallant, Sebastien Roy, Ozzy Mermut September 2017 Introduction Protein-protein
Compact and Reliable Speckle Reduction
 Compact and Reliable Speckle Reduction August 2017 Mark Ventura, Vice President Sales & Marketing Optotune Switzerland AG Bernstrasse 388 CH-8953 Dietikon Switzerland Phone +41 58 856 3011 www.optotune.com
Compact and Reliable Speckle Reduction August 2017 Mark Ventura, Vice President Sales & Marketing Optotune Switzerland AG Bernstrasse 388 CH-8953 Dietikon Switzerland Phone +41 58 856 3011 www.optotune.com
Nikon Eclipse Ti2-E Widefield/Spinning Disk Confocal Microscope Standard Operation Protocol
 Nikon Eclipse Ti-E Widefield/Spinning Disk Confocal Microscope Standard Operation Protocol Please sign on the log sheet before switching on system. Turn on system Turn on A only if confocal mode or laser
Nikon Eclipse Ti-E Widefield/Spinning Disk Confocal Microscope Standard Operation Protocol Please sign on the log sheet before switching on system. Turn on system Turn on A only if confocal mode or laser
When Failure Analysis Meets Side-Channel Attacks
 When Failure Analysis Meets Side-Channel Attacks Jérôme DI-BATTISTA (THALES), Jean-Christophe COURREGE (THALES), Bruno ROUZEYRE (LIRMM), Lionel TORRES (LIRMM), Philippe PERDU (CNES) Outline Introduction
When Failure Analysis Meets Side-Channel Attacks Jérôme DI-BATTISTA (THALES), Jean-Christophe COURREGE (THALES), Bruno ROUZEYRE (LIRMM), Lionel TORRES (LIRMM), Philippe PERDU (CNES) Outline Introduction
Laser tests of Wide Band Gap power devices. Using Two photon absorption process
 Laser tests of Wide Band Gap power devices Using Two photon absorption process Frederic Darracq Associate professor IMS, CNRS UMR5218, Université Bordeaux, 33405 Talence, France 1 Outline Two-Photon absorption
Laser tests of Wide Band Gap power devices Using Two photon absorption process Frederic Darracq Associate professor IMS, CNRS UMR5218, Université Bordeaux, 33405 Talence, France 1 Outline Two-Photon absorption
Microscope - Exercise 1
 Microscope - Exercise 1 Objectives -Familiarize parts and functions of the microscope. -Calculate total magnifications. -Determining the Diameter of the field of view for different magnifications. -Estimate
Microscope - Exercise 1 Objectives -Familiarize parts and functions of the microscope. -Calculate total magnifications. -Determining the Diameter of the field of view for different magnifications. -Estimate
ScanArray Overview. Principle of Operation. Instrument Components
 ScanArray Overview The GSI Lumonics ScanArrayÒ Microarray Analysis System is a scanning laser confocal fluorescence microscope that is used to determine the fluorescence intensity of a two-dimensional
ScanArray Overview The GSI Lumonics ScanArrayÒ Microarray Analysis System is a scanning laser confocal fluorescence microscope that is used to determine the fluorescence intensity of a two-dimensional
7 CHAPTER 7: REFRACTIVE INDEX MEASUREMENTS WITH COMMON PATH PHASE SENSITIVE FDOCT SETUP
 7 CHAPTER 7: REFRACTIVE INDEX MEASUREMENTS WITH COMMON PATH PHASE SENSITIVE FDOCT SETUP Abstract: In this chapter we describe the use of a common path phase sensitive FDOCT set up. The phase measurements
7 CHAPTER 7: REFRACTIVE INDEX MEASUREMENTS WITH COMMON PATH PHASE SENSITIVE FDOCT SETUP Abstract: In this chapter we describe the use of a common path phase sensitive FDOCT set up. The phase measurements
3 Choose the Channels button and set the Channel Settings. Set the Pinhole to 1 Airy unit.
 1 Set up the confocal light path for imaging a green dye (e.g. Alexa488-EGFP). For example, under the Configuration Control window the light path could be set up as shown here using the 488 nm LASER (found
1 Set up the confocal light path for imaging a green dye (e.g. Alexa488-EGFP). For example, under the Configuration Control window the light path could be set up as shown here using the 488 nm LASER (found
MULTIFOCAL MULTISPECTRAL DESCANNED DETECTION IN TPLSM
 MULTIFOCAL MULTISPECTRAL DESCANNED DETECTION IN TPLSM T. Bergmann a1, M. Tiemann a, J. Martini a,b, K. Tönsing a and D. Anselmetti a a Bielefeld University, Experimental Biophysics & Applied Nanoscience,
MULTIFOCAL MULTISPECTRAL DESCANNED DETECTION IN TPLSM T. Bergmann a1, M. Tiemann a, J. Martini a,b, K. Tönsing a and D. Anselmetti a a Bielefeld University, Experimental Biophysics & Applied Nanoscience,
Quick Guide. LSM 5 MP, LSM 510 and LSM 510 META. Laser Scanning Microscopes. We make it visible. M i c r o s c o p y f r o m C a r l Z e i s s
 LSM 5 MP, LSM 510 and LSM 510 META M i c r o s c o p y f r o m C a r l Z e i s s Quick Guide Laser Scanning Microscopes LSM Software ZEN 2007 August 2007 We make it visible. Contents Page Contents... 1
LSM 5 MP, LSM 510 and LSM 510 META M i c r o s c o p y f r o m C a r l Z e i s s Quick Guide Laser Scanning Microscopes LSM Software ZEN 2007 August 2007 We make it visible. Contents Page Contents... 1
Scanning Ion Conductance Microscope ICnano
 Sperm Cell Epithelial Cells I nner Ear Hair Cells I nner Ear Hair Cell Neurons E- Coli Bac teria Scanning Ion Conductance Microscope ICnano About ionscope About ionscope The ionscope scanning ion conductance
Sperm Cell Epithelial Cells I nner Ear Hair Cells I nner Ear Hair Cell Neurons E- Coli Bac teria Scanning Ion Conductance Microscope ICnano About ionscope About ionscope The ionscope scanning ion conductance
Fast, high-contrast imaging of animal development with scanned light sheet based structured-illumination microscopy
 nature methods Fast, high-contrast imaging of animal development with scanned light sheet based structured-illumination microscopy Philipp J Keller, Annette D Schmidt, Anthony Santella, Khaled Khairy,
nature methods Fast, high-contrast imaging of animal development with scanned light sheet based structured-illumination microscopy Philipp J Keller, Annette D Schmidt, Anthony Santella, Khaled Khairy,
Very short introduction to light microscopy and digital imaging
 Very short introduction to light microscopy and digital imaging Hernan G. Garcia August 1, 2005 1 Light Microscopy Basics In this section we will briefly describe the basic principles of operation and
Very short introduction to light microscopy and digital imaging Hernan G. Garcia August 1, 2005 1 Light Microscopy Basics In this section we will briefly describe the basic principles of operation and
Leica SPEII confocal microscope. Short Manual
 Leica SPEII confocal microscope Short Manual Switching ON sequence: 1. Turn on the Workstation under the bench (top, far right). 2. Turn on the Supply Unit - Laser box (big green switch first and then
Leica SPEII confocal microscope Short Manual Switching ON sequence: 1. Turn on the Workstation under the bench (top, far right). 2. Turn on the Supply Unit - Laser box (big green switch first and then
Evaluation of laser-based active thermography for the inspection of optoelectronic devices
 More info about this article: http://www.ndt.net/?id=15849 Evaluation of laser-based active thermography for the inspection of optoelectronic devices by E. Kollorz, M. Boehnel, S. Mohr, W. Holub, U. Hassler
More info about this article: http://www.ndt.net/?id=15849 Evaluation of laser-based active thermography for the inspection of optoelectronic devices by E. Kollorz, M. Boehnel, S. Mohr, W. Holub, U. Hassler
SUPPLEMENTARY INFORMATION
 Optically reconfigurable metasurfaces and photonic devices based on phase change materials S1: Schematic diagram of the experimental setup. A Ti-Sapphire femtosecond laser (Coherent Chameleon Vision S)
Optically reconfigurable metasurfaces and photonic devices based on phase change materials S1: Schematic diagram of the experimental setup. A Ti-Sapphire femtosecond laser (Coherent Chameleon Vision S)
Megapixel FLIM with bh TCSPC Modules
 Megapixel FLIM with bh TCSPC Modules The New SPCM 64-bit Software Abstract: Becker & Hickl have recently introduced version 9.60 of their SPCM TCSPC data acquisition software. SPCM version 9.60 not only
Megapixel FLIM with bh TCSPC Modules The New SPCM 64-bit Software Abstract: Becker & Hickl have recently introduced version 9.60 of their SPCM TCSPC data acquisition software. SPCM version 9.60 not only
Get the full picture of your sample. Applications
 Follow the Experts Get the full picture of your sample The new generation of confocal Raman microscopes offers a non-destructive and non-contact method of sample analysis at the sub-micron level. More
Follow the Experts Get the full picture of your sample The new generation of confocal Raman microscopes offers a non-destructive and non-contact method of sample analysis at the sub-micron level. More
LSM 800 Confocal Microscope Standard Operation Protocol
 LSM 800 Confocal Microscope Standard Operation Protocol Turning on the system 1. Switch on the Main switch (labeled 1 and 2 ) mounted on the wall. 2. Turn the Laser Key (labeled 3 ) 90 clockwise for power
LSM 800 Confocal Microscope Standard Operation Protocol Turning on the system 1. Switch on the Main switch (labeled 1 and 2 ) mounted on the wall. 2. Turn the Laser Key (labeled 3 ) 90 clockwise for power
Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center
 Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center For any questions or concerns, please contact: Linda Nieman lnieman@mgh.harvard.edu Office: (617) 643-9684 Cell: (512) 565-8076 Chenyue
Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center For any questions or concerns, please contact: Linda Nieman lnieman@mgh.harvard.edu Office: (617) 643-9684 Cell: (512) 565-8076 Chenyue
Nikon C1si Spectral Laser Scanning Confocal Microscope. User Guide
 Nikon C1si Spectral Laser Scanning Confocal Microscope User Guide Contents: C1Si Turn-On/ShutDown Procedures... 2 Overview... 4 Setup for epi-illumination to view through the eyepieces:... 5 Setup for
Nikon C1si Spectral Laser Scanning Confocal Microscope User Guide Contents: C1Si Turn-On/ShutDown Procedures... 2 Overview... 4 Setup for epi-illumination to view through the eyepieces:... 5 Setup for
