Multiphoton confocal microscope. Multiphoton Confocal Microscope A1 MP + /A1R MP +
|
|
|
- Christian Lewis
- 6 years ago
- Views:
Transcription
1 Multiphoton confocal microscope Multiphoton Confocal Microscope A1 MP + /A1R MP +
2 The A1 MP+/A1R MP+ multiphoton confocal microscopes provide faster and sharper imaging from deeper within living organisms, extending the boundaries of traditional research techniques in biological sciences. Ultrahigh-speed imaging up to 420 frames per second (fps) (512 x 32 pixels) with multiphoton imaging using A1R MP+ high efficiency optics and resonant scanner. Deep specimen imaging with high-sensitive non-descanned detectors (NDD) located close to the back aperture of the objective lens. Newly developed ultrasensitive gallium arsenide phosphide (GaAsP) NDD allows much deeper in vivo imaging of mouse brain over 1.2 mm. Auto laser alignment function quickly corrects the IR laser beam shift caused after changing the multiphoton excitation wavelength. The IR laser is coupled to the microscope using a compact Incident Optical Unit that contains an acousto-optic modulator and features autoalignment functions. Compatible with both upright and inverted microscopes. Provides optimum multiphoton imaging configurations for brain research, other neuroscience applications and in vivo imaging of living specimens. In combination with Ni-E Amazingly deep A1 MP + /A1R MP + sharply visualize ultra-deep dynamics within living organisms. In combination with Ti-E In combination with FN1
3 Ultra-deep imaging with the new GaAsP NDD Fast multiphoton imaging, powerful enough for in vivo imaging The new ultrasensitive GaAsP NDD allows clear in vivo imaging in deeper areas than ever before and is powerful enough to analyze the mechanisms, such as brain neurons, of living specimens. Deep brain imaging in in vivo mouse In vivo imaging of an anesthetized YFP-H mouse (4-week-old) via open skull method. Visualization of the entire layer V pyramidal neurons and the deeper hippocampal neurons. Deep imaging achieved for 3-dimensional imaging of hippocampal dendrites over 1.1 mm into the brain. 0mm 0.1mm GaAsP The Nikon resonant scanner is capable of high-speed 420-fps imaging, the world s fastest for a multiphoton microscope using point scanning technology. Unique to this design is a resonant scan mirror capable of imaging full fields of view at much higher speeds than traditional galvano scanners. Nikon's optical pixel clock system, which monitors the position of the resonant mirror in real time, adjusts the pixel clock to ensure more stable, geometrically correct and more evenly illuminated imaging even at high speeds. This enables the successful visualization of in vivo rapid changes, such as reactions in living organisms, dynamics and cell interactions. Visualization of intravital microcirculation Blood cells in blood vessels within a living organism were excited by a femtosecond pulsed IR laser with the A1R MP + ultrahighspeed resonant scanner, and their movements were simultaneously captured in three successive fluorescence images at 30 fps (30 msec), with three separate color channels. The arrowhead indicates the tracking movement of the white blood cell nucleus. Three fluorescent probes are simultaneously excited and imaged nucleus (blue), endothelium (green), and plasma (red). The long-wavelength ultrafast laser in combination with the ultrahigh-speed resonant scanner effectively reduces photodamage and makes time resolved multiphoton imaging of biomolecules possible. 0.2mm 0.3mm 0.4mm Pyramidal cells in layer V Scale bar 20 µm 0.5mm 0.6mm 0.7mm 3.46 sec 3.49 sec 3.52 sec 3.55 sec Image resolution: 512 x 512 pixels, Image acquisition speed: 30 fps, Objective: water immersion objective 60x Dr. Satoshi Nishimura, Department of Cardiovascular Medicine, the University of Tokyo, TSBMI, the University of Tokyo, PRESTO, Japan Science and Technology Agency White matter Scale bar 20 µm 0.8mm 0.9mm Mouse brain in vivo high-speed imaging The cerebral cortex of an anesthetized YFP-H mouse (4-week-old) was studied with the open skull method. SRB (Sulforhodamine B) was injected into the tail vein. Using resonant scanning with episcopic GaAsP NDD, blood flow can be imaged at various deep Z positions. GaAsP 1.0mm Yellow: EYFP pyramidal cells in layer V of the cortex Red: SRB-labeled blood vessels 0mm 1.1mm Blood flow 0.1mm Alveus Scale bar 20 µm 1.2mm 60 fps 0.2mm 15 fps 0.3mm 0.4mm 0.5mm 0.6mm 15 fps 7.5 fps 0.7mm Hippocampal pyramidal cells Scale bar 20 µm Hippocampus 3D zoom image 0.8mm Scale bar 5 µm 0.9mm Captured with episcopic GaAsP NDD and CFI75 Apochromat 25xW MP objective lens (NA 1.10, WD 2.0 mm) Dr. Ryosuke Kawakami, Dr. Terumasa Hibi, Dr. Tomomi Nemoto, Research Institute for Electronic Science, Hokkaido University Dr. Ryosuke Kawakami, Dr. Terumasa Hibi, Dr. Tomomi Nemoto, Research Institute for Electronic Science, Hokkaido University
4 Deep imaging of living specimens with highly efficient standard NDD In vivo image of deep areas of cerebral cortex of a mouse 0.00mm 0.10mm 0.20mm 0.30mm 0.40mm 0.50mm 0.60mm 0.70mm 0.80mm 0.90mm The cerebral cortex of an H-line 5-week-old mouse was studied with the open skull method. The entire shape of dendrites of pyramidal cells in layer V expressing EYFP were visualized from the bottom layer into a superficial layer. In addition, the fluorescence signal of white matter in deeper areas was also studied. Left) 3D reconstruction image Right) Z-stack images Top: dendrites located in superficial layers in the layer V pyramidal cells 25 µm from the surface Middle: basal dendrites in the layer V pyramidal cells 625 µm from the surface Bottom: fluorescence from white matter Excitation wavelength: 930 nm Objective: CFI75 Apochromat 25xW MP (NA 1.10 WD 2.0) Dr. Tomomi Nemoto, Research Institute for Electronic Science, Hokkaido University Dr. Shigenori Nonaka, National Institute for Basic Biology Dr. Takeshi Imamura, Graduate School of Medicine, Ehime University Channel unmixing With multiphoton excitation, fluorophores have a considerably broader profile of the absorption spectra than with single photon excitation. Therefore simultaneous excitation of multiple fluorophores with single excitation wavelength is possible. Additionally, the wavelength of a pulsed laser for multiphoton excitation can be changed and the user can select a suitable and well-balanced wavelength for the excitation of multiple fluorophores. A1 MP+/A1R MP+ NDD and channel unmixing technology enables the user to clearly isolate multiple fluorophores and obtain information on the minute structure of a specimen deep within a living organism. Unmixing with three-color simultaneous excitation Simultaneous imaging of three colors in anesthetized YFP-H mouse with IR excitation of 950 nm The upper four images are acquired original data and the lower four images are unmixed images by utilizing the unmixing function. Blood vessels and neurons are clearly separated. Acquired All channels merged Dura mater Pyramidal neuron Blood vessels Unmixed 1.00mm 1.10mm Mouse cerebral cortex multi-color imaging Simultaneous acquisition of three channels in anesthetized YFP-H mouse using IR excitation of 950 nm and imaging Second Harmonic Generation (SHG) and two fluorescence emissions. Cyan: SHG signal of dura mater Yellow: EYFP pyramidal neurons in layer V of the cortex Red: SRB-labeled blood vessels Dr. Ryosuke Kawakami, Dr. Terumasa Hibi, Dr. Tomomi Nemoto, Research Institute for Electronic Science, Hokkaido University 0mm 0.2mm 0.4mm 0.6mm 0.8mm Unmixing with two-color simultaneous excitation Spinal cord primordia (neural tube) of a 12.5-day-old rat embryo The entire embryo was cultured for approximately 44 hours after transfection of the right and left nerve cells with egfp and YFP (Venus) by electroporation. A cross-sectional slice of spinal cord was embedded in gel and simultaneous excitation of egfp and YFP was conducted using pulsed IR laser (930 nm). The image is captured with NDD and processed by the unmixing function. Observation of interneuron and its commissural axon is clearly achieved. 1.0mm Cyan: SHG signal of dura mater Yellow: EYFP pyramidal neurons in layer V of the cortex Red: SRB-labeled blood vessels Dr. Ryosuke Kawakami, Dr. Terumasa Hibi, Dr. Tomomi Nemoto, Research Institute for Electronic Science, Hokkaido University Dr. Noriko Osumi, Dr. Masanori Takahashi, Division of Developmental Neuroscience, United Center for Advanced Research and Translational Medicine (ART), Tohoku University Graduate School of Medicine
5 Multiphoton imaging gallery Four-color imaging of human colon cancer cells in in vivo Three-dimensional volume rendering of implanted subcutaneous tumor of HCT116 expressing Fucci. The cell cycle of tumor cells and the environment (collagen fiber and vessels) are visualized. Upper right, only collagen fiber and vessels are shown. SHG image of the brain surface of a mouse The neocortex of an H-line 5-week-old mouse was studied with the open skull method. The SHG signals from dura mater and EYFP fluorescence signals were simultaneously acquired using the NDD. Red: Fucci mko2/cancer cell Green: Fucci mag/cancer cell Cyan: SHG/collagen fiber Purple: Qtracker655/neovascular vessels Objective: CFI Plan Fluor 20xA MI Excitation Wavelength: 940 nm Photographed with the cooperation of Dr. Yoshinori Kagawa and Dr. Masaru Ishii, Immunology Frontier Research Center, Osaka University EYFP fluorescent image SHG image of the dura mater overlay image Excitation wavelength: 950 nm Objective: CFI75 Apochromat 25xW MP (NA 1.10 WD 2.0) Dynamic in vivo imaging of granulocytes in live adipose tissues The epididymal adipose tissue of a LysM-EGFP mouse was observed using intravital multiphoton microscopy. Granulocytes patrolling around adipocytes were visualized. Time-lapse images show the movement of the granulocytes. (arrowhead : granulocyte-a, arrow : granulocyte-b) Dr. Takeshi Imamura, Graduate School of Medicine, Ehime University Dr. Yusuke Oshima, Dr. Shigenori Nonaka, National Institute for Basic Biology Dr. Terumasa Hibi, Dr. Ryoshuke Kawakami, Dr. Tomomi Nemoto, Research Institute for Electronic Science, Hokkaido University 3D volume rendering images Three-dimensional volume renderings of a kidney labeled with Hoxb7/myrVenus marker (Chi et al, 2009 Genesis), using depth-code pseudocolor volume rendering to reference Z depths (pseudocolored by depth - 1 µm step for 550 µm). A B Red: BODIPY /fat droplet Green: EGFP /granulocyte Cyan: Hoechst /nucleus and SHG/collagen fiber Photographed with the cooperation of Junichi Kikuta, Shoko Yasuda and Dr. Masaru Ishii, Laboratory of Cellular Dynamics, Immunology Frontier Research Center, Osaka University Objective: CFI Apochromat LWD 40x WI λs Excitation Wavelength: 920 nm Knitted stitch structure of colon wall muscle by SHG imaging NOD/SCID mouse colon wall was observed toward mucosal membrane from serosal membrane side. Knitted stitch structure of colon wall muscle fibers was clearly visualized using SHG. Left, maximum intensity projection calculated from Z stack. Right, three-dimensional volume rendering using depth-code pseudo color. Objective: CFI Apochromat 25xW MP, Scan zoom: 1x, Z step size: 1 µm, IR excitation wavelength: 930 nm Image resolution: 1024x1024 pixels, Image volume: 460 µm (length) x 460 µm (width) x 600 µm (height) Photographed with the cooperation of Dr. Frank Costantini and Dr. Liza Pon, Columbia University Medical Center, New York Ca 2+ signals from the layer V pyramidal neuron SHG of collagen fiber Objective: CFI Plan Fluor 20xA MI Excitation Wavelength: 840 nm Photographed with the cooperation of Dr. Yoshinori Kagawa and Dr. Masaru Ishii, Immunology Frontier Research Center, Osaka University Left, Two-photon image of Alexa 594 fluorescence. Lateral-medial (x axis) and dorsal-ventral (y axis) projections were calculated from 3D stacks. The soma was located at > 500 µm from the surface. Right, Fluorescent change evoked by action potentials. The soma and dendrites were loaded with Oregon Green 488 BAPTA-1 using a patch pipette. The duration of current pulses was 500 ms or 1 s. Photographed with the cooperation of Dr. Satoshi Manita and Dr. Masanori Murayama, Brain Science Institute (BSI), Riken 500 µm 1 s 10% F/F 100 mv 50% F/F 100 V 1 s
6 A1 MP + /A1R MP + achieve the most advanced multiphoton imaging Standard NDD The fluorescence emissions from deep within a specimen are highly scattered in multiphoton excitation, and therefore the conventional detector using a pinhole cannot provide bright fluorescent images. The episcopic NDD in the A1 MP+/A1R MP+ is located close to the back aperture of the objective to detect the maximum amount of scattered emission signals from deep within living specimens. The use of this four-channel detector in combination with special spectral mirrors, together with Nikon s unmixing algorithm, eliminates cross talk between fluorescent probes with highly overlapping emission spectra. Background auto-fluorescence is also eliminated, enabling high-contrast image capture from deep within the specimen. Using diascopic NDD* together with episcopic NDD, brighter images can be acquired by detecting fluorescence signals from both reflected and transmitted. *Compatible with Ni-E focusing nosepiece microscope Nikon s high-na objectives are ideal for multiphoton imaging High-NA objectives have been developed that highly correct chromatic aberrations over a wide wavelength range, from ultraviolet to infrared. Transmission is increased through the use of Nikon s exclusive Nano Crystal Coat technology. In particular, the CFI Apochromat 25xW MP objective lens provides an industry leading highest numerical aperture of 1.10 while still maintaining a 2.0 mm working distance. It also has a collar that corrects chromatic aberrations depending on the depth of the specimen and a 33 manipulator pipette access angle, making it ideal for deep multiphoton imaging and physiology research applications. Nano Crystal Coat is a Nikon exclusive lens coating technology using an ultralow refractive index nanoparticle thin film originally developed for the semiconductor fabrications industry. The Nano Crystal Coat particle structure dramatically reduces stray reflections and boosts transmission over a wide wavelength range, producing images with higher signal-to-noise (S/N) ratios. 4-channel episcopic NDD Objectives CFI75 Apochromat 25xW MP NA 1.10 WD 2.0 Nano Crystal Coat CFI Apochromat LWD 40xWI S NA 1.15 WD 0.6 Nano Crystal Coat CFI Apochromat 40xWI S NA 1.25 WD 0.18 Nano Crystal Coat CFI Plan Apochromat IR 60xWI NA 1.27 WD 0.17 Nano Crystal Coat 4-channel diascopic NDD Auto laser alignment when changing multiphoton excitation wavelength When the multiphoton laser wavelength or group velocity dispersion pre-compensation is changed, the multiphoton laser beam positional pointing at the objective back aperture may also change, resulting in uneven intensity across the image, or a slight misalignment between the IR and visible laser light paths. Super High-sensitive GaAsP NDD The newly developed GaAsP NDD* has approximately twice the sensitivity of a standard NDD and allows clear imaging of deeper areas of living specimens than ever before. Its ability to acquire bright images enables faster imaging and higher quality Z-stack imaging. Its high sensitivity allows acquisition of fluorescent signals with less laser power, resulting in less photo damage to living specimens. * Compatible with FN1 fixed stage microscope Verifying the IR laser beam pointing and setting the alignment has traditionally been difficult. Nikon s A1 MP+ series' auto laser alignment function, housed in the Incident Optical Unit for the multiphoton excitation light path, automatically maximizes IR laser alignments with a single click in NIS-Elements C. Auto laser alignment with a single click
7 Two types of scanning head enable high-speed, high-quality imaging A1 MP+ is equipped with a galvano (non-resonant) scanner for high-resolution imaging. A1R MP+ is a hybrid scanning head that incorporates both galvano and ultrahigh-speed resonant scanners. A1R MP+ allows imaging and photoactivation at ultrafast speeds necessary for revealing cell dynamics and interaction. Simultaneous photoactivation and imaging Simultaneous photoactivation and fluorescence imaging is conducted using galvano and resonant scanners. Because the resonant scanner can capture images at 30 fps, image acquisition of high-speed biological processes after photoactivation is possible. High-resolution imaging The A1 MP+/A1R MP+ galvano scanner enables high-resolution imaging of up to 4096 x 4096 pixels. In addition, with the newly developed scanner driving and sampling systems, plus Nikon s unique image correction technology, high-speed acquisition of 10 fps (512 x 512 pixels) is also possible. 1D scanning 2D scanning Full frame scanning 5,200 lps (lines per second) 130 fps (512 x 32 pixels) 10 fps (512 x 512 pixels) High- speed A1 MP + A1R MP + High- resolution High-speed imaging of photoactivation Imaged at video rate (30 fps) while photo activating the target area with a 405 nm laser 33 ms Generated with Nikon confocal software Points within the cell and changes of fluorescence intensity (From the point closer to the activated point: red, blue and purple) Optical path in the A1R MP + scanning head Ultrafast imaging A1R MP+ is a hybrid scanning head equipped with both a galvano scanner and a resonant scanner with an ultrahigh resonance frequency of 7.8 khz. It allows ultrafast imaging and photoactivation at 420 fps (512 x 32 pixels), the world's fastest image acquisition. 1D scanning 2D scanning Full frame scanning 15,600 lps 420 fps (512 x 32 pixels) 30 fps (512 x 512 pixels) Ultrafast High- speed High- resolution A1R MP + Optical output ports A detector port for the 4-PMT detector, spectral detector port and optional detector port is incorporated. Excitation input ports Up to seven lasers (maximum nine colors) can be loaded. Continuous variable hexagonal pinhole Resonant scanner For high-speed imaging of up to 420 fps (512 x 32 pixels). During simultaneous photoactivation and imaging, the resonant scanner is used for image capture. Resonant Galvano Galvano Low-angle incidence dichroic mirror Stable, ultrafast imaging The Nikon original optical clock generation method is used for high-speed imaging with a resonant scanner. Stable clock pulses are generated optically, offering images that have neither flicker nor distortion even at the highest speed. Galvano scanner For High-quality and high-resolution imaging of up to 4096 x 4096 pixels. High-speed imaging of 10 fps (512 x 512 pixels) is also possible. During simultaneous photoactivation and imaging, the galvano scanner is used for photo stimulation. High-speed data transfer with fiber-optic communication High-speed data transfer with fiber-optic communication The fiber-optic communication data transfer system can transfer data at a maximum of 4 Gbps. This allows the transfer of five channels of image data (512 x 512 pixels, 16 bit) at 30 fps. Wide field of view Resonant scanners do not suffer from overheating of the motor during highspeed image acquisition. Therefore, it is not necessary to reduce the field of view of the scanned image in order to avoid overheating, thus enabling a wide field of view. Wide field of view of resonant scanner Field of view of galvano scanner Photoactivation laser High-speed imaging laser Hyper selector Resonant scanner Imaging Hyper selector Photoactivation What is a hybrid scanner? This mechanism allows flexible switching or simultaneous use of two scanners (resonant and galvano) with the use of a hyper selector. Galvano scanner
8 Key Nikon innovations for improving image quality Enhanced spectral detector The best image quality is achieved by an increased light sensitivity resulting from comprehensive technological innovations in electronics, optics and software. Nikon s original spectral performance is even further enhanced in the A1 MP+ series, allowing high-speed spectral acquisition with a single scan. In addition, advanced functions, including real-time unmixing, are incorporated. Low-angle incidence dichroic mirror creates a 30% increase in fluorescence efficiency With the A1 MP+ series, the industry s first low-angle incidence method is utilized on the dichroic mirrors and a 30% increase of fluorescence efficiency is realized. Conventional 45º incidence angle method Reflection-transmission characteristics have high polarization dependence Low-angle incidence method Reflection-transmission characteristics have lower polarization dependence Transmission rate (%) Increased fluorescence efficiency Low-angle incidence method 45º incidence angle method DEES system High diffraction efficiency is achieved by matching the polarization direction of light entering a grating to the polarizing light beam S. Non-polarized light Polarized beam splitter Polarization rotator P S1 S2 S2 S1 Optical fiber The wavelength resolution is independent of pinhole diameter. Wavelength (nm) Comparison of fluorescence efficiency Brighter images with continuous variable hexagonal pinhole Instead of a continuous variable square pinhole, the industry s first hexagonal pinhole is employed. Higher brightness, equivalent to that of an ideal circular pinhole is achieved while maintaining the confocality. Square pinhole 30% more light Hexagonal pinhole Multiple gratings Wavelength resolution can be varied between 2.5/6/10 nm with three gratings. Each position is precisely controlled for high wavelength reproducibility. 32-channel detector A precisely corrected 32-PMT array detector is used. A three-mobile-shielding mechanism allows simultaneous excitation by up to four lasers. 64% of the area of a circle 83% of the area of a circle High-quality spectral data acquisition DISP improves electrical efficiency Nikon s original Dual Integration Signal Processing (DISP) technology has been implemented in the image processing circuitry to improve electrical efficiency, preventing signal loss while the digitizer processes pixel data and resets. The signal is monitored for the entire pixel time resulting in an extremely high S/N ratio. DISP Integrator (1) Integrator (2) Pixel time Image of a zebrafish labeled with four probes (captured with galvano scanner) Nucleus (blue): Hoechst33342, Pupil (green): GFP, Nerve (yellow): Alexa555, Muscle (red): Alexa647 Dr. Kazuki Horikawa and Prof. Takeharu Nagai, Research Institute for Electronic Science, Hokkaido University Integration Hold Reset Diffraction Efficiency Enhancement System (DEES) With the DEES, non-polarized fluorescence light emitted by the specimen is separated into two polarizing light beams P and S by a polarizing beam splitter. P is then converted by a polarization rotator into S, which has higher diffraction efficiency than P, achieving vastly increased overall diffraction efficiency. Diffraction efficiency (%) Characteristics of grating S polarizing light beam P polarizing light beam Wavelength (nm) 750 (Brightness) High-efficiency fluorescence transmission technology The ends of the fluorescence fibers and detector surfaces use a proprietary antireflective coating to reduce signal loss to a minimum, achieving high optical transmission. Accurate, reliable spectral data: three correction techniques Three correction techniques allow for the acquisition of accurate spectra: interchannel sensitivity correction, which adjusts offset and sensitivity of each channel; spectral sensitivity correction, which adjusts diffraction grating spectral efficiency and detector spectral sensitivity; and correction of spectral transmission of optical devices in scanning heads and microscopes. Pre-correction (Channel) Multi-anode PMT sensitivity correction (Brightness) Post-correction (Channel) Two integrators work in parallel as the optical signal is read to ensure there are no gaps.
9 Intuitive, easy-to-use software for multiphoton imaging NIS-Elements C Acquisition and Analysis software Simple operations common with Nikon confocal microscopes All necessary operations for image capture are displayed in one window. Lasers and detectors for visible laser excitation can be switched simply by selecting fluorescent probe to be used. One-touch switching of high speed resonant scanner and high-resolution galvano (non-resonant) scanner Simultaneous photoactivation with high speed imaging is possible with visible laser excitation. Channel unmixing function Nikon's channel unmixing allows you to obtain emissions from multiple NDD PMTs simultaneously, using one IR excitation wavelength, and unmix overlapping emission spectra. Multiphoton laser Detector for multiphoton emission Image capture mode selector Resonant/galvano scanner switch Sensitivity controller Three color simultaneous fluorescent imaging with 850 nm pulsed IR excitation (left: before unmixing, right: after unmixing) Channel unmixing reduces crosstalk (left: before unmixing, right: after unmixing) Functions for high quality multiphoton imaging Auto laser alignment function The IR laser alignment can be quickly optimized with a single click when changing the multiphoton excitation wavelength Scanning mode controller Z-intensity control function Users can define the laser power and PMT gain to use at different depths in a Z series using the Z intensity control function, so that even when imaging dense and thick specimens, the intensity of the emission is maintained throughout the specimen. External trigger function A1 MP+/A1R MP+ and NIS Elements C support triggering applications. This is effective for synchronizing frame and scanning times with electrophysiology recordings, or to externally trigger the confocal to scan. Principle of multiphoton excitation When two photons are absorbed simultaneously by a single fluorescent molecule (two-photon excitation), the excitation efficiency is proportional to the square of the excitation light intensity. In order to achieve multiphoton excitation, a pulsed beam with high photon density or flux is used. Because the laser beam is delivered in very short (femtosecond) pulses and is converged on a focal point through an objective lens, the probability of simultaneous absorption of two photons becomes high enough to be useful for imaging. In two-photon excitation, the excitation efficiency decreases inversely with the fourth power of the distance from the center of the focal volume. As a result, only fluorescence molecules located within the diffraction-limited focal volume of the objective lens are excited and can emit fluorescence. This principle allows the use of non-descanned detectors (NDD s), where an emission pinhole is not necessary to achieve confocal results. There is less absorption and scattering of near infrared light than visible wavelengths through a specimen so the excitation beam can easily penetrate deep into thick tissue. Because two photon excitation is highly confined to only the diffraction-limited focal volume of the objective lens, the need for a confocal pinhole aperture to block the emitted fluorescence from out of focus plane from reaching the detector is eliminated. Photo damage to a specimen can be minimized, and maximum fluorescence detection is made possible, creating conditions suitable for in vivo imaging of living tissue. The combination of the group velocity dispersion pre-compensation "pre-chirping" system incorporated in the multiphoton laser and the use of the non-descanned detector (NDD) allows fluorescence imaging deeper into a specimen than is possible with standard confocal technique. Confocal (single-photon) microscopy Multiphoton microscopy Excitation area in confocal microscopy and multiphoton microscopy Focal plane Excited level Virtual level Ground level Transition of energy levels of fluorescence molecule
10 System diagram Specifications A1 MP+ A1R MP+ Input/output port 3 laser input ports 4 signal output ports for 4-PMT detector, spectral detector, VAAS (optional), and third-party detector (FCS/FCCS/FLIM) Laser for multiphoton Compatible laser Mai Tai HP/eHP DeepSee (Newport Corp.) microscopy Chameleon Vision II (Coherent Inc.) Modulation Method: AOM (Acousto-Optic Modulator) device Control: power control, return mask, ROI exposure control Incident optics nm, auto alignment Laser for confocal microscopy Compatible laser 405 nm, 440/445 nm, 488 nm, 561/594 nm, 638/640nm, Ar laser (457 nm, 488 nm, 514 nm), HeNe laser (543 nm) (option) Modulation Method: AOTF (Acousto-Optic Tunable Filter) or AOM (Acousto-Optic Modulator) device Control: power control for each wavelength, return mask, ROI exposure control Laser unit Standard: LU4A 4-laser module A or C-LU3EX 3-laser module EX Optional: C-LU3EX 3-laser module EX (when 4-laser module is chosen as standard laser unit) NDD for multiphoton Wavelength nm microscopy Detector 4 PMT Filter cube Filter cubes commonly used for a microscope Recommended filter sets for multiphoton: 492SP, 525/50, 575/25, 629/53, DM458, DM495, DM511, DM560, DM593 Detector type Episcopic NDD (for Ni-E/FN1/Ti-E) Diascopic NDD (for Ni-E) Episcopic GaAsP NDD (for FN1) Standard fluorescence detector Wavelength nm ( nm for multiphoton observation) (option) Detector 4 PMT Femtosecond pulsed lasers When pulsed light of very short duration, typically about 100 femtoseconds, passes through microscope optics (e.g. objective), the pulse is spread out in time on its way to the specimen because of group velocity dispersion, (the variation by wavelength in velocity of the speed of light through glass substrates),causing a reduction of peak power. To prevent the reduction of peak pulse power, Nikon has equipped the femtosecond pulsed lasers for multiphoton microscopy with built-in group velocity dispersion precompensation that restores the original pulse width at the specimen. The parameters of the precompensation have been optimized for Nikon s optical system. This enables bright fluorescence imaging of areas deep within a specimen with minimum laser power. Mai Tai HP/eHP DeepSee, Newport Corp., Spectra-Physics Lasers Division (Nikon specifications) Chameleon Vision II, Coherent Inc. (Nikon specifications) Filter cube Diascopic detector (option) Wavelength nm FOV Detector 6 filter cubes commonly used for a microscope mountable on each of three filter wheels Recommended wavelengths for multiphoton/confocal observation: 450/50, 482/35, 515/30, 525/50, 540/30, 550/49, 585/65, 595/50, 700/75 PMT Square inscribed in a ø18 mm circle Image bit depth 4096 gray intensity levels (12 bit) Scanning head Standard image acquisition Scanner: galvano scanner x2 Pixel size: max x 4096 pixels Scanning speed: Standard mode: 2 fps (512 x 512 pixels, bi-direction), 24 fps (512 x 32 pixels, bi-direction) Fast mode: 10fps (512 x 512 pixels, bi-direction), 130 fps (512 x 32 pixels bi-direction) *1 Zoom: x continuously variable Scanning mode: X-Y, X-T, X-Z, XY rotation, Free line High-speed image acquisition Scanner: resonant scanner (X-axis, resonance frequency 7.8 khz), galvano scanner (Y-axis) Pixel size: max. 512 x 512 pixels Scanning speed: 30 fps (512 x 512 pixels) to 420 fps (512 x 32 pixels), 15,600 lines/sec (line speed) Zoom: 7 steps (1x, 1.5x, 2x, 3x, 4x, 6x, 8x) Scanning mode: X-Y, X-T, X-Z Acquisition method: Standard image acquisition, High-speed image acquisition, Simultaneous photoactivation and image acquisition Dichroic mirror Low-angle incidence method Position: 8 Standard filter: 405/488, 405/488/561, 405/488/561/638, /514/IR, 405/488/543/638, BS20/80, IR, 405/488/561/IR Pinhole µm variable (1st image plane) Spectral detector Wavelength detection range 400 nm-750 nm (400 nm-650 nm with multiphoton microscopy) (with galvano scanner) Number of channels 32 channels (option) Spectral image acquisition speed 4 fps (256 x 256 pixels), 1000 lps Wavelength resolution 80 nm (2.5 nm), 192 nm (6 nm), 320 nm (10 nm) Wavelength range variable in 0.25 nm steps Unmixing High-speed unmixing, Precision unmixing Compatible microscopes ECLIPSE Ti-E inverted microscope, ECLIPSE FN1 fixed stage microscope, ECLIPSE Ni-E upright microscope (focusing nosepiece type and focusing stage type) Z step Ti-E: µm, FN1 stepping motor: 0.05 µm Ni-E: µm Option Motorized XY stage (for Ti-E/Ni-E), High-speed Z stage (for Ti-E), High-speed piezo objective-positioning system (for FN1/Ni-E), VAAS Software Display/image generation 2D analysis, 3D volume rendering/orthogonal, 4D analysis, spectral unmixing Image format Application JP2, JPG, TIFF, BMP, GIF, PNG, ND2, JFF, JTF, AVI, ICS/IDS FRAP, FLIP, FRET, photo activation, three-dimensional time-lapse imaging, multipoint time-lapse imaging, colocalization Control computer OS Microsoft Windows 7 Professional 64 bits SP1 (Japanese version/english version) CPU Intel Xeon X5672 (3.20 GHz/8 MB/1333 MHz/Quad Core) or higher Memory 12 GB (2 GB x 3 + additional 2 GB x3) Hard disk Data transfer Network interface Monitor 300 GB SAS (15,000 rpm) x2, RAID 0 configuration Dedicated data transfer I/F 10/100/1000 Gigabit Ethernet 1600 x 1200 or higher resolution, dual monitor configuration recommended Vibration isolated table 1800 (W) x 1500 (D) mm recommended, or 1500 (W) x 1500 (D) mm *1 Fast mode is compatible with zoom x and scanning modes X-Y and X-T. It is not compatible with Rotation, Free line, CROP, ROI, Spectral imaging, Stimulation, CLEM and FLIM.
11 4-laser Module A, Layout Unit: mm With Ti With FN1 Approx Approx Incident Optical Unit Laser Chiller for Multiphoton Microscopy Incident Optical Unit Laser for Multiphoton Microscopy Laser for Multiphoton Microscopy Laser Controller for Multiphoton Microscopy Laser Chiller for Multiphoton Microscopy Laser Controller for Multiphoton Microscopy 1500 Approx laser Module A, 4-detector Unit, 4-laser Power Source Spectral Detector Unit, Rack Controller Scanning Head Non-Descanned Detector Vibration Isolated Table Approx laser Power Source Rack 4-detector Unit, Spectral Detector Unit, Controller Non-Descanned Detector Scanning Head Vibration Isolated Table PC Monitor PC Monitor Remote Controller Remote Controller Operation conditions Temperature: 20 ºC to 25 ºC (± 1 ºC), with 24-hour air conditioning Humidity: 75 % (RH) or less, with no condensation Completely dark room or light shield for microscope 1003 Approx Power source 120 VAC 6.7 A Multiphoton system (scanner set, laser unit) Multiphoton 220 VAC 3.6 A system 120 VAC 12.2 A Computer unit 220 VAC 6.6 A Lazer Microscope Ar laser (457 nm, 488 nm, 514 nm) Except Ar laser (457 nm, 488 nm, 514 nm) Laser for multiphoton microscopy (laser, water chiller, others) Inverted microscope Ti-E with HUB-A and epi-fluorescence illuminator 120 VAC 12.5 A 220 VAC 6.8 A 120 VAC 2.5 A 220 VAC 1.4 A 120 VAC 19.2 A 220 VAC 10.5 A 120 VAC 4.4 A 220 VAC 2.4 A Specifications and equipment are subject to change without any notice or obligation on the part of the manufacturer. December NIKON CORPORATION TO ENSURE CORRECT USAGE, READ THE CORRESPONDING WARNING MANUALS CAREFULLY BEFORE USING YOUR EQUIPMENT. Monitor images are simulated. Company names and product names appearing in this brochure are their registered trademarks or trademarks. N.B. Export of the products* in this brochure is controlled under the Japanese Foreign Exchange and Foreign Trade Law. Appropriate export procedure shall be required in case of export from Japan. *Products: Hardware and its technical information (including software) Dimensions and weight Scanning head 276 (W) x 163 (H) x 364 (D) mm Approx. 10 kg Incident optical unit (A1-IOU) 363 (W) x 186 (H) x 676(D) mm Approx. 16 kg Controller 360 (W) x 580 (H) x 600 (D) mm Approx. 40 kg 4-detector unit 360 (W) x 199 (H) x (D) mm Approx. 16 kg (approx. 22 kg with VAAS) Spectral detector unit 360 (W) x 323 (H) x 595 (D) mm Approx. 26 kg Episcopic NDD (for Ti-E) 206 (W) x 60 (H) x 262 (D) mm Approx. 5 kg Episcopic NDD(for FN1, Ni-E) 216 (W) x 112 (H) x 425 (D) mm Approx. 10 kg Diascopic NDD (for Ni-E) 301 (W) x 66 (H) x 185 (D) mm Approx. 10 kg 4-laser module 438 (W) x 301 (H) x 690 (D) mm Approx. 43 kg (without laser) 4-laser power source rack 438 (W) x 400 (H) x 800 (D) mm Approx. 20 kg (without laser power source) 3-laser module EX 365 (W) x 133 (H) x 702 (D) mm Approx. 22 kg (without laser) Dimensions exclude projections. The AOTF incorporated into the 4-laser unit and the AOM optionally incorporated into the 3-laser unit are classified as controlled products (including provisions applicable to controlled technology) under foreign exchange and trade control laws. You must obtain government permission and complete all required procedures before exporting this system. 650 NIKON CORPORATION Shin-Yurakucho Bldg., 12-1, Yurakucho 1-chome, Chiyoda-ku, Tokyo , Japan phone: fax: NIKON INSTRUMENTS INC Walt Whitman Road, Melville, N.Y , U.S.A. phone: ; NIKON (within the U.S.A. only) fax: NIKON INSTRUMENTS EUROPE B.V. Laan van Kronenburg 2, 1183 AS Amstelveen, The Netherlands phone: fax: NIKON INSTRUMENTS (SHANGHAI) CO., LTD. CHINA phone: fax: (Beijing branch) phone: fax: (Guangzhou branch) phone: fax: Printed in Japan ( )T NIKON SINGAPORE PTE LTD SINGAPORE phone: fax: NIKON MALAYSIA SDN. BHD. MALAYSIA phone: fax: NIKON INSTRUMENTS KOREA CO., LTD. KOREA phone: fax: NIKON CANADA INC. CANADA phone: fax: NIKON FRANCE S.A.S. FRANCE phone: fax: NIKON GMBH GERMANY phone: fax: NIKON INSTRUMENTS S.p.A. ITALY phone: fax: NIKON AG SWITZERLAND phone: fax: Code No.2CE-SCAH-3 NIKON UK LTD. UNITED KINGDOM phone: fax: NIKON GMBH AUSTRIA AUSTRIA phone: fax: NIKON BELUX BELGIUM phone: fax: This brochure is printed on recycled paper made from 40% used material. En
Multiphoton confocal microscope. Multiphoton confocal microscope A1R MP
 Multiphoton confocal microscope Multiphoton confocal microscope A1R MP Nikon's provides deeper, faster and sharper imaging. The confocal microscope A1R, which has an excellent reputation for its high speed,
Multiphoton confocal microscope Multiphoton confocal microscope A1R MP Nikon's provides deeper, faster and sharper imaging. The confocal microscope A1R, which has an excellent reputation for its high speed,
Multiphoton confocal microscope. Multiphoton confocal microscope A1R MP
 confocal microscope confocal microscope A1R MP A1R MP boosts multiphoton imaging Fast multiphoton imaging, powerful enough for in vivo imaging The A1R MP is capable of high-speed 420-fps imaging, the world's
confocal microscope confocal microscope A1R MP A1R MP boosts multiphoton imaging Fast multiphoton imaging, powerful enough for in vivo imaging The A1R MP is capable of high-speed 420-fps imaging, the world's
Multiphoton confocal microscope. Multiphoton Confocal Microscope A1 MP + /A1R MP +
 Multiphoton confocal microscope Multiphoton Confocal Microscope A1 MP + /A1R MP + The A1 MP+/A1R MP+ multiphoton confocal microscopes provide faster and sharper imaging from deeper within living organisms,
Multiphoton confocal microscope Multiphoton Confocal Microscope A1 MP + /A1R MP + The A1 MP+/A1R MP+ multiphoton confocal microscopes provide faster and sharper imaging from deeper within living organisms,
Multiphoton confocal microscope. Multiphoton Confocal Microscope A1 MP + /A1R MP +
 Multiphoton confocal microscope Multiphoton Confocal Microscope A1 MP + /A1R MP + The A1 MP + /A1R MP + multiphoton confocal microscopes provide faster and sharper imaging from deeper within living organisms,
Multiphoton confocal microscope Multiphoton Confocal Microscope A1 MP + /A1R MP + The A1 MP + /A1R MP + multiphoton confocal microscopes provide faster and sharper imaging from deeper within living organisms,
Confocal Microscope. Confocal Microscope C2
 Confocal Microscope Confocal Microscope C2 Confocal Microscope An essential microscopy laboratory insturument The C2 confocal microscope system comprises a new generation of Nikon confocal instruments
Confocal Microscope Confocal Microscope C2 Confocal Microscope An essential microscopy laboratory insturument The C2 confocal microscope system comprises a new generation of Nikon confocal instruments
Confocal Microscope. Confocal Microscope C2
 Confocal Microscope Confocal Microscope C2 Confocal Microscope An essential microscopy laboratory instrument The C2 confocal microscope system comprises a new generation of Nikon confocal instruments designed
Confocal Microscope Confocal Microscope C2 Confocal Microscope An essential microscopy laboratory instrument The C2 confocal microscope system comprises a new generation of Nikon confocal instruments designed
Multiphoton Confocal Microscope A1 MP+/A1R MP+ Multiphoton Confocal Microscope
 Multiphoton Confocal Microscope A1 MP+/A1R MP+ Multiphoton Confocal Microscope 2 Amazingly deep A1 MP+/A1R MP+ sharply visualizes ultra-deep dynamics within living organisms. The A1 MP+ and A1R MP+ multiphoton
Multiphoton Confocal Microscope A1 MP+/A1R MP+ Multiphoton Confocal Microscope 2 Amazingly deep A1 MP+/A1R MP+ sharply visualizes ultra-deep dynamics within living organisms. The A1 MP+ and A1R MP+ multiphoton
Super High Vertical Resolution Non-Contact 3D Surface Profiler BW-S500/BW-D500 Series
 Super High Vertical Resolution Non-Contact 3D Surface Profiler BW-S500/BW-D500 Series Nikon's proprietary scanning-type optical interference measurement technology achieves 1pm* height resolution. * Height
Super High Vertical Resolution Non-Contact 3D Surface Profiler BW-S500/BW-D500 Series Nikon's proprietary scanning-type optical interference measurement technology achieves 1pm* height resolution. * Height
Upright Microscope. Upright Microscope ECLIPSE Ci-E/Ci-L
 Upright Microscope Upright Microscope ECLIPSE Ci-E/Ci-L Feel the evolution Compact research microscope with superior optical performance Nikon has drawn on its proven optics and mechanical design technologies
Upright Microscope Upright Microscope ECLIPSE Ci-E/Ci-L Feel the evolution Compact research microscope with superior optical performance Nikon has drawn on its proven optics and mechanical design technologies
Confocal Microscope A1+/A1R+ Confocal Microscope
 Confocal Microscope A1+/A1R+ Confocal Microscope 1 Smart Tools for Superior Results Nikon s modular A1+/A1R+ confocal laser scanning microscope system can meet the most demanding imaging requirements with
Confocal Microscope A1+/A1R+ Confocal Microscope 1 Smart Tools for Superior Results Nikon s modular A1+/A1R+ confocal laser scanning microscope system can meet the most demanding imaging requirements with
Confocal Microscope A1+/A1R+ Confocal Microscope
 Confocal Microscope A1+/A1R+ Confocal Microscope AM 1 The ultimate confocal microscope Smart Tools for Superior Results Nikon s modular A1+/A1R+ confocal laser scanning microscope system can meet the most
Confocal Microscope A1+/A1R+ Confocal Microscope AM 1 The ultimate confocal microscope Smart Tools for Superior Results Nikon s modular A1+/A1R+ confocal laser scanning microscope system can meet the most
LSM 510 META in Chang Gung University
 Content LSM 510 META in Chang ung University LSM 510 META 路 理 The features and applications of LSM 510 META 01-09 Introduction of the hardware 10-12 Fluorescence observation in conventional microscope
Content LSM 510 META in Chang ung University LSM 510 META 路 理 The features and applications of LSM 510 META 01-09 Introduction of the hardware 10-12 Fluorescence observation in conventional microscope
Opterra. Multipoint Scanning Confocal Microscope. Innovation with Integrity. Cell-Friendly, High-Speed, High-Resolution Imaging
 Opterra Multipoint Scanning Confocal Microscope Cell-Friendly, High-Speed, High-Resolution Imaging Innovation with Integrity Fluorescence Microscopy Opterra Multipoint Scanning Confocal Microscope Superior
Opterra Multipoint Scanning Confocal Microscope Cell-Friendly, High-Speed, High-Resolution Imaging Innovation with Integrity Fluorescence Microscopy Opterra Multipoint Scanning Confocal Microscope Superior
Confocal Microscope. Confocal Microscope A1 + /A1R +
 Confocal Microscope Confocal Microscope A1 + /A1R + A1 + /A1R + the ultimate confocal microscope 2 3 Capturing high-quality images of cells and molecular events at high speed, Nikon s superior A1 + confocal
Confocal Microscope Confocal Microscope A1 + /A1R + A1 + /A1R + the ultimate confocal microscope 2 3 Capturing high-quality images of cells and molecular events at high speed, Nikon s superior A1 + confocal
Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev. Microscopy course, Michmoret Dec 2005
 Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev Why use confocal microscopy? Principles of the laser scanning confocal microscope. Image resolution. Manipulating the
Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev Why use confocal microscopy? Principles of the laser scanning confocal microscope. Image resolution. Manipulating the
NIS-Elements C (For CONFOCAL MICROSCOPE A1) Instructions (Ver. 4.40)
 M487E 15.4.NF.17 (1/4) *M487EN17* NIS-Elements C (For CONFOCAL MICROSCOPE A1) Instructions (Ver. 4.40) Preface Thank you for purchasing the Nikon products. This instruction manual has been prepared for
M487E 15.4.NF.17 (1/4) *M487EN17* NIS-Elements C (For CONFOCAL MICROSCOPE A1) Instructions (Ver. 4.40) Preface Thank you for purchasing the Nikon products. This instruction manual has been prepared for
TRAINING MANUAL. Multiphoton Microscopy LSM 510 META-NLO
 TRAINING MANUAL Multiphoton Microscopy LSM 510 META-NLO September 2010 Multiphoton Microscopy Training Manual Multiphoton microscopy is only available on the LSM 510 META-NLO system. This system is equipped
TRAINING MANUAL Multiphoton Microscopy LSM 510 META-NLO September 2010 Multiphoton Microscopy Training Manual Multiphoton microscopy is only available on the LSM 510 META-NLO system. This system is equipped
MULTIPHOTON MICROSCOPY. Matyas Molnar Dirk Pacholsky
 MULTIPHOTON MICROSCOPY Matyas Molnar Dirk Pacholsky Information Information given here about 2 Photon microscopy were mainly taken from these sources: Background information on 2-Photon microscopy: http://micro.magnet.fsu.edu/primer/techniques/fluorescence/multiphoton/
MULTIPHOTON MICROSCOPY Matyas Molnar Dirk Pacholsky Information Information given here about 2 Photon microscopy were mainly taken from these sources: Background information on 2-Photon microscopy: http://micro.magnet.fsu.edu/primer/techniques/fluorescence/multiphoton/
Super Resolution Microscope N-SIM E. Super Resolution Microscope
 Super Resolution Microscope N-SIM E Super Resolution Microscope Explore Nano world with Nikon N-SIM E is a streamlined, affordable superresolution system that provides double the resolution of conventional
Super Resolution Microscope N-SIM E Super Resolution Microscope Explore Nano world with Nikon N-SIM E is a streamlined, affordable superresolution system that provides double the resolution of conventional
Confocal Microscope A1 + /A1R + Confocal Microscope
 Confocal Microscope A1 + /A1R + Confocal Microscope 1 The ultimate confocal microscope Capturing high-quality images of cells and molecular events at high speed, Nikon s superior A1+ confocal laser microscope
Confocal Microscope A1 + /A1R + Confocal Microscope 1 The ultimate confocal microscope Capturing high-quality images of cells and molecular events at high speed, Nikon s superior A1+ confocal laser microscope
Multifluorescence The Crosstalk Problem and Its Solution
 Multifluorescence The Crosstalk Problem and Its Solution If a specimen is labeled with more than one fluorochrome, each image channel should only show the emission signal of one of them. If, in a specimen
Multifluorescence The Crosstalk Problem and Its Solution If a specimen is labeled with more than one fluorochrome, each image channel should only show the emission signal of one of them. If, in a specimen
Microscope objectives
 Microscope objectives Our objectives help you focus on yours Nikon is a leader in the development and manufacture of optical and digital imaging technology for advanced science and clinical research. With
Microscope objectives Our objectives help you focus on yours Nikon is a leader in the development and manufacture of optical and digital imaging technology for advanced science and clinical research. With
FEMTOSMART. Benefits. Features
 FEMTOSMART Extremely large space under the objective For in vivo studies Field upgradability Patented imaging technologies Flexible scanning methods Maximal photon collection Elevated, column-based body
FEMTOSMART Extremely large space under the objective For in vivo studies Field upgradability Patented imaging technologies Flexible scanning methods Maximal photon collection Elevated, column-based body
Upright Microscope. Upright Microscope ECLIPSE Ci-E/Ci-L
 Upright Microscope Upright Microscope ECLIPSE Ci-E/Ci-L Feel the evolution Compact research microscope with superior optical performance Nikon has drawn on its proven optics and mechanical design technologies
Upright Microscope Upright Microscope ECLIPSE Ci-E/Ci-L Feel the evolution Compact research microscope with superior optical performance Nikon has drawn on its proven optics and mechanical design technologies
Confocal NEXIV VMZ-K Series. CNC Video Measuring System CONFOCAL NEXIV. VMZ-K Series
 Confocal NEXIV VMZ-K Series CNC Video Measuring System CONFOCAL NEXIV VMZ-K Series 3D FOV Measurements Generated with Confocal Images The Confocal NEXIV VMZ-K series, a ground-breaking multifunctional
Confocal NEXIV VMZ-K Series CNC Video Measuring System CONFOCAL NEXIV VMZ-K Series 3D FOV Measurements Generated with Confocal Images The Confocal NEXIV VMZ-K series, a ground-breaking multifunctional
Maria Smedh, Centre for Cellular Imaging. Maria Smedh, Centre for Cellular Imaging
 Nonlinear microscopy I: Two-photon fluorescence microscopy Multiphoton Microscopy What is multiphoton imaging? Applications Different imaging modes Advantages/disadvantages Scattering of light in thick
Nonlinear microscopy I: Two-photon fluorescence microscopy Multiphoton Microscopy What is multiphoton imaging? Applications Different imaging modes Advantages/disadvantages Scattering of light in thick
Confocal NEXIV VMZ-K Series. CNC Video Measuring System CONFOCAL NEXIV. VMZ-K Series
 Confocal NEXIV VMZ-K Series CNC Video Measuring System CONFOCAL NEXIV VMZ-K Series The VMZ-K series enables microscopic height measurements using various objective lenses, with two models to choose from,
Confocal NEXIV VMZ-K Series CNC Video Measuring System CONFOCAL NEXIV VMZ-K Series The VMZ-K series enables microscopic height measurements using various objective lenses, with two models to choose from,
Practical work no. 3: Confocal Live Cell Microscopy
 Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Shreyash Tandon M.S. III Year
 Shreyash Tandon M.S. III Year 20091015 Confocal microscopy is a powerful tool for generating high-resolution images and 3-D reconstructions of a specimen by using point illumination and a spatial pinhole
Shreyash Tandon M.S. III Year 20091015 Confocal microscopy is a powerful tool for generating high-resolution images and 3-D reconstructions of a specimen by using point illumination and a spatial pinhole
CNC Video Measuring System NEXIV VMZ-K series. CNC Video Measuring System. Confocal Model
 CNC Video Measuring System NEXIV VMZ-K series CNC Video Measuring System Confocal Model D FOV Measurements Generated with s A ground-breaking multifunctional video measuring system developed on the strength
CNC Video Measuring System NEXIV VMZ-K series CNC Video Measuring System Confocal Model D FOV Measurements Generated with s A ground-breaking multifunctional video measuring system developed on the strength
ADVANCED METHODS FOR CONFOCAL MICROSCOPY II. Jean-Yves Chatton Sept. 2006
 ADVANCED METHODS FOR CONFOCAL MICROSCOPY II Jean-Yves Chatton Sept. 2006 Workshop outline Confocal microscopy of living cells and tissues X-Z scanning Time series Bleach: FRAP, photoactivation Emission
ADVANCED METHODS FOR CONFOCAL MICROSCOPY II Jean-Yves Chatton Sept. 2006 Workshop outline Confocal microscopy of living cells and tissues X-Z scanning Time series Bleach: FRAP, photoactivation Emission
Imaging Retreat - UMASS Customized real-time confocal and 2-photon imaging
 Imaging Retreat - UMASS 2012 Customized real-time confocal and 2-photon imaging Mike Sanderson Department of Microbiology and Physiological Systems University of Massachusetts Medical School Thanks for
Imaging Retreat - UMASS 2012 Customized real-time confocal and 2-photon imaging Mike Sanderson Department of Microbiology and Physiological Systems University of Massachusetts Medical School Thanks for
VMZ-K3040 CONFOCAL. Confocal Imaging & Metrology. CNC Video Measuring System. Specifications. Dimensional Diagram
 Specifications Types Type - S Objectives Magnification W.D. Confocal Imaging & Metrology Type - H 3x 7.5x 15x 30x 24mm 5mm 20mm 5mm Confocal optics (Area height measurement) Maximum scan height Field of
Specifications Types Type - S Objectives Magnification W.D. Confocal Imaging & Metrology Type - H 3x 7.5x 15x 30x 24mm 5mm 20mm 5mm Confocal optics (Area height measurement) Maximum scan height Field of
Opterra II Multipoint Scanning Confocal Microscope. Innovation with Integrity
 Opterra II Multipoint Scanning Confocal Microscope Enabling 4D Live-Cell Fluorescence Imaging through Speed, Sensitivity, Viability and Simplicity Innovation with Integrity Fluorescence Microscopy The
Opterra II Multipoint Scanning Confocal Microscope Enabling 4D Live-Cell Fluorescence Imaging through Speed, Sensitivity, Viability and Simplicity Innovation with Integrity Fluorescence Microscopy The
Quality Performance, Innovative Design
 Dimensions Confocal Laser Scanning Biological Microscope Table size (mm): 1400(W) 800(D) * Table is not available from Olympus. Avoid placing the controller directly on the floor. Dimensions / Weight /
Dimensions Confocal Laser Scanning Biological Microscope Table size (mm): 1400(W) 800(D) * Table is not available from Olympus. Avoid placing the controller directly on the floor. Dimensions / Weight /
Multiphoton Detection Unit (MDU)
 Experts in Electrophysiology Microscope Equipment Multiphoton Detection Unit (MDU) For integration with the Scientifica SliceScope Pro motorised microscopy system 2 Multiphoton Detection Unit Two-photon
Experts in Electrophysiology Microscope Equipment Multiphoton Detection Unit (MDU) For integration with the Scientifica SliceScope Pro motorised microscopy system 2 Multiphoton Detection Unit Two-photon
Components of confocal and two-photon microscopes
 Components of confocal and two-photon microscopes Internal training 07/04/2016 A. GRICHINE Platform Optical microscopy Cell imaging, IAB, ISdV Plan Confocal laser scanning microscope o o o Principle Main
Components of confocal and two-photon microscopes Internal training 07/04/2016 A. GRICHINE Platform Optical microscopy Cell imaging, IAB, ISdV Plan Confocal laser scanning microscope o o o Principle Main
Confocal Microscope A1 + /A1R + Confocal Microscope
 Confocal Microscope A1 + /A1R + Confocal Microscope 1 Bring Imaging to Life! Capturing high-quality images of cells and molecular events at high speed, Nikon s superior A1+ confocal laser microscope series,
Confocal Microscope A1 + /A1R + Confocal Microscope 1 Bring Imaging to Life! Capturing high-quality images of cells and molecular events at high speed, Nikon s superior A1+ confocal laser microscope series,
Akinori Mitani and Geoff Weiner BGGN 266 Spring 2013 Non-linear optics final report. Introduction and Background
 Akinori Mitani and Geoff Weiner BGGN 266 Spring 2013 Non-linear optics final report Introduction and Background Two-photon microscopy is a type of fluorescence microscopy using two-photon excitation. It
Akinori Mitani and Geoff Weiner BGGN 266 Spring 2013 Non-linear optics final report Introduction and Background Two-photon microscopy is a type of fluorescence microscopy using two-photon excitation. It
DCS-120. Confocal Scanning FLIM Systems. Based on bh s Multidimensional Megapixel FLIM Technology
 Based on bh s Multidimensional Megapixel FLIM Technology Complete Laser Scanning FLIM Microscopes FLIM Upgrades for Existing Conventional Microscopes Multidimensional TCSPC technique High throughput dual-channel
Based on bh s Multidimensional Megapixel FLIM Technology Complete Laser Scanning FLIM Microscopes FLIM Upgrades for Existing Conventional Microscopes Multidimensional TCSPC technique High throughput dual-channel
Cell culture, observation and image capture three functions incorporated into one single long-term time-lapse imaging system
 BioStation IM New! Time Lapse Imaging System Cell culture, observation and image capture three functions incorporated into one single long-term time-lapse imaging system All-in-one package allows perfect
BioStation IM New! Time Lapse Imaging System Cell culture, observation and image capture three functions incorporated into one single long-term time-lapse imaging system All-in-one package allows perfect
Life Science Instrumentation. New Generation. Light Sheet Fluorescence Microscope. Alph
 Life Science Instrumentation Light Sheet Fluorescence Microscope New Generation Alph Modular Light Sheet Microscope Alpha 3 is a new generation of light sheet fluorescence microscope addressing the needs
Life Science Instrumentation Light Sheet Fluorescence Microscope New Generation Alph Modular Light Sheet Microscope Alpha 3 is a new generation of light sheet fluorescence microscope addressing the needs
BASICS OF CONFOCAL IMAGING (PART I)
 BASICS OF CONFOCAL IMAGING (PART I) INTERNAL COURSE 2012 LIGHT MICROSCOPY Lateral resolution Transmission Fluorescence d min 1.22 NA obj NA cond 0 0 rairy 0.61 NAobj Ernst Abbe Lord Rayleigh Depth of field
BASICS OF CONFOCAL IMAGING (PART I) INTERNAL COURSE 2012 LIGHT MICROSCOPY Lateral resolution Transmission Fluorescence d min 1.22 NA obj NA cond 0 0 rairy 0.61 NAobj Ernst Abbe Lord Rayleigh Depth of field
Training Guide for Leica SP8 Confocal/Multiphoton Microscope
 Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
Basics of confocal imaging (part I)
 Basics of confocal imaging (part I) Swiss Institute of Technology (EPFL) Faculty of Life Sciences Head of BIOIMAGING AND OPTICS BIOP arne.seitz@epfl.ch Lateral resolution BioImaging &Optics Platform Light
Basics of confocal imaging (part I) Swiss Institute of Technology (EPFL) Faculty of Life Sciences Head of BIOIMAGING AND OPTICS BIOP arne.seitz@epfl.ch Lateral resolution BioImaging &Optics Platform Light
Point Spread Function. Confocal Laser Scanning Microscopy. Confocal Aperture. Optical aberrations. Alternative Scanning Microscopy
 Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
Non-Descanned FLIM Detection in Multiphoton Microscopes
 Non-Descanned FLIM Detection in Multiphoton Microscopes Abstract. Multiphoton microscopes use a femtosecond NIR laser to excite fluorescence in the sample. Excitation is performed via a multi-photon absorption
Non-Descanned FLIM Detection in Multiphoton Microscopes Abstract. Multiphoton microscopes use a femtosecond NIR laser to excite fluorescence in the sample. Excitation is performed via a multi-photon absorption
Nature Neuroscience: doi: /nn Supplementary Figure 1. Optimized Bessel foci for in vivo volume imaging.
 Supplementary Figure 1 Optimized Bessel foci for in vivo volume imaging. (a) Images taken by scanning Bessel foci of various NAs, lateral and axial FWHMs: (Left panels) in vivo volume images of YFP + neurites
Supplementary Figure 1 Optimized Bessel foci for in vivo volume imaging. (a) Images taken by scanning Bessel foci of various NAs, lateral and axial FWHMs: (Left panels) in vivo volume images of YFP + neurites
Microscopy from Carl Zeiss
 Microscopy from Carl Zeiss Contents Page Contents... 1 Introduction... 1 Starting the System... 2 Introduction to ZEN Efficient Navigation... 5 Setting up the microscope... 10 Configuring the beam path
Microscopy from Carl Zeiss Contents Page Contents... 1 Introduction... 1 Starting the System... 2 Introduction to ZEN Efficient Navigation... 5 Setting up the microscope... 10 Configuring the beam path
Nikon A1R. Multi-Photon & Laser Scanning Confocal Microscope. Kyle Marchuk Adam Fries Jordan Briscoe Kaitlin Corbin. April 2017.
 Nikon A1R Multi-Photon & Laser Scanning Confocal Microscope Kyle Marchuk Adam Fries Jordan Briscoe Kaitlin Corbin April 2017 Contents 1 Introduction 2 2 Start-Up 2 3 Imaging 4 3.1 Sample Alignment...........................................
Nikon A1R Multi-Photon & Laser Scanning Confocal Microscope Kyle Marchuk Adam Fries Jordan Briscoe Kaitlin Corbin April 2017 Contents 1 Introduction 2 2 Start-Up 2 3 Imaging 4 3.1 Sample Alignment...........................................
Things to check before start-up.
 Byeong Cha Page 1 11/24/2009 Manual for Leica SP2 Confocal Microscope Enter you name, the date, the time, and the account number in the user log book. Things to check before start-up. Make sure that your
Byeong Cha Page 1 11/24/2009 Manual for Leica SP2 Confocal Microscope Enter you name, the date, the time, and the account number in the user log book. Things to check before start-up. Make sure that your
Objectives for biological microscopes. Objectives for biological microscopes
 Objectives for biological microscopes Objectives for biological microscopes The Ultimate in Optical Performance and System Flexibility Nikon had two distinct goals in mind when creating its CFI60 optical
Objectives for biological microscopes Objectives for biological microscopes The Ultimate in Optical Performance and System Flexibility Nikon had two distinct goals in mind when creating its CFI60 optical
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement
 Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
 07 Setting Place a specimen, and select a fluorescence dye. The FV10i automatically selects the most suitable imaging conditions based on the fluorescence dye selection. Set Image mapping menu Just click
07 Setting Place a specimen, and select a fluorescence dye. The FV10i automatically selects the most suitable imaging conditions based on the fluorescence dye selection. Set Image mapping menu Just click
Zeiss 780 Training Notes
 Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
Image Analysis Software. Advanced Solutions for your Imaging World
 Image Analysis Software Advanced Solutions for your Imaging World Nikon offers total software solution covering image capture, archiving, and analysis NIS-Elements is an integrated platform of imaging
Image Analysis Software Advanced Solutions for your Imaging World Nikon offers total software solution covering image capture, archiving, and analysis NIS-Elements is an integrated platform of imaging
Fastest high definition Raman imaging. Fastest Laser Raman Microscope RAMAN
 Fastest high definition Raman imaging Fastest Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Observation A New Generation in Raman Observation RAMAN-11 developed by Nanophoton was newly created by
Fastest high definition Raman imaging Fastest Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Observation A New Generation in Raman Observation RAMAN-11 developed by Nanophoton was newly created by
INTRODUCTION TO MICROSCOPY. Urs Ziegler THE PROBLEM
 INTRODUCTION TO MICROSCOPY Urs Ziegler ziegler@zmb.uzh.ch THE PROBLEM 1 ORGANISMS ARE LARGE LIGHT AND ELECTRONS: ELECTROMAGNETIC WAVES v = Wavelength ( ) Speed (v) Frequency ( ) Amplitude (A) Propagation
INTRODUCTION TO MICROSCOPY Urs Ziegler ziegler@zmb.uzh.ch THE PROBLEM 1 ORGANISMS ARE LARGE LIGHT AND ELECTRONS: ELECTROMAGNETIC WAVES v = Wavelength ( ) Speed (v) Frequency ( ) Amplitude (A) Propagation
Nikon. King s College London. Imaging Centre. N-SIM guide NIKON IMAGING KING S COLLEGE LONDON
 N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
DCS-120. Confocal Scanning FLIM Systems. Based on bh s Multidimensional Megapixel FLIM Technology
 DCS-120 Based on bh s Multidimensional Megapixel FLIM Technology Complete Laser Scanning FLIM Microscopes FLIM Upgrades for Existing Conventional Microscopes FLIM with up to 2048 x 2048 pixels Decay curves
DCS-120 Based on bh s Multidimensional Megapixel FLIM Technology Complete Laser Scanning FLIM Microscopes FLIM Upgrades for Existing Conventional Microscopes FLIM with up to 2048 x 2048 pixels Decay curves
Confocal imaging on the Leica TCS SP8. 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software:
 Confocal imaging on the Leica TCS SP8 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software: 4) Do not touch the microscope while the software is initializing. Choose your options: Turn
Confocal imaging on the Leica TCS SP8 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software: 4) Do not touch the microscope while the software is initializing. Choose your options: Turn
Zeiss 880 Training Notes Zen 2.3
 Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
Spectral Imaging with the Opterra Multipoint Scanning Confocal
 Spectral Imaging with the Opterra Multipoint Scanning Confocal Outline Opterra design overview Scan Modes Light Path Spectral Imaging with Opterra Drosophila larva heart. Opterra Design Overview Supravideo
Spectral Imaging with the Opterra Multipoint Scanning Confocal Outline Opterra design overview Scan Modes Light Path Spectral Imaging with Opterra Drosophila larva heart. Opterra Design Overview Supravideo
Confocal Microscopy. (Increasing contrast and resolu6on using op6cal sec6oning) Lecture 7. November 2017
 Confocal Microscopy (Increasing contrast and resolu6on using op6cal sec6oning) Lecture 7 November 2017 3 Flavours of Microscope Confocal Laser Scanning Problem: Out of Focus Light Spinning disc 2-Photon
Confocal Microscopy (Increasing contrast and resolu6on using op6cal sec6oning) Lecture 7 November 2017 3 Flavours of Microscope Confocal Laser Scanning Problem: Out of Focus Light Spinning disc 2-Photon
Complete support to all your needs
 CNC Video Measuring Systems Ryf AG Bettlachstrasse 2 2540 Grenchen tel 032 654 21 00 fax 032 654 21 09 www.ryfag.ch Complete support to all your needs The perfect answer to all your measurement needs The
CNC Video Measuring Systems Ryf AG Bettlachstrasse 2 2540 Grenchen tel 032 654 21 00 fax 032 654 21 09 www.ryfag.ch Complete support to all your needs The perfect answer to all your measurement needs The
FPD/LSI Inspection Microscopes
 FPD/LSI Inspection Microscopes With improved observation and operation, and environmentally friendlier, the four ECLIPSE models are ideally suited for inspection of large FPD/LSI. For ø300mm wafer/episcopic
FPD/LSI Inspection Microscopes With improved observation and operation, and environmentally friendlier, the four ECLIPSE models are ideally suited for inspection of large FPD/LSI. For ø300mm wafer/episcopic
NeoScope. Simple Operation to 40,000. Table Top SEM. Serving Advanced Technology
 Table Top SEM Simple Operation to 40,000 Serving Advanced Technology From 10 to 40,000 Table Top SEM Notebook PC version Just plug it to a wall outlet after placing it on a table Desktop PC version Option
Table Top SEM Simple Operation to 40,000 Serving Advanced Technology From 10 to 40,000 Table Top SEM Notebook PC version Just plug it to a wall outlet after placing it on a table Desktop PC version Option
Systematic Workflow via Intuitive GUI. Easy operation accomplishes your goals faster than ever.
 Systematic Workflow via Intuitive GUI Easy operation accomplishes your goals faster than ever. 16 With the LEXT OLS4100, observation or measurement begins immediately once the sample is placed on the stage.
Systematic Workflow via Intuitive GUI Easy operation accomplishes your goals faster than ever. 16 With the LEXT OLS4100, observation or measurement begins immediately once the sample is placed on the stage.
Confocal Microscopy. Kristin Jensen
 Confocal Microscopy Kristin Jensen 17.11.05 References Cell Biological Applications of Confocal Microscopy, Brian Matsumoto, chapter 1 Studying protein dynamics in living cells,, Jennifer Lippincott-Schwartz
Confocal Microscopy Kristin Jensen 17.11.05 References Cell Biological Applications of Confocal Microscopy, Brian Matsumoto, chapter 1 Studying protein dynamics in living cells,, Jennifer Lippincott-Schwartz
長庚大學共軛焦顯微鏡課程 長庚大學共軛焦顯微鏡課程. Spot light 長庚大學
 長庚大學共軛焦顯微鏡課程 Spot light 長庚大學共軛焦顯微鏡課程 20071030 長庚大學 Basic principle of Laser Scanning Confocal Microscopy The application of LSM 510 META detector Multiphoton microscopy basic principle and introduction
長庚大學共軛焦顯微鏡課程 Spot light 長庚大學共軛焦顯微鏡課程 20071030 長庚大學 Basic principle of Laser Scanning Confocal Microscopy The application of LSM 510 META detector Multiphoton microscopy basic principle and introduction
MEASUREMENT APPLICATION GUIDE OUTER/INNER
 MEASUREMENT APPLICATION GUIDE OUTER/INNER DIAMETER Measurement I N D E X y Selection Guide P.2 y Measurement Principle P.3 y P.4 y X and Y Axes Synchronous Outer Diameter Measurement P.5 y of a Large Diameter
MEASUREMENT APPLICATION GUIDE OUTER/INNER DIAMETER Measurement I N D E X y Selection Guide P.2 y Measurement Principle P.3 y P.4 y X and Y Axes Synchronous Outer Diameter Measurement P.5 y of a Large Diameter
Nikon C1si Spectral Laser Scanning Confocal Microscope. User Guide
 Nikon C1si Spectral Laser Scanning Confocal Microscope User Guide Contents: C1Si Turn-On/ShutDown Procedures... 2 Overview... 4 Setup for epi-illumination to view through the eyepieces:... 5 Setup for
Nikon C1si Spectral Laser Scanning Confocal Microscope User Guide Contents: C1Si Turn-On/ShutDown Procedures... 2 Overview... 4 Setup for epi-illumination to view through the eyepieces:... 5 Setup for
Dynamic Confocal Imaging of Living Brain. Advantages and risks of multiphoton microscopy in physiology
 Dynamic Confocal Imaging of Living Brain Advantages and risks of multiphoton microscopy in physiology Confocal laser scanning microscopy In conventional optical microscopy focused and out-offocus light
Dynamic Confocal Imaging of Living Brain Advantages and risks of multiphoton microscopy in physiology Confocal laser scanning microscopy In conventional optical microscopy focused and out-offocus light
3D light microscopy techniques
 3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
ZEISS LSM510META confocal manual
 ZEISS LSM510META confocal manual Switching on the system 1) Switch on the Remote Control button located on the table to the right of the microscope. This is the main switch for the whole system including
ZEISS LSM510META confocal manual Switching on the system 1) Switch on the Remote Control button located on the table to the right of the microscope. This is the main switch for the whole system including
Fast Laser Raman Microscope RAMAN
 Fast Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Fast Raman Imaging A New Generation of Raman Microscope RAMAN-11 developed by Nanophoton was created by combining confocal laser microscope technology
Fast Laser Raman Microscope RAMAN - 11 www.nanophoton.jp Fast Raman Imaging A New Generation of Raman Microscope RAMAN-11 developed by Nanophoton was created by combining confocal laser microscope technology
Training Guide for Carl Zeiss LSM 7 MP Multiphoton Microscope
 Training Guide for Carl Zeiss LSM 7 MP Multiphoton Microscope ZEN 2009 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn Chameleon TiS laser key from Standby
Training Guide for Carl Zeiss LSM 7 MP Multiphoton Microscope ZEN 2009 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn Chameleon TiS laser key from Standby
Wafer Loaders for IC Inspection Microscopes NWL200 Series. Wafer Loaders for IC Inspection Microscopes
 Wafer Loaders for IC Inspection Microscopes NWL200 Wafer Loaders for IC Inspection Microscopes Wafer Loaders for IC Inspection Microscopes I Nikon s original technology ensures safe, reliable loading of
Wafer Loaders for IC Inspection Microscopes NWL200 Wafer Loaders for IC Inspection Microscopes Wafer Loaders for IC Inspection Microscopes I Nikon s original technology ensures safe, reliable loading of
Instructions for the Experiment
 Instructions for the Experiment Excitonic States in Atomically Thin Semiconductors 1. Introduction Alongside with electrical measurements, optical measurements are an indispensable tool for the study of
Instructions for the Experiment Excitonic States in Atomically Thin Semiconductors 1. Introduction Alongside with electrical measurements, optical measurements are an indispensable tool for the study of
Biological Microscope. Biological Microscope ECLIPSE E100
 Biological Microscope Biological Microscope ECLIPSE E100 Bright LED illumination, superb optical quality, durability and ergonomic touches, all exceed your expectations High-intensity LED Eco-illumination
Biological Microscope Biological Microscope ECLIPSE E100 Bright LED illumination, superb optical quality, durability and ergonomic touches, all exceed your expectations High-intensity LED Eco-illumination
Working Simultaneously. The Next Level of TIRF Microscopy. cell^tirf Illuminator Motorized Total Internal Reflection Fluorescence
 cell^tirf Illuminator Motorized Total Internal Reflection Fluorescence Four individually aligned illumination beams for simultaneous multi-color TIRF imaging Working Simultaneously The Next Level of TIRF
cell^tirf Illuminator Motorized Total Internal Reflection Fluorescence Four individually aligned illumination beams for simultaneous multi-color TIRF imaging Working Simultaneously The Next Level of TIRF
Nature Methods: doi: /nmeth Supplementary Figure 1. Schematic of 2P-ISIM AO optical setup.
 Supplementary Figure 1 Schematic of 2P-ISIM AO optical setup. Excitation from a femtosecond laser is passed through intensity control and shuttering optics (1/2 λ wave plate, polarizing beam splitting
Supplementary Figure 1 Schematic of 2P-ISIM AO optical setup. Excitation from a femtosecond laser is passed through intensity control and shuttering optics (1/2 λ wave plate, polarizing beam splitting
Leica TCS SP8 Quick Start Guide
 Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, Fluorescent Light for the microscope stand. 2. Turn on the Scanner Power (1) on the front
Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, Fluorescent Light for the microscope stand. 2. Turn on the Scanner Power (1) on the front
Quick Guide. LSM 5 MP, LSM 510 and LSM 510 META. Laser Scanning Microscopes. We make it visible. M i c r o s c o p y f r o m C a r l Z e i s s
 LSM 5 MP, LSM 510 and LSM 510 META M i c r o s c o p y f r o m C a r l Z e i s s Quick Guide Laser Scanning Microscopes LSM Software ZEN 2007 August 2007 We make it visible. Contents Page Contents... 1
LSM 5 MP, LSM 510 and LSM 510 META M i c r o s c o p y f r o m C a r l Z e i s s Quick Guide Laser Scanning Microscopes LSM Software ZEN 2007 August 2007 We make it visible. Contents Page Contents... 1
Fundamentals of Light Microscopy II: Fluorescence, Deconvolution, Confocal, Multiphoton, Spectral microscopy. Integrated Microscopy Course
 Fundamentals of Light Microscopy II: Fluorescence, Deconvolution, Confocal, Multiphoton, Spectral microscopy Integrated Microscopy Course Review Lecture 1: Microscopy Basics Light train Kohler illumination*
Fundamentals of Light Microscopy II: Fluorescence, Deconvolution, Confocal, Multiphoton, Spectral microscopy Integrated Microscopy Course Review Lecture 1: Microscopy Basics Light train Kohler illumination*
Imaging Beyond the Basics: Optimizing Settings on the Leica SP8 Confocal
 Imaging Beyond the Basics: Optimizing Settings on the Leica SP8 Confocal Todays Goal: Introduce some additional functionalities of the Leica SP8 confocal HyD vs. PMT detectors Dye Assistant Scanning By
Imaging Beyond the Basics: Optimizing Settings on the Leica SP8 Confocal Todays Goal: Introduce some additional functionalities of the Leica SP8 confocal HyD vs. PMT detectors Dye Assistant Scanning By
LSM 710 Confocal Microscope Standard Operation Protocol
 LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
Nature Methods: doi: /nmeth Supplementary Figure 1
 . Supplementary Figure 1 Schematics and characterization of our AO two-photon fluorescence microscope. (a) Essential components of our AO two-photon fluorescence microscope: Ti:Sapphire laser; optional
. Supplementary Figure 1 Schematics and characterization of our AO two-photon fluorescence microscope. (a) Essential components of our AO two-photon fluorescence microscope: Ti:Sapphire laser; optional
Guide to Confocal 5. Starting session
 Guide to Confocal 5 Remember that when booking and before starting session you can check for any problems at https://www.bris.ac.uk/biochemistry/uobonly/cif/index.html Starting session Switch on microscope
Guide to Confocal 5 Remember that when booking and before starting session you can check for any problems at https://www.bris.ac.uk/biochemistry/uobonly/cif/index.html Starting session Switch on microscope
Camera Overview. Digital Microscope Cameras for Material Science: Clear Images, Precise Analysis. Digital Cameras for Microscopy
 Digital Cameras for Microscopy Camera Overview For Materials Science Microscopes Digital Microscope Cameras for Material Science: Clear Images, Precise Analysis Passionate about Imaging: Olympus Digital
Digital Cameras for Microscopy Camera Overview For Materials Science Microscopes Digital Microscope Cameras for Material Science: Clear Images, Precise Analysis Passionate about Imaging: Olympus Digital
Contents. Introduction
 Contents Page Contents... 1 Introduction... 1 Starting the System... 2 Introduction to ZEN Efficient Navigation... 5 Setting up the microscope... 10 Configuring the beam path and lasers... 12 Scanning
Contents Page Contents... 1 Introduction... 1 Starting the System... 2 Introduction to ZEN Efficient Navigation... 5 Setting up the microscope... 10 Configuring the beam path and lasers... 12 Scanning
Nikon Instruments Europe
 Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
Camera Overview. Digital Microscope Cameras for Material Science: Clear Images, Precise Analysis. Digital Cameras for Microscopy
 Digital Cameras for Microscopy Camera Overview For Materials Science Microscopes Digital Microscope Cameras for Material Science: Clear Images, Precise Analysis Passionate about Imaging: Olympus Digital
Digital Cameras for Microscopy Camera Overview For Materials Science Microscopes Digital Microscope Cameras for Material Science: Clear Images, Precise Analysis Passionate about Imaging: Olympus Digital
Modes of light microscopy Choosing the appropriate system
 Modes of light microscopy Choosing the appropriate system Wide-field microscopy Confocal microscopy Multi-photon microscopy Wide-field, inverted fluorescence microscope Nikon MicroscopyU Endosome migration
Modes of light microscopy Choosing the appropriate system Wide-field microscopy Confocal microscopy Multi-photon microscopy Wide-field, inverted fluorescence microscope Nikon MicroscopyU Endosome migration
Resolution. Diffraction from apertures limits resolution. Rayleigh criterion θ Rayleigh = 1.22 λ/d 1 peak at 2 nd minimum. θ f D
 Microscopy Outline 1. Resolution and Simple Optical Microscope 2. Contrast enhancement: Dark field, Fluorescence (Chelsea & Peter), Phase Contrast, DIC 3. Newer Methods: Scanning Tunneling microscopy (STM),
Microscopy Outline 1. Resolution and Simple Optical Microscope 2. Contrast enhancement: Dark field, Fluorescence (Chelsea & Peter), Phase Contrast, DIC 3. Newer Methods: Scanning Tunneling microscopy (STM),
Bio 407. Applied microscopy. Introduction into light microscopy. José María Mateos. Center for Microscopy and Image Analysis
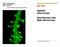 Center for Microscopy and Image Analysis Bio 407 Applied Introduction into light José María Mateos Fundamentals of light Compound microscope Microscope composed of an objective and an additional lens (eyepiece,
Center for Microscopy and Image Analysis Bio 407 Applied Introduction into light José María Mateos Fundamentals of light Compound microscope Microscope composed of an objective and an additional lens (eyepiece,
Invitation for a walk through microscopy. Sebastian Schuchmann Jörg Rösner
 Invitation for a walk through microscopy Sebastian Schuchmann Jörg Rösner joerg.roesner@charite.de Techniques in microscopy Conventional (light) microscopy bright & dark field, phase & interference contrast
Invitation for a walk through microscopy Sebastian Schuchmann Jörg Rösner joerg.roesner@charite.de Techniques in microscopy Conventional (light) microscopy bright & dark field, phase & interference contrast
Digital Microscope ShuttlePix P-400R. Digital Microscope. A new era of microscopy: Shuttle style.
 Digital Microscope ShuttlePix P-400R Digital Microscope new era of microscopy: Shuttle style. ShuttlePix Revolutionary New Portable, Shuttle Style Digital Microscope ShuttlePix Head Handy image capture
Digital Microscope ShuttlePix P-400R Digital Microscope new era of microscopy: Shuttle style. ShuttlePix Revolutionary New Portable, Shuttle Style Digital Microscope ShuttlePix Head Handy image capture
Megapixel FLIM with bh TCSPC Modules
 Megapixel FLIM with bh TCSPC Modules The New SPCM 64-bit Software Abstract: Becker & Hickl have recently introduced version 9.60 of their SPCM TCSPC data acquisition software. SPCM version 9.60 not only
Megapixel FLIM with bh TCSPC Modules The New SPCM 64-bit Software Abstract: Becker & Hickl have recently introduced version 9.60 of their SPCM TCSPC data acquisition software. SPCM version 9.60 not only
Light Microscopy. Upon completion of this lecture, the student should be able to:
 Light Light microscopy is based on the interaction of light and tissue components and can be used to study tissue features. Upon completion of this lecture, the student should be able to: 1- Explain the
Light Light microscopy is based on the interaction of light and tissue components and can be used to study tissue features. Upon completion of this lecture, the student should be able to: 1- Explain the
