Point Spread Function Estimation Tool, Alpha Version. A Plugin for ImageJ
|
|
|
- Wilfrid Felix Payne
- 5 years ago
- Views:
Transcription
1 Tutorial Point Spread Function Estimation Tool, Alpha Version A Plugin for ImageJ Benedikt Baumgartner Jo Helmuth jo.helmuth@inf.ethz.ch MOSAIC Lab, ETH Zurich
2 This tutorial explains the use of the Point Spread Function Estimation Tool, implemented as a plugin for ImageJ. The plugin is still under active development. Your comments on functionality, design, and bugs are highly appreciated. 1 General Description Your microscope setup constitutes an expensive and precious resource from which you want to get the best possible performance. Therefore, you should not rely on purely qualitative performance metrics like it looks pretty good. Your eyes are not capable of telling the difference between a well-adjusted microscope and a poorly adjusted one. In all real equipment, the imaging results are worse than the theoretically possible value (e.g. by a factor of 1.5 in resolution). The reasons for this include imaging noise (prefer images with long exposure!), imperfections of the lenses, distortions caused by variations in cover slide thickness (your objectives are corrected for a specific thickness of slides and they are very sensitive to this), the finite width of the emission wavelength spectrum (we also collect some light at longer wavelengths), point sources of finite extension, under-sampling due to a mismatch of magnification and camera pixel size, and so on. Or you might simply not have focused well enough. D oh! Most likely, many people in your lab share the use of a microscope. You can thus benchmark your results against the ones of your colleges to get a first clue about the quality of your images. The present tool gives you a quantitative way of doing so, enables you to adjust your microscope optimally, and to compare your images to the maximum possible theoretical quality. This is done on the basis of the microscope s Point Spread Function (PSF). The PSF is widely used to describe the imaging properties of optical systems such as light microscopes. Most importantly, the shape of the PSF is a measure for the resolving power of the optical system. In a nutshell, the PSF is the smeared image of a point-like source. The present tool is intended to be used for characterizing light microscopes (of any kind, e.g. fluorescence, wide-field, confocal,...) equipped with a digital camera. With this tool you are able to estimate the PSF of a microscope setup and to compare it to the theoretical PSF as calculated from elementary parameters of the microscope and the camera. This serves two purposes: First, you might want to know whether you adjusted your equipment optimally, and second, the PSF can be used for image restoration purposes (in future versions of the plugin) such as deconvolution. 1
3 Presently, this tool features: user-defined parameters, free selection of images of point sources to be considered for PSF estimation, automatic source position refinement based on intensity centroid estimation, plots of the measured and the theoretical PSF, and a report file containing all relevant input and output. 2 Installation The plugin requires a working installation of ImageJ (version 1.36 or later) on your system. To install the plugin, simply drag the PSF Tool folder into the plugins folder of your local ImageJ installation. After restarting ImageJ, PSF Tool should be visible in the Plugins menu. 3 How to use the Plugin In the following, we will walk through an example in order to describe the main features of the tool and their usage. We have, therefore, attached a sample image to the zip-file, called sample.tif (courtesy of Christoph Burckhardt, Greber lab, University of Zurich). The image shows endosomes and small vesicles in a human cell. While these objects are not strictly point sources, they are still smaller than the resolution limit of the microscope (their size is about 100 nm). For real applications, we strongly encourage you to spend some time on imaging small sources of known size, such as fluorescent beads, virus particles, quantum dots, or whatever is available in your lab. The smaller and brighter the better. Also consider that the sources should not move during exposure. 3.1 Start and Configuration To start, open sample.tif in ImageJ. Currently, the plugin only handles 8-bit gray-scale images so we need to convert the Image. Select Image Type 8-bit in the menu. Now start the plugin. A configuration dialog will appear (see Figure 1). Here, you have to enter necessary microscope, camera, and image parameters on which the calculations will be based. 2
4 Figure 1: Configuration menu Radius of Point Sources (pixel) The apparent radius of the point sources in the image. The parameter is needed for position refinement. In our example, set it to 2. Maximum Sampling Radius (pixel) Determines the radius around the centroid up to which the PSF is sampled. It is important to not set it below the first minimum of the PSF. Be conservative and set it to 4 in our example (which is twice the value of the apparent radius used above). Pixel Size on Camera Chip (µm) Have a look at the data sheet of your microscope or camera. For the tutorial data this parameter is 16. Numerical Aperture of Microscope Objective For the sample image the NA is 1.3. Wavelength of detected light (nm) We imaged green fluorescent proteins and set the value to the emission maximum at 532 nm. Microscope Magnification Factor Overall microscope magnification factor. In our case 100. Click OK after you have entered all parameters. 3.2 Source Selection and PSF Estimation Once you have entered the configuration parameters, a GUI with 4 Buttons is displayed (see Figure 2). 3
5 Figure 2: Graphical User Interface (GUI) Start New Selection Starts a new selection of point sources. Once pressed, every click on the image will result in a PSF estimation (be careful!). Enhance Contrast Enhances Image Contrast by linearly re-scaling all pixel intensity values to the possible range from 0 to 255 using the following equation: (I min (I)) I enhanced = 255 (max (I) min (I)) This does not affect the PSF estimation and is only done to enhance image viewing. Estimate PSF Estimates the Point Spread Function from all selected sources. Create Report Creates a text file containing all relevant result data. Let us now select some single-point sources. To make the selection more convenient, first zoom in to a specific area of the image using the ImageJ Magnifying Glass cursor. Now click on Start New Selection and select some point sources in the image (make sure your cursor is set to single selection again after magnification!). Try to choose apparently small points (the less bright ones might be the smallest) outside noisy areas and without strong background variations and close neighboring sources. Also, make sure not to select spots that contain saturated pixels. This would render your PSF estimate meaningless! Each time you select a source, the position of the source is refined to sub-pixel accuracy (as described in I. F. Sbalzarini and P. Koumoutsakos. Feature Point Tracking and Trajectory Analysis for Video Imaging in Cell Biology, Journal of Structural Biology 151(2): , 2005 ) and a plot will show the estimated PSF of the last selection (see Figure 3). Your selection is also added to the 4
6 Figure 3: Plot of Point Spread Function. The average intensity on concentric circles around the intensity centroid is plotted against the radius. program s GUI and highlighted in the image by a red dot (see Figure 4). Figure 4: Point Source Selection For each selection has its own checkbox in the GUI, stating the refined centroid position of the source and the width of the PSF at half maximum. Using these checkboxes you can select the sources that are to be considered for the overall PSF estimation. To calculate the average PSF click Estimate PSF. A new plot ap- 5
7 pears showing the average PSF. In addition, a theoretical PSF is calculated from the microscope and camera parameters. This allows you to compare the measurement with the theoretical curve. A good measure of comparison is the width at half maximum. The closer the measured PSF is to the theoretical one, the better the performance of your equipment. The theoretical PSF corresponds to the maximum possible resolution permitted by the laws of optics. It is computed from the wavelength λ of the detected light, the microscope s numerical aperture NA, and C defining the intensity value at r = 0. Using the Bessel function J 1 (z), the theoretical PSF is then given by (as described in Y. Iketaki et. al. Theoretical investigation of the point-spread function given by super-resolving fluorescence microscopy using two-color fluorescence dip spectroscopy, Optical Engineering 44(3), 2005 ): PSF (r) = C 2J 1 ( 2π λ NAr) 2π λ NAr Click on Create Report in order to save the PSF data and the configuration parameters to a text file for later use or reference. 2 4 Development and contact The present PSF tool is still under active development. It is currently limited to 2D-PSFs. If you are dealing with image z-stacks, you could either process them slice-by-slice to get an idea of z-resolution (I would not recommend this, however) or wait for a 3D version of our tool. Alternatively, you could make a maximum intensity projection using ImageJ s ZProjection function. Note, however, that you will now measure the effect of your optical system plus the projection! Please send comments, wishes, and reports to jo.helmuth@inf.ethz.ch or benedikt.baumgartner@inf.ethz.ch. 6
ECEN 4606, UNDERGRADUATE OPTICS LAB
 ECEN 4606, UNDERGRADUATE OPTICS LAB Lab 3: Imaging 2 the Microscope Original Version: Professor McLeod SUMMARY: In this lab you will become familiar with the use of one or more lenses to create highly
ECEN 4606, UNDERGRADUATE OPTICS LAB Lab 3: Imaging 2 the Microscope Original Version: Professor McLeod SUMMARY: In this lab you will become familiar with the use of one or more lenses to create highly
Leica DMi8A Quick Guide
 Leica DMi8A Quick Guide 1 Optical Microscope Quick Start Guide The following instructions are provided as a Quick Start Guide for powering up, running measurements, and shutting down Leica s DMi8A Inverted
Leica DMi8A Quick Guide 1 Optical Microscope Quick Start Guide The following instructions are provided as a Quick Start Guide for powering up, running measurements, and shutting down Leica s DMi8A Inverted
Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each
 Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each lens with a 1 Airy unit pinhole setting. Many water lenses
Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each lens with a 1 Airy unit pinhole setting. Many water lenses
Camera Test Protocol. Introduction TABLE OF CONTENTS. Camera Test Protocol Technical Note Technical Note
 Technical Note CMOS, EMCCD AND CCD CAMERAS FOR LIFE SCIENCES Camera Test Protocol Introduction The detector is one of the most important components of any microscope system. Accurate detector readings
Technical Note CMOS, EMCCD AND CCD CAMERAS FOR LIFE SCIENCES Camera Test Protocol Introduction The detector is one of the most important components of any microscope system. Accurate detector readings
Nikon. King s College London. Imaging Centre. N-SIM guide NIKON IMAGING KING S COLLEGE LONDON
 N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
Training Guide for Leica SP8 Confocal/Multiphoton Microscope
 Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
Spatial intensity distribution analysis Matlab user guide
 Spatial intensity distribution analysis Matlab user guide August 2011 Guide on how to use the SpIDA graphical user interface. This little tutorial provides a step by step tutorial explaining how to get
Spatial intensity distribution analysis Matlab user guide August 2011 Guide on how to use the SpIDA graphical user interface. This little tutorial provides a step by step tutorial explaining how to get
User manual for Olympus SD-OSR spinning disk confocal microscope
 User manual for Olympus SD-OSR spinning disk confocal microscope Ved Prakash, PhD. Research imaging specialist Imaging & histology core University of Texas, Dallas ved.prakash@utdallas.edu Once you open
User manual for Olympus SD-OSR spinning disk confocal microscope Ved Prakash, PhD. Research imaging specialist Imaging & histology core University of Texas, Dallas ved.prakash@utdallas.edu Once you open
Optical Performance of Nikon F-Mount Lenses. Landon Carter May 11, Measurement and Instrumentation
 Optical Performance of Nikon F-Mount Lenses Landon Carter May 11, 2016 2.671 Measurement and Instrumentation Abstract In photographic systems, lenses are one of the most important pieces of the system
Optical Performance of Nikon F-Mount Lenses Landon Carter May 11, 2016 2.671 Measurement and Instrumentation Abstract In photographic systems, lenses are one of the most important pieces of the system
MRI Grid. The MRI Grid is a tool in MRI Cell Image Analyzer, that can be used to associate measurements with labeled positions on a board.
 Abstract The is a tool in MRI Cell Image Analyzer, that can be used to associate measurements with labeled positions on a board. Illustration 2: A grid on a binary image. Illustration 1: The interface
Abstract The is a tool in MRI Cell Image Analyzer, that can be used to associate measurements with labeled positions on a board. Illustration 2: A grid on a binary image. Illustration 1: The interface
Zeiss LSM 510 Confocor III Training Notes. Center for Cell Analysis & Modeling
 Zeiss LSM 510 Confocor III Training Notes Center for Cell Analysis & Modeling Confocor 3 Start Up Go to System Module Turn on Main Switch, System/ PC, and Components Switches Do you need the arc lamp?
Zeiss LSM 510 Confocor III Training Notes Center for Cell Analysis & Modeling Confocor 3 Start Up Go to System Module Turn on Main Switch, System/ PC, and Components Switches Do you need the arc lamp?
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 1: THE LIGHT MICROSCOPE
 Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 1: THE LIGHT MICROSCOPE Prior to lab: 1) Read these instructions (p 1-6) 2) Go through the online tutorial, the microscopy pre-lab
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 1: THE LIGHT MICROSCOPE Prior to lab: 1) Read these instructions (p 1-6) 2) Go through the online tutorial, the microscopy pre-lab
Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center
 Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center For any questions or concerns, please contact: Linda Nieman lnieman@mgh.harvard.edu Office: (617) 643-9684 Cell: (512) 565-8076 Chenyue
Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center For any questions or concerns, please contact: Linda Nieman lnieman@mgh.harvard.edu Office: (617) 643-9684 Cell: (512) 565-8076 Chenyue
The Zeiss AiryScan System, Confocal Four.
 The Zeiss AiryScan System, Confocal Four. Overview. The Zeiss AiryScan module is a segmented, radially stacked GaASP detector and collector system designed to subsample the airy disk of a point emission
The Zeiss AiryScan System, Confocal Four. Overview. The Zeiss AiryScan module is a segmented, radially stacked GaASP detector and collector system designed to subsample the airy disk of a point emission
Bi/BE 227 Winter Assignment #3. Adding the third dimension: 3D Confocal Imaging
 Bi/BE 227 Winter 2016 Assignment #3 Adding the third dimension: 3D Confocal Imaging Schedule: Jan 20: Assignment Jan 20-Feb 8: Work on assignment Feb 10: Student PowerPoint presentations. Goals for this
Bi/BE 227 Winter 2016 Assignment #3 Adding the third dimension: 3D Confocal Imaging Schedule: Jan 20: Assignment Jan 20-Feb 8: Work on assignment Feb 10: Student PowerPoint presentations. Goals for this
ScanArray Overview. Principle of Operation. Instrument Components
 ScanArray Overview The GSI Lumonics ScanArrayÒ Microarray Analysis System is a scanning laser confocal fluorescence microscope that is used to determine the fluorescence intensity of a two-dimensional
ScanArray Overview The GSI Lumonics ScanArrayÒ Microarray Analysis System is a scanning laser confocal fluorescence microscope that is used to determine the fluorescence intensity of a two-dimensional
Training Guide for Carl Zeiss LSM 510 META Confocal Microscope
 Training Guide for Carl Zeiss LSM 510 META Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON Components and System/PC switches
Training Guide for Carl Zeiss LSM 510 META Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON Components and System/PC switches
GXCapture 8.1 Instruction Manual
 GT Vision image acquisition, managing and processing software GXCapture 8.1 Instruction Manual Contents of the Instruction Manual GXC is the shortened name used for GXCapture Square brackets are used to
GT Vision image acquisition, managing and processing software GXCapture 8.1 Instruction Manual Contents of the Instruction Manual GXC is the shortened name used for GXCapture Square brackets are used to
Point Spread Function. Confocal Laser Scanning Microscopy. Confocal Aperture. Optical aberrations. Alternative Scanning Microscopy
 Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
3D light microscopy techniques
 3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
Topic 04 What is a digital image?
 Topic 04 What is a digital image? Exercise 4.1 How is the image represented by the computer - Pixels Images can have 2 or 3 spacial dimensions, a time dimensions and a number of colour-channels. An image
Topic 04 What is a digital image? Exercise 4.1 How is the image represented by the computer - Pixels Images can have 2 or 3 spacial dimensions, a time dimensions and a number of colour-channels. An image
Introduction to BioImage Analysis
 Introduction to BioImage Analysis Qi Gao CellNetworks Math-Clinic core facility 22-23.02.2018 MATH- CLINIC Math-Clinic core facility Data analysis services on bioimage analysis & bioinformatics: 1-to-1
Introduction to BioImage Analysis Qi Gao CellNetworks Math-Clinic core facility 22-23.02.2018 MATH- CLINIC Math-Clinic core facility Data analysis services on bioimage analysis & bioinformatics: 1-to-1
What is a digital image?
 Chapter 4 What is a digital image? 4.1 How is the image represented by the computer? Pixels Images can have 2 or 3 spacial dimensions, a time dimensions and a number of colour-channels. An image is a rectilinear
Chapter 4 What is a digital image? 4.1 How is the image represented by the computer? Pixels Images can have 2 or 3 spacial dimensions, a time dimensions and a number of colour-channels. An image is a rectilinear
Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope
 Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Verify that main power switches on the
Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Verify that main power switches on the
Manual. Cell Border Tracker. Jochen Seebach Institut für Anatomie und Vaskuläre Biologie, WWU Münster
 Manual Cell Border Tracker Jochen Seebach Institut für Anatomie und Vaskuläre Biologie, WWU Münster 1 Cell Border Tracker 1. System Requirements The software requires Windows XP operating system or higher
Manual Cell Border Tracker Jochen Seebach Institut für Anatomie und Vaskuläre Biologie, WWU Münster 1 Cell Border Tracker 1. System Requirements The software requires Windows XP operating system or higher
Be aware that there is no universal notation for the various quantities.
 Fourier Optics v2.4 Ray tracing is limited in its ability to describe optics because it ignores the wave properties of light. Diffraction is needed to explain image spatial resolution and contrast and
Fourier Optics v2.4 Ray tracing is limited in its ability to describe optics because it ignores the wave properties of light. Diffraction is needed to explain image spatial resolution and contrast and
LSM 780 Confocal Microscope Standard Operation Protocol
 LSM 780 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Sign on log sheet according to Actual start time 2. Check Compressed Air supply for the air table 3. Switch
LSM 780 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Sign on log sheet according to Actual start time 2. Check Compressed Air supply for the air table 3. Switch
Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center
 Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center For any questions or concerns, please contact: Linda Nieman lnieman@mgh.harvard.edu Office: (617) 643-9684 Cell: (512) 565-8076 Chenyue
Quick Guide for Zeiss 710 Laser Scanning Confocal MGH Cancer Center For any questions or concerns, please contact: Linda Nieman lnieman@mgh.harvard.edu Office: (617) 643-9684 Cell: (512) 565-8076 Chenyue
Nature Protocols: doi: /nprot Supplementary Figure 1. Schematic diagram of Kőhler illumination.
 Supplementary Figure 1 Schematic diagram of Kőhler illumination. The green beam path represents the excitation path and the red represents the emission path. Supplementary Figure 2 Microscope base components
Supplementary Figure 1 Schematic diagram of Kőhler illumination. The green beam path represents the excitation path and the red represents the emission path. Supplementary Figure 2 Microscope base components
1 Co Localization and Working flow with the lsm700
 1 Co Localization and Working flow with the lsm700 Samples -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ BrDU with alexa 488. -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ no BrDU (but with
1 Co Localization and Working flow with the lsm700 Samples -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ BrDU with alexa 488. -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ no BrDU (but with
CHAPTER1: QUICK START...3 CAMERA INSTALLATION... 3 SOFTWARE AND DRIVER INSTALLATION... 3 START TCAPTURE...4 TCAPTURE PARAMETER SETTINGS... 5 CHAPTER2:
 Image acquisition, managing and processing software TCapture Instruction Manual Key to the Instruction Manual TC is shortened name used for TCapture. Help Refer to [Help] >> [About TCapture] menu for software
Image acquisition, managing and processing software TCapture Instruction Manual Key to the Instruction Manual TC is shortened name used for TCapture. Help Refer to [Help] >> [About TCapture] menu for software
Overview. About other software. Administrator password. 58. UltraVIEW VoX Getting Started Guide
 Operation 58. UltraVIEW VoX Getting Started Guide Overview This chapter outlines the basic methods used to operate the UltraVIEW VoX system. About other software Volocity places great demands on the computer
Operation 58. UltraVIEW VoX Getting Started Guide Overview This chapter outlines the basic methods used to operate the UltraVIEW VoX system. About other software Volocity places great demands on the computer
FLUORESCENCE MICROSCOPY. Matyas Molnar and Dirk Pacholsky
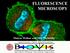 FLUORESCENCE MICROSCOPY Matyas Molnar and Dirk Pacholsky 1 The human eye perceives app. 400-700 nm; best at around 500 nm (green) Has a general resolution down to150-300 μm (human hair: 40-250 μm) We need
FLUORESCENCE MICROSCOPY Matyas Molnar and Dirk Pacholsky 1 The human eye perceives app. 400-700 nm; best at around 500 nm (green) Has a general resolution down to150-300 μm (human hair: 40-250 μm) We need
Introduction to BioImage Analysis using Fiji
 Introduction to BioImage Analysis using Fiji CellNetworks Math-Clinic core facility Qi Gao Carlo A. Beretta 12.05.2017 Math-Clinic core facility Data analysis services on bioinformatics & bioimage analysis:
Introduction to BioImage Analysis using Fiji CellNetworks Math-Clinic core facility Qi Gao Carlo A. Beretta 12.05.2017 Math-Clinic core facility Data analysis services on bioinformatics & bioimage analysis:
Examination, TEN1, in courses SK2500/SK2501, Physics of Biomedical Microscopy,
 KTH Applied Physics Examination, TEN1, in courses SK2500/SK2501, Physics of Biomedical Microscopy, 2009-06-05, 8-13, FB51 Allowed aids: Compendium Imaging Physics (handed out) Compendium Light Microscopy
KTH Applied Physics Examination, TEN1, in courses SK2500/SK2501, Physics of Biomedical Microscopy, 2009-06-05, 8-13, FB51 Allowed aids: Compendium Imaging Physics (handed out) Compendium Light Microscopy
Renishaw InVia Raman microscope
 Laser Spectroscopy Labs Renishaw InVia Raman microscope Operation instructions 1. Turn On the power switch, system power switch is located towards the back of the system on the right hand side. Wait ~10
Laser Spectroscopy Labs Renishaw InVia Raman microscope Operation instructions 1. Turn On the power switch, system power switch is located towards the back of the system on the right hand side. Wait ~10
User Manual. Copyright 2010 Lumos. All rights reserved
 User Manual The contents of this document may not be copied nor duplicated in any form, in whole or in part, without prior written consent from Lumos. Lumos makes no warranties as to the accuracy of the
User Manual The contents of this document may not be copied nor duplicated in any form, in whole or in part, without prior written consent from Lumos. Lumos makes no warranties as to the accuracy of the
LSM 800 Confocal Microscope Standard Operation Protocol
 LSM 800 Confocal Microscope Standard Operation Protocol Turning on the system 1. Switch on the Main switch (labeled 1 and 2 ) mounted on the wall. 2. Turn the Laser Key (labeled 3 ) 90 clockwise for power
LSM 800 Confocal Microscope Standard Operation Protocol Turning on the system 1. Switch on the Main switch (labeled 1 and 2 ) mounted on the wall. 2. Turn the Laser Key (labeled 3 ) 90 clockwise for power
3D light microscopy techniques
 3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 2D imaging 3D imaging Resolution
3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 2D imaging 3D imaging Resolution
Education in Microscopy and Digital Imaging
 Contact Us Carl Zeiss Education in Microscopy and Digital Imaging ZEISS Home Products Solutions Support Online Shop ZEISS International ZEISS Campus Home Interactive Tutorials Basic Microscopy Spectral
Contact Us Carl Zeiss Education in Microscopy and Digital Imaging ZEISS Home Products Solutions Support Online Shop ZEISS International ZEISS Campus Home Interactive Tutorials Basic Microscopy Spectral
LSM 510 Meta Training Notes
 LSM 510 Meta Training Notes Turning on the system Turn on X-Cite power supply. This supplies light for epifluorescence for viewing your samples through the microscope. Turn on the remote control switch.
LSM 510 Meta Training Notes Turning on the system Turn on X-Cite power supply. This supplies light for epifluorescence for viewing your samples through the microscope. Turn on the remote control switch.
Katarina Logg, Kristofer Bodvard, Mikael Käll. Dept. of Applied Physics. 12 September Optical Microscopy. Supervisor s signature:...
 Katarina Logg, Kristofer Bodvard, Mikael Käll Dept. of Applied Physics 12 September 2007 O1 Optical Microscopy Name:.. Date:... Supervisor s signature:... Introduction Over the past decades, the number
Katarina Logg, Kristofer Bodvard, Mikael Käll Dept. of Applied Physics 12 September 2007 O1 Optical Microscopy Name:.. Date:... Supervisor s signature:... Introduction Over the past decades, the number
Swept-Field User Guide
 Swept-Field User Guide Note: for more details see the Prairie user manual at http://www.prairietechnologies.com/resources/software/prairieview.html Please report any problems to Julie Last (jalast@wisc.edu)
Swept-Field User Guide Note: for more details see the Prairie user manual at http://www.prairietechnologies.com/resources/software/prairieview.html Please report any problems to Julie Last (jalast@wisc.edu)
Diffraction at Circular Edges
 Diffraction at Circular Edges References: Equipment: Born and Wolf, Principles of Optics nd ed., Pergamon Press 1964 pp. 395-398. Giles, Robin A., Waves and Optics Simulations:The Consortium for Upper
Diffraction at Circular Edges References: Equipment: Born and Wolf, Principles of Optics nd ed., Pergamon Press 1964 pp. 395-398. Giles, Robin A., Waves and Optics Simulations:The Consortium for Upper
LSM 510 Training Notes
 LSM 510 Training Notes Turning on the system Turn on the arc lamp, found on the bench top left of the microscope. This supplies light for epifluorescence for viewing your samples through the microscope.
LSM 510 Training Notes Turning on the system Turn on the arc lamp, found on the bench top left of the microscope. This supplies light for epifluorescence for viewing your samples through the microscope.
LSM 710 Confocal Microscope Standard Operation Protocol
 LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement
 Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
Things to check before start-up.
 Byeong Cha Page 1 11/24/2009 Manual for Leica SP2 Confocal Microscope Enter you name, the date, the time, and the account number in the user log book. Things to check before start-up. Make sure that your
Byeong Cha Page 1 11/24/2009 Manual for Leica SP2 Confocal Microscope Enter you name, the date, the time, and the account number in the user log book. Things to check before start-up. Make sure that your
Title: Leica SP5 Confocal User Manual
 Title: Leica SP5 Confocal User Manual Date of first issue: 23/10/2015 Date of review: Version: Admin For assistance or to report an issue Office: CG07 or 05 Email: Igmm-imaginghelpdesk@igmm.ed.ac.uk Website:
Title: Leica SP5 Confocal User Manual Date of first issue: 23/10/2015 Date of review: Version: Admin For assistance or to report an issue Office: CG07 or 05 Email: Igmm-imaginghelpdesk@igmm.ed.ac.uk Website:
Practical work no. 3: Confocal Live Cell Microscopy
 Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Single Photon Interference Katelynn Sharma and Garrett West University of Rochester, Institute of Optics, 275 Hutchison Rd. Rochester, NY 14627
 Single Photon Interference Katelynn Sharma and Garrett West University of Rochester, Institute of Optics, 275 Hutchison Rd. Rochester, NY 14627 Abstract: In studying the Mach-Zender interferometer and
Single Photon Interference Katelynn Sharma and Garrett West University of Rochester, Institute of Optics, 275 Hutchison Rd. Rochester, NY 14627 Abstract: In studying the Mach-Zender interferometer and
Bioimage Informatics
 Bioimage Informatics Lecture 5, Spring 01 Fundamentals of Fluorescence Microscopy (II) Bioimage Data Analysis (I): Basic Operations Lecture 5 January 5, 01 1 Outline Performance metrics of a microscope
Bioimage Informatics Lecture 5, Spring 01 Fundamentals of Fluorescence Microscopy (II) Bioimage Data Analysis (I): Basic Operations Lecture 5 January 5, 01 1 Outline Performance metrics of a microscope
Multicolor 4D Fluorescence Microscopy using Ultrathin Bessel Light sheets
 SUPPLEMENTARY MATERIAL Multicolor 4D Fluorescence Microscopy using Ultrathin Bessel Light sheets Teng Zhao, Sze Cheung Lau, Ying Wang, Yumian Su, Hao Wang, Aifang Cheng, Karl Herrup, Nancy Y. Ip, Shengwang
SUPPLEMENTARY MATERIAL Multicolor 4D Fluorescence Microscopy using Ultrathin Bessel Light sheets Teng Zhao, Sze Cheung Lau, Ying Wang, Yumian Su, Hao Wang, Aifang Cheng, Karl Herrup, Nancy Y. Ip, Shengwang
AgilEye Manual Version 2.0 February 28, 2007
 AgilEye Manual Version 2.0 February 28, 2007 1717 Louisiana NE Suite 202 Albuquerque, NM 87110 (505) 268-4742 support@agiloptics.com 2 (505) 268-4742 v. 2.0 February 07, 2007 3 Introduction AgilEye Wavefront
AgilEye Manual Version 2.0 February 28, 2007 1717 Louisiana NE Suite 202 Albuquerque, NM 87110 (505) 268-4742 support@agiloptics.com 2 (505) 268-4742 v. 2.0 February 07, 2007 3 Introduction AgilEye Wavefront
STORM/ PALM ANSWER KEY
 STORM/ PALM ANSWER KEY Phys598BP Spring 2016 University of Illinois at Urbana-Champaign Questions for Lab Report 1. How do you define a resolution in STORM imaging? If you are given a STORM setup, how
STORM/ PALM ANSWER KEY Phys598BP Spring 2016 University of Illinois at Urbana-Champaign Questions for Lab Report 1. How do you define a resolution in STORM imaging? If you are given a STORM setup, how
MICROSCOPY MICROSCOPE TERMINOLOGY
 1 MICROSCOPY Most of the microorganisms that we talk about in this class are too small to be seen with the naked eye. The instruments we will use to visualize these microbes are microscopes. The laboratory
1 MICROSCOPY Most of the microorganisms that we talk about in this class are too small to be seen with the naked eye. The instruments we will use to visualize these microbes are microscopes. The laboratory
Match the microscope structures given in the left column with the statements in the right column that identify or describe them.
 49 Prelab for Name Match the microscope structures given in the left column with the statements in the right column that identify or describe them. Key: a. coarse adjustment knob f. turret or nosepiece
49 Prelab for Name Match the microscope structures given in the left column with the statements in the right column that identify or describe them. Key: a. coarse adjustment knob f. turret or nosepiece
Zeiss Axiovert 135 Fluorescence Microscope Quick Guide / Operations Manual (v. 1.0 February 09)
 University of Chicago Integrated Light Microscopy Core Dr. Vytas Bindokas, Director http://digital.bsd.uchicago.edu By: Christine Labno, Assistant Director Room: AB-129 Phone: 4-9040 Zeiss Axiovert 135
University of Chicago Integrated Light Microscopy Core Dr. Vytas Bindokas, Director http://digital.bsd.uchicago.edu By: Christine Labno, Assistant Director Room: AB-129 Phone: 4-9040 Zeiss Axiovert 135
Microscope anatomy, image formation and resolution
 Microscope anatomy, image formation and resolution Ian Dobbie Buy this book for your lab: D.B. Murphy, "Fundamentals of light microscopy and electronic imaging", ISBN 0-471-25391-X Visit these websites:
Microscope anatomy, image formation and resolution Ian Dobbie Buy this book for your lab: D.B. Murphy, "Fundamentals of light microscopy and electronic imaging", ISBN 0-471-25391-X Visit these websites:
Widefield 1. Switching on
 Widefield 1 Switching on 1. Ignite DG5 lamp - must be switched on first (if previous user has switched off, wait 30 min before igniting) 2. Wait 5s and then turn on the main DG5 controller switch. 3. DG5
Widefield 1 Switching on 1. Ignite DG5 lamp - must be switched on first (if previous user has switched off, wait 30 min before igniting) 2. Wait 5s and then turn on the main DG5 controller switch. 3. DG5
User Guide for TWAIN / DirectX interface for GRYPHAX USB 3.0 cameras
 User Guide for TWAIN / DirectX interface for GRYPHAX USB 3.0 cameras The TWAIN & DirectX driver for PROGRES GRYPHAX USB 3.0 cameras enables user to operate with TWAIN and DirectX supported 3 rd party software
User Guide for TWAIN / DirectX interface for GRYPHAX USB 3.0 cameras The TWAIN & DirectX driver for PROGRES GRYPHAX USB 3.0 cameras enables user to operate with TWAIN and DirectX supported 3 rd party software
Measurement of the Modulation Transfer Function (MTF) of a camera lens. Laboratoire d Enseignement Expérimental (LEnsE)
 Measurement of the Modulation Transfer Function (MTF) of a camera lens Aline Vernier, Baptiste Perrin, Thierry Avignon, Jean Augereau, Lionel Jacubowiez Institut d Optique Graduate School Laboratoire d
Measurement of the Modulation Transfer Function (MTF) of a camera lens Aline Vernier, Baptiste Perrin, Thierry Avignon, Jean Augereau, Lionel Jacubowiez Institut d Optique Graduate School Laboratoire d
Modulation Transfer Function
 Modulation Transfer Function The Modulation Transfer Function (MTF) is a useful tool in system evaluation. t describes if, and how well, different spatial frequencies are transferred from object to image.
Modulation Transfer Function The Modulation Transfer Function (MTF) is a useful tool in system evaluation. t describes if, and how well, different spatial frequencies are transferred from object to image.
Science in the Classroom. Actin Network Architecture can determine Myosin Motor Activity Student Activity
 Science in the Classroom Actin Network Architecture can determine Myosin Motor Activity Student Activity Learning objective In this activity, students will learn how to extract quantitative information
Science in the Classroom Actin Network Architecture can determine Myosin Motor Activity Student Activity Learning objective In this activity, students will learn how to extract quantitative information
CAPTURING IMAGES ON THE HIGH-MAGNIFICATION MICROSCOPE
 University of Virginia ITC Academic Computing Health Sciences CAPTURING IMAGES ON THE HIGH-MAGNIFICATION MICROSCOPE Introduction The Olympus BH-2 microscope in ACHS s microscope lab has objectives from
University of Virginia ITC Academic Computing Health Sciences CAPTURING IMAGES ON THE HIGH-MAGNIFICATION MICROSCOPE Introduction The Olympus BH-2 microscope in ACHS s microscope lab has objectives from
ECEN. Spectroscopy. Lab 8. copy. constituents HOMEWORK PR. Figure. 1. Layout of. of the
 ECEN 4606 Lab 8 Spectroscopy SUMMARY: ROBLEM 1: Pedrotti 3 12-10. In this lab, you will design, build and test an optical spectrum analyzer and use it for both absorption and emission spectroscopy. The
ECEN 4606 Lab 8 Spectroscopy SUMMARY: ROBLEM 1: Pedrotti 3 12-10. In this lab, you will design, build and test an optical spectrum analyzer and use it for both absorption and emission spectroscopy. The
Optika ISview. Image acquisition and processing software. Instruction Manual
 Optika ISview Image acquisition and processing software Instruction Manual Key to the Instruction Manual IS is shortened name used for OptikaISview Square brackets are used to indicate items such as menu
Optika ISview Image acquisition and processing software Instruction Manual Key to the Instruction Manual IS is shortened name used for OptikaISview Square brackets are used to indicate items such as menu
Ph 3455 The Photoelectric Effect
 Ph 3455 The Photoelectric Effect Required background reading Tipler, Llewellyn, section 3-3 Prelab Questions 1. In this experiment you will be using a mercury lamp as the source of photons. At the yellow
Ph 3455 The Photoelectric Effect Required background reading Tipler, Llewellyn, section 3-3 Prelab Questions 1. In this experiment you will be using a mercury lamp as the source of photons. At the yellow
Applications of Optics
 Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 26 Applications of Optics Marilyn Akins, PhD Broome Community College Applications of Optics Many devices are based on the principles of optics
Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 26 Applications of Optics Marilyn Akins, PhD Broome Community College Applications of Optics Many devices are based on the principles of optics
Light Microscopy. Upon completion of this lecture, the student should be able to:
 Light Light microscopy is based on the interaction of light and tissue components and can be used to study tissue features. Upon completion of this lecture, the student should be able to: 1- Explain the
Light Light microscopy is based on the interaction of light and tissue components and can be used to study tissue features. Upon completion of this lecture, the student should be able to: 1- Explain the
Horiba LabRAM ARAMIS Raman Spectrometer Revision /28/2016 Page 1 of 11. Horiba Jobin-Yvon LabRAM Aramis - Raman Spectrometer
 Page 1 of 11 Horiba Jobin-Yvon LabRAM Aramis - Raman Spectrometer The Aramis Raman system is a software selectable multi-wavelength Raman system with mapping capabilities with a 400mm monochromator and
Page 1 of 11 Horiba Jobin-Yvon LabRAM Aramis - Raman Spectrometer The Aramis Raman system is a software selectable multi-wavelength Raman system with mapping capabilities with a 400mm monochromator and
Nikon Instruments Europe
 Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
PHY385H1F Introductory Optics. Practicals Session 7 Studying for Test 2
 PHY385H1F Introductory Optics Practicals Session 7 Studying for Test 2 Entrance Pupil & Exit Pupil A Cooke-triplet consists of three thin lenses in succession, and is often used in cameras. It was patented
PHY385H1F Introductory Optics Practicals Session 7 Studying for Test 2 Entrance Pupil & Exit Pupil A Cooke-triplet consists of three thin lenses in succession, and is often used in cameras. It was patented
Microscopy. CS/CME/BioE/Biophys/BMI 279 Nov. 2, 2017 Ron Dror
 Microscopy CS/CME/BioE/Biophys/BMI 279 Nov. 2, 2017 Ron Dror 1 Outline Microscopy: the basics Fluorescence microscopy Resolution limits The diffraction limit Beating the diffraction limit 2 Microscopy:
Microscopy CS/CME/BioE/Biophys/BMI 279 Nov. 2, 2017 Ron Dror 1 Outline Microscopy: the basics Fluorescence microscopy Resolution limits The diffraction limit Beating the diffraction limit 2 Microscopy:
Digital Camera Technologies for Scientific Bio-Imaging. Part 2: Sampling and Signal
 Digital Camera Technologies for Scientific Bio-Imaging. Part 2: Sampling and Signal Yashvinder Sabharwal, 1 James Joubert 2 and Deepak Sharma 2 1. Solexis Advisors LLC, Austin, TX, USA 2. Photometrics
Digital Camera Technologies for Scientific Bio-Imaging. Part 2: Sampling and Signal Yashvinder Sabharwal, 1 James Joubert 2 and Deepak Sharma 2 1. Solexis Advisors LLC, Austin, TX, USA 2. Photometrics
Training Guide for Carl Zeiss LSM 880 with AiryScan FAST
 Training Guide for Carl Zeiss LSM 880 with AiryScan FAST ZEN 2.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2018) Power ON Routine 1 2 Turn ON Main Switch from the remote control
Training Guide for Carl Zeiss LSM 880 with AiryScan FAST ZEN 2.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2018) Power ON Routine 1 2 Turn ON Main Switch from the remote control
Supplementary Figure S1: Schematic view of the confocal laser scanning STED microscope used for STED-RICS. For a detailed description of our
 Supplementary Figure S1: Schematic view of the confocal laser scanning STED microscope used for STED-RICS. For a detailed description of our home-built STED microscope used for the STED-RICS experiments,
Supplementary Figure S1: Schematic view of the confocal laser scanning STED microscope used for STED-RICS. For a detailed description of our home-built STED microscope used for the STED-RICS experiments,
Cell Biology and Bioimaging Core
 Cell Biology and Bioimaging Core Leica TCS SP5 Operating Instructions Starting up the instrument 1. First, log in the log book located on the confocal desk. Include your name, your lab s PI, an account
Cell Biology and Bioimaging Core Leica TCS SP5 Operating Instructions Starting up the instrument 1. First, log in the log book located on the confocal desk. Include your name, your lab s PI, an account
VISUAL PHYSICS ONLINE DEPTH STUDY: ELECTRON MICROSCOPES
 VISUAL PHYSICS ONLINE DEPTH STUDY: ELECTRON MICROSCOPES Shortly after the experimental confirmation of the wave properties of the electron, it was suggested that the electron could be used to examine objects
VISUAL PHYSICS ONLINE DEPTH STUDY: ELECTRON MICROSCOPES Shortly after the experimental confirmation of the wave properties of the electron, it was suggested that the electron could be used to examine objects
Topics. - How to calibrate the LSM scanner. - How to clean the microscope. - How to adjust the pinhole alignment. - How to adjust the Collimator
 Topics - How to calibrate the LSM scanner - How to measure the PSF - How to clean the microscope - How to adjust the pinhole alignment - How to adjust the Collimator How to calibrate the LSM scanner The
Topics - How to calibrate the LSM scanner - How to measure the PSF - How to clean the microscope - How to adjust the pinhole alignment - How to adjust the Collimator How to calibrate the LSM scanner The
New Features in Release 2.2
 Release 2.2 New Features 1 New Features in Release 2.2 This short manual gives an overview over the new features in the Software v. 2.2.0.8, Analysis Software v. 1.2.0.4). release v. 2.2 (Acquisition 2
Release 2.2 New Features 1 New Features in Release 2.2 This short manual gives an overview over the new features in the Software v. 2.2.0.8, Analysis Software v. 1.2.0.4). release v. 2.2 (Acquisition 2
Introduction to light microscopy
 Center for Microscopy and Image Anaylsis Introduction to light microscopy Basic concepts of imaging with light Urs Ziegler ziegler@zmb.uzh.ch Light interacting with matter Absorbtion Refraction Diffraction
Center for Microscopy and Image Anaylsis Introduction to light microscopy Basic concepts of imaging with light Urs Ziegler ziegler@zmb.uzh.ch Light interacting with matter Absorbtion Refraction Diffraction
INTRODUCTION TO CCD IMAGING
 ASTR 1030 Astronomy Lab 85 Intro to CCD Imaging INTRODUCTION TO CCD IMAGING SYNOPSIS: In this lab we will learn about some of the advantages of CCD cameras for use in astronomy and how to process an image.
ASTR 1030 Astronomy Lab 85 Intro to CCD Imaging INTRODUCTION TO CCD IMAGING SYNOPSIS: In this lab we will learn about some of the advantages of CCD cameras for use in astronomy and how to process an image.
Focus on an optical blind spot A closer look at lenses and the basics of CCTV optical performances,
 Focus on an optical blind spot A closer look at lenses and the basics of CCTV optical performances, by David Elberbaum M any security/cctv installers and dealers wish to know more about lens basics, lens
Focus on an optical blind spot A closer look at lenses and the basics of CCTV optical performances, by David Elberbaum M any security/cctv installers and dealers wish to know more about lens basics, lens
Zeiss 880 Training Notes Zen 2.3
 Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
OPTICAL SYSTEMS OBJECTIVES
 101 L7 OPTICAL SYSTEMS OBJECTIVES Aims Your aim here should be to acquire a working knowledge of the basic components of optical systems and understand their purpose, function and limitations in terms
101 L7 OPTICAL SYSTEMS OBJECTIVES Aims Your aim here should be to acquire a working knowledge of the basic components of optical systems and understand their purpose, function and limitations in terms
Bringing Answers to the Surface
 3D Bringing Answers to the Surface 1 Expanding the Boundaries of Laser Microscopy Measurements and images you can count on. Every time. LEXT OLS4100 Widely used in quality control, research, and development
3D Bringing Answers to the Surface 1 Expanding the Boundaries of Laser Microscopy Measurements and images you can count on. Every time. LEXT OLS4100 Widely used in quality control, research, and development
Assignment 1 Examining the Solar Spectrum with a diffraction grating
 Module 1 Assignments 1 & 2 Before we begin, note that some of the activities require posting answers and results on the Blog. In the document below, the Blog assignments are written in green for ease of
Module 1 Assignments 1 & 2 Before we begin, note that some of the activities require posting answers and results on the Blog. In the document below, the Blog assignments are written in green for ease of
Zeiss 780 Training Notes
 Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
CCAM Microscope Objectives
 CCAM Microscope Objectives Things to consider when selecting an objective Magnification Numerical Aperture (NA) resolving power and light intensity of the objective Working Distance distance between the
CCAM Microscope Objectives Things to consider when selecting an objective Magnification Numerical Aperture (NA) resolving power and light intensity of the objective Working Distance distance between the
You won t be able to measure the incident power precisely. The readout of the power would be lower than the real incident power.
 1. a) Given the transfer function of a detector (below), label and describe these terms: i. dynamic range ii. linear dynamic range iii. sensitivity iv. responsivity b) Imagine you are using an optical
1. a) Given the transfer function of a detector (below), label and describe these terms: i. dynamic range ii. linear dynamic range iii. sensitivity iv. responsivity b) Imagine you are using an optical
Signal-to-Noise Ratio (SNR) discussion
 Signal-to-Noise Ratio (SNR) discussion The signal-to-noise ratio (SNR) is a commonly requested parameter for hyperspectral imagers. This note is written to provide a description of the factors that affect
Signal-to-Noise Ratio (SNR) discussion The signal-to-noise ratio (SNR) is a commonly requested parameter for hyperspectral imagers. This note is written to provide a description of the factors that affect
Optical design of a high resolution vision lens
 Optical design of a high resolution vision lens Paul Claassen, optical designer, paul.claassen@sioux.eu Marnix Tas, optical specialist, marnix.tas@sioux.eu Prof L.Beckmann, l.beckmann@hccnet.nl Summary:
Optical design of a high resolution vision lens Paul Claassen, optical designer, paul.claassen@sioux.eu Marnix Tas, optical specialist, marnix.tas@sioux.eu Prof L.Beckmann, l.beckmann@hccnet.nl Summary:
Olympus Digital Microscope Camera (DP70) checklist
 Smith College - July 2005 Olympus Digital Microscope Camera (DP70) checklist CONTENT, page no. Camera Information, 1 Startup, 1 Retrieve an Image, 2 Microscope Setup, 2 Capture, 3 Preview. 3 Color Balans,
Smith College - July 2005 Olympus Digital Microscope Camera (DP70) checklist CONTENT, page no. Camera Information, 1 Startup, 1 Retrieve an Image, 2 Microscope Setup, 2 Capture, 3 Preview. 3 Color Balans,
Zeiss LSM 880 Protocol
 Zeiss LSM 880 Protocol 1) System Startup Please note put sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time.
Zeiss LSM 880 Protocol 1) System Startup Please note put sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time.
Standard Operating Procedure (SOP) for Shared Equipment: Spinning Disk Confocal Microscope
 Standard Operating Procedure (SOP) for Shared Equipment: Spinning Disk Confocal Microscope This document is to be used as a supplementary guide and not as a replacement for formal training. DO NOT operate
Standard Operating Procedure (SOP) for Shared Equipment: Spinning Disk Confocal Microscope This document is to be used as a supplementary guide and not as a replacement for formal training. DO NOT operate
Application Note #548 AcuityXR Technology Significantly Enhances Lateral Resolution of White-Light Optical Profilers
 Application Note #548 AcuityXR Technology Significantly Enhances Lateral Resolution of White-Light Optical Profilers ContourGT with AcuityXR TM capability White light interferometry is firmly established
Application Note #548 AcuityXR Technology Significantly Enhances Lateral Resolution of White-Light Optical Profilers ContourGT with AcuityXR TM capability White light interferometry is firmly established
attocfm I for Surface Quality Inspection NANOSCOPY APPLICATION NOTE M01 RELATED PRODUCTS G
 APPLICATION NOTE M01 attocfm I for Surface Quality Inspection Confocal microscopes work by scanning a tiny light spot on a sample and by measuring the scattered light in the illuminated volume. First,
APPLICATION NOTE M01 attocfm I for Surface Quality Inspection Confocal microscopes work by scanning a tiny light spot on a sample and by measuring the scattered light in the illuminated volume. First,
The microscope is useful in making observations and collecting data in scientific experiments. Microscopy involves three basic concepts:
 AP BIOLOGY Chapter 6 NAME DATE Block MICROSCOPE LAB PART I: COMPOUND MICROSCOPE OBJECTIVES: After completing this exercise you should be able to: Demonstrate proper care and use of a compound microscope.
AP BIOLOGY Chapter 6 NAME DATE Block MICROSCOPE LAB PART I: COMPOUND MICROSCOPE OBJECTIVES: After completing this exercise you should be able to: Demonstrate proper care and use of a compound microscope.
40 Digital Photo Retouching Techniques COPYRIGHTED MATERIAL
 40 Digital Photo Retouching Techniques COPYRIGHTED MATERIAL C h a p t e r Correcting Contrast If you are a photography enthusiast, you know that light is the defining factor in photography. You probably
40 Digital Photo Retouching Techniques COPYRIGHTED MATERIAL C h a p t e r Correcting Contrast If you are a photography enthusiast, you know that light is the defining factor in photography. You probably
