PREP Course 10: Electronic External Interests Review & The HRPP Review Process Presented by: Hallie Kassan Scott Beardsley Gerry Kassoff
|
|
|
- Myron Barrett
- 5 years ago
- Views:
Transcription
1 PREP Course 10: Electronic External Interests Review & The HRPP Review Process Presented by: Hallie Kassan Scott Beardsley Gerry Kassoff January 18,
2 CME Disclosure Statement The Northwell Health adheres to the ACCME s new Standards for Commercial Support. Any individuals in a position to control the content of a CME activity, including faculty, planners, and managers, are required to disclose all financial relationships with commercial interests. All identified potential conflicts of interest are thoroughly vetted by the Northwell Health for fair balance and scientific objectivity and to ensure appropriateness of patient care recommendations. Course Director and Course Planner, Kevin Tracey, MD and Tina Chuck, MPH have nothing to disclose. Hallie Kassan, Scott Beardsley and Gerry Kassoff have nothing to disclose. January 18,
3 Objectives Identify COI requirements in human subject research Review how our unique electronic platform and tiered review may be best utilized by researchers to reduce administrative burden January 18,
4 January 18,
5 Purpose of External Interests Review Promote objectivity and mitigate actual or perceived bias in research Provide guidance and education on how to comply with local policies and federal regulations Aid in maintaining the public trust in the mission of Northwell Health and its facilities. January 18,
6 January 18,
7 Paper Problems 1. Administrative Burden: High 2. Accessibility of information: Low 3. Opportunity for errors: High THE CHALLENGE: Build a more efficient process January 18,
8 Tiered Review Process 1. Admin review No significant financial interests reported 2. Expedited review Conducted by the COI Committee chair or a designee. 3. Full Committee review Reserved for cases where a standard management plan is inadequate or; When Northwell has a direct financial interest in the companies or products involved in research taking place at Northwell. January 18,
9 Going Electronic January 18,
10 Compliance Pitfalls Minimum requirement is annual submission Updates must be submitted within 30 days of changes to your SFIs Consistency across platforms COI Smart CMS Open Payments era Commons COI submissions to industry sponsors and external IRBs January 18,
11 Audience Participation You are tasked with gathering all of the required information for your PI s progress report/continuing review submission to the IRB. The study is sponsored by AbbyZabby Pharma, Inc. The PI last submitted an external interest disclosure form via COI Smart 8 months ago, and has a management plan. You ask for a copy from the PI and it does not list AbbyZabby. Open Payments data was released four months ago and your PI is reported as accepting $100K in general payments last year from AbbyZabby. WHAT SHOULD YOU DO? January 18,
12 COI requirements for Clinical Research Protocols Must have current annual disclosure on file If disclosure is missing or expired: o For co-investigators they cannot participate in the study until the current disclosure is on file o For PIs study will not be approved until PI completes his/her disclosure. 12
13 Finding COI Disclosure Dates On Feinstein website: 13
14 How do I communicate my COI disclosure date to IRB/HRPP? COI disclosure dates are automatically loaded into IRBmanager Same fields exist any time you add personnel new studies, modifications or continuing review 14
15 What if someone on the study team has a management plan? Answer is yes Alerts HRPP staff to check MP in COI Smart HRPP staff confirm that MP is being applied correctly 15
16 Northwell IRB review of Management Plans (MPs) Review the MP as issued by the COI committee Determine if MP ok as is, or if extra precautions are necessary 16
17 External IRB review and Management Plans HRPP staff will perform the same COI review for studies that are being reviewed by an external IRB MP issued by COI Committee applies regardless of which IRB reviews your study. 17
18 Conflict Mitigation Possible Management Plans Remedies are based on the severity of the potential COI, level of risk of affected studies, and involvement of human subjects. Add language to the consent form Investigator cannot participate in obtaining consent Investigator cannot recruit for a study 18
19 Appeals If you do not agree with this management plan you may alternatively divest yourself of the interest or decline participation in studies involving these companies. If the individual disputes the terms and conditions of a management plan the case will be referred to the COI Committee for resolution. Study activities may not take place until the MP dispute has been resolved. 19
20 Thank you. Questions?? Contact: 20
PREP Course #11: Interactions with Industry What Most Docs Ask About
 PREP Course #11: Interactions with Industry What Most Docs Ask About Presented by: Lou DiGiovanni, Corporate Compliance & Emmelyn Kim, Research Compliance January 23, 2018 1 CME Disclosure Statement The
PREP Course #11: Interactions with Industry What Most Docs Ask About Presented by: Lou DiGiovanni, Corporate Compliance & Emmelyn Kim, Research Compliance January 23, 2018 1 CME Disclosure Statement The
Lurie Children s Panel #2
 Lurie Children s Panel #2 Tricia E ifler, CIP Assistant Director Office of Research Integrity & Compliance teifler@luriechildrens.org 773-755- 7482 Michelle Brown, CIP Biomedical IRB Manager Northwestern
Lurie Children s Panel #2 Tricia E ifler, CIP Assistant Director Office of Research Integrity & Compliance teifler@luriechildrens.org 773-755- 7482 Michelle Brown, CIP Biomedical IRB Manager Northwestern
Clinical Safety & Effectiveness Cohort # 11
 Clinical Safety & Effectiveness Cohort # 11 Compliance in the Documentation of Verbal Disclosure of Relevant Financial Relationships with Commercial Interests from Presenters DATE Educating for Quality
Clinical Safety & Effectiveness Cohort # 11 Compliance in the Documentation of Verbal Disclosure of Relevant Financial Relationships with Commercial Interests from Presenters DATE Educating for Quality
A POLICY in REGARDS to INTELLECTUAL PROPERTY. OCTOBER UNIVERSITY for MODERN SCIENCES and ARTS (MSA)
 A POLICY in REGARDS to INTELLECTUAL PROPERTY OCTOBER UNIVERSITY for MODERN SCIENCES and ARTS (MSA) OBJECTIVE: The objective of October University for Modern Sciences and Arts (MSA) Intellectual Property
A POLICY in REGARDS to INTELLECTUAL PROPERTY OCTOBER UNIVERSITY for MODERN SCIENCES and ARTS (MSA) OBJECTIVE: The objective of October University for Modern Sciences and Arts (MSA) Intellectual Property
DATE OF REVISION March 15, ADMINISTRATIVE OFFICE Office of Research
 ADMINISTRATIVE DIVISION RSCH Office of Research POLICY TITLE Data Access, Retention, and Ownership SCOPE OF POLICY USC System RESPONSIBLE OFFICER Vice President for Research DATE OF REVISION March 15,
ADMINISTRATIVE DIVISION RSCH Office of Research POLICY TITLE Data Access, Retention, and Ownership SCOPE OF POLICY USC System RESPONSIBLE OFFICER Vice President for Research DATE OF REVISION March 15,
APPEAL TO BOARD OF VETERANS APPEALS
 Form Approved: OMB No. 2900-0085 Respondent Burden: 1 Hour APPEAL TO BOARD OF VETERANS APPEALS IMPORTANT: Read the attached instructions before you fill out this form. VA also encourages you to get assistance
Form Approved: OMB No. 2900-0085 Respondent Burden: 1 Hour APPEAL TO BOARD OF VETERANS APPEALS IMPORTANT: Read the attached instructions before you fill out this form. VA also encourages you to get assistance
4.1. Accurate: The information is a true reflection of the original observation.
 SOP #: DOC-101 Page: 1 of 7 Effective Date: 1. POLICY STATEMENT: The Principal Investigator and research team members are required to prepare and maintain adequate and accurate case histories designed
SOP #: DOC-101 Page: 1 of 7 Effective Date: 1. POLICY STATEMENT: The Principal Investigator and research team members are required to prepare and maintain adequate and accurate case histories designed
Intellectual Property
 Tennessee Technological University Policy No. 732 Intellectual Property Effective Date: July 1January 1, 20198 Formatted: Highlight Formatted: Highlight Formatted: Highlight Policy No.: 732 Policy Name:
Tennessee Technological University Policy No. 732 Intellectual Property Effective Date: July 1January 1, 20198 Formatted: Highlight Formatted: Highlight Formatted: Highlight Policy No.: 732 Policy Name:
F98-3 Intellectual/Creative Property
 F98-3 (A.S. 1041) Page 1 of 7 F98-3 Intellectual/Creative Property Legislative History: At its meeting of October 5, 1998, the Academic Senate approved the following policy recommendation presented by
F98-3 (A.S. 1041) Page 1 of 7 F98-3 Intellectual/Creative Property Legislative History: At its meeting of October 5, 1998, the Academic Senate approved the following policy recommendation presented by
Guidance for Industry
 Guidance for Industry Formal Dispute Resolution: Scientific and Technical Issues Related to Pharmaceutical CGMP U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
Guidance for Industry Formal Dispute Resolution: Scientific and Technical Issues Related to Pharmaceutical CGMP U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
Frequently Asked Questions
 Table of Contents Who should an Investigator contact to submit an ERP proposal? What type of information is needed for Medtronic to review? Is a protocol required? Why is so much information necessary?
Table of Contents Who should an Investigator contact to submit an ERP proposal? What type of information is needed for Medtronic to review? Is a protocol required? Why is so much information necessary?
IRB APPLICATION PROCESS. Claire V. Murphy, PharmD, BCPS
 IRB APPLICATION PROCESS Claire V. Murphy, PharmD, BCPS What is the IRB? Institutional Review Board responsible for the risks to research participants Mission is to protect the rights, dignity, welfare
IRB APPLICATION PROCESS Claire V. Murphy, PharmD, BCPS What is the IRB? Institutional Review Board responsible for the risks to research participants Mission is to protect the rights, dignity, welfare
Policy on Patents (CA)
 RESEARCH Effective Date: Date Revised: N/A Supersedes: N/A Related Policies: Policy on Copyright (CA) Responsible Office/Department: Center for Research Innovation (CRI) Keywords: Patent, Intellectual
RESEARCH Effective Date: Date Revised: N/A Supersedes: N/A Related Policies: Policy on Copyright (CA) Responsible Office/Department: Center for Research Innovation (CRI) Keywords: Patent, Intellectual
WANT TO PARTICIPATE IN RESEARCH? THERE S AN APP FOR THAT!
 WANT TO PARTICIPATE IN RESEARCH? THERE S AN APP FOR THAT! SECURITY AND PRIVACY ISSUES WITH THE INCREASED USE OF CONNECTED DEVICES, APPS, AND SOCIAL MEDIA IN RESEARCH. PRESENTED BY: DAVID MATA, SENIOR ASSOCIATE,
WANT TO PARTICIPATE IN RESEARCH? THERE S AN APP FOR THAT! SECURITY AND PRIVACY ISSUES WITH THE INCREASED USE OF CONNECTED DEVICES, APPS, AND SOCIAL MEDIA IN RESEARCH. PRESENTED BY: DAVID MATA, SENIOR ASSOCIATE,
Intellectual Property Ownership and Disposition Policy
 Intellectual Property Ownership and Disposition Policy PURPOSE: To provide a policy governing the ownership of intellectual property and associated University employee responsibilities. I. INTRODUCTION
Intellectual Property Ownership and Disposition Policy PURPOSE: To provide a policy governing the ownership of intellectual property and associated University employee responsibilities. I. INTRODUCTION
Loyola University Maryland Provisional Policies and Procedures for Intellectual Property, Copyrights, and Patents
 Loyola University Maryland Provisional Policies and Procedures for Intellectual Property, Copyrights, and Patents Approved by Loyola Conference on May 2, 2006 Introduction In the course of fulfilling the
Loyola University Maryland Provisional Policies and Procedures for Intellectual Property, Copyrights, and Patents Approved by Loyola Conference on May 2, 2006 Introduction In the course of fulfilling the
Policy Contents. Policy Information. Purpose and Summary. Scope. Published on Policies and Procedures (http://policy.arizona.edu)
 Published on Policies and Procedures (http://policy.arizona.edu) Home > Intellectual Property Policy Policy Contents Purpose and Summary Scope Definitions Policy Related Information* Revision History*
Published on Policies and Procedures (http://policy.arizona.edu) Home > Intellectual Property Policy Policy Contents Purpose and Summary Scope Definitions Policy Related Information* Revision History*
Lewis-Clark State College No Date 2/87 Rev. Policy and Procedures Manual Page 1 of 7
 Policy and Procedures Manual Page 1 of 7 1.0 Policy Statement 1.1 As a state supported public institution, Lewis-Clark State College's primary mission is teaching, research, and public service. The College
Policy and Procedures Manual Page 1 of 7 1.0 Policy Statement 1.1 As a state supported public institution, Lewis-Clark State College's primary mission is teaching, research, and public service. The College
CASE REPORT FORM DESIGN AND IMPLEMENTATION
 CASE REPORT FORM DESIGN AND IMPLEMENTATION DOCUMENT NO.: CR013 v1.0 AUTHOR: Elizabeth Craig ISSUE DATE: 25 October 2016 EFFECTIVE DATE: 08 November 2016 1 INTRODUCTION 1.1 The Academic & Clinical Central
CASE REPORT FORM DESIGN AND IMPLEMENTATION DOCUMENT NO.: CR013 v1.0 AUTHOR: Elizabeth Craig ISSUE DATE: 25 October 2016 EFFECTIVE DATE: 08 November 2016 1 INTRODUCTION 1.1 The Academic & Clinical Central
Academic Medical Center Security and Privacy Conference (AMCSP)
 Academic Medical Center Security and Privacy Conference (AMCSP) econsent Guidance and Use Cases JUNE 27, 2016 Colleen Lawrence, PhD, CCRP, Research Services Consultant, Vanderbilt University Medical Center
Academic Medical Center Security and Privacy Conference (AMCSP) econsent Guidance and Use Cases JUNE 27, 2016 Colleen Lawrence, PhD, CCRP, Research Services Consultant, Vanderbilt University Medical Center
Heart Valve Summit: Medical, Surgical and Interventional Decision Making
 Disclosure/Conflict of Interest Statement As a provider accredited by the Accreditation Council for Continuing Medical Education (ACCME) and the American Nurses Credentialing Center (ANCC), the American
Disclosure/Conflict of Interest Statement As a provider accredited by the Accreditation Council for Continuing Medical Education (ACCME) and the American Nurses Credentialing Center (ANCC), the American
Mistakes to Avoid in Your Catering Business. With Rob Martin Recovering Caterer and Business Consultant
 With Rob Martin Recovering Caterer and Business Consultant Understand your own ability 1. Providing a catered event is different than in your restaurant. 2. Know which type of catering you want to pursue.
With Rob Martin Recovering Caterer and Business Consultant Understand your own ability 1. Providing a catered event is different than in your restaurant. 2. Know which type of catering you want to pursue.
MARCH 21 24, 2018 COLORADO CONVENTION CENTER DENVER, CO
 Guidelines for Commercially Supported Symposia (CME and NCE) HOPA 14TH ANNUAL CONFERENCE MARCH 21 24, 2018 COLORADO CONVENTION CENTER DENVER, CO Industry Partner, We invite you to participate in the Hematology/Oncology
Guidelines for Commercially Supported Symposia (CME and NCE) HOPA 14TH ANNUAL CONFERENCE MARCH 21 24, 2018 COLORADO CONVENTION CENTER DENVER, CO Industry Partner, We invite you to participate in the Hematology/Oncology
Public and Aboriginal Engagement Public Information and Disclosure REGDOC-3.2.1
 Public and Aboriginal Engagement Public Information and Disclosure REGDOC-3.2.1 May 2018 Public Information and Disclosure Regulatory document REGDOC-3.2.1 Canadian Nuclear Safety Commission (CNSC) 2018
Public and Aboriginal Engagement Public Information and Disclosure REGDOC-3.2.1 May 2018 Public Information and Disclosure Regulatory document REGDOC-3.2.1 Canadian Nuclear Safety Commission (CNSC) 2018
 IRBNet: https://training.irbnet.org/training/study/wizard.do 1 of 1 01/10/2014 11:17 AM IRBNet ID: 52832-1 My Reminders (26) UC Davis - Project Information Worksheet - [52832-1] Smart Form Screen Shots
IRBNet: https://training.irbnet.org/training/study/wizard.do 1 of 1 01/10/2014 11:17 AM IRBNet ID: 52832-1 My Reminders (26) UC Davis - Project Information Worksheet - [52832-1] Smart Form Screen Shots
ESI 7735 Old Georgetown Road Bethesda, MD 20814
 Office of the Assistant Secretary of Preparedness and Response (ASPR) KEY PLAYERS TRAVEL CONTRACTOR ESI 7735 Old Georgetown Road Bethesda, MD 20814 John Latz (240) 744-7016 office (202) 439-9097 cellular
Office of the Assistant Secretary of Preparedness and Response (ASPR) KEY PLAYERS TRAVEL CONTRACTOR ESI 7735 Old Georgetown Road Bethesda, MD 20814 John Latz (240) 744-7016 office (202) 439-9097 cellular
SR (FPC)(RC)
 Marshall University Marshall Digital Scholar Recommendations Faculty Senate 5-21-1996 SR-95-96-46 (FPC)(RC) Marshall University Follow this and additional works at: http://mds.marshall.edu/fs_recommendations
Marshall University Marshall Digital Scholar Recommendations Faculty Senate 5-21-1996 SR-95-96-46 (FPC)(RC) Marshall University Follow this and additional works at: http://mds.marshall.edu/fs_recommendations
California State University, Northridge Policy Statement on Inventions and Patents
 Approved by Research and Grants Committee April 20, 2001 Recommended for Adoption by Faculty Senate Executive Committee May 17, 2001 Revised to incorporate friendly amendments from Faculty Senate, September
Approved by Research and Grants Committee April 20, 2001 Recommended for Adoption by Faculty Senate Executive Committee May 17, 2001 Revised to incorporate friendly amendments from Faculty Senate, September
Non-Police Personnel Vetting - (NPPV) Basic
 RESTRICTED - STAFF (WHEN COMPLETE) FORCE VETTING UNIT Non-Police Personnel Vetting - (NPPV) Basic Form No.: 330(g) Rev. 02/2014 White Please complete ALL of the following questionnaire IN FULL. We have
RESTRICTED - STAFF (WHEN COMPLETE) FORCE VETTING UNIT Non-Police Personnel Vetting - (NPPV) Basic Form No.: 330(g) Rev. 02/2014 White Please complete ALL of the following questionnaire IN FULL. We have
Public Information and Disclosure RD/GD-99.3
 Public Information and Disclosure RD/GD-99.3 March, 2012 Public Information and Disclosure Regulatory Document RD/GD-99.3 Minister of Public Works and Government Services Canada 2012 Catalogue number CC172-82/2012E-PDF
Public Information and Disclosure RD/GD-99.3 March, 2012 Public Information and Disclosure Regulatory Document RD/GD-99.3 Minister of Public Works and Government Services Canada 2012 Catalogue number CC172-82/2012E-PDF
DEPARTMENT OF PUBLIC SAFETY DIVISION OF FIRE COLUMBUS, OHIO. SOP Revision Social Media Digital Imagery
 DEPARTMENT OF PUBLIC SAFETY DIVISION OF FIRE COLUMBUS, OHIO 17-007 SUBJECT: TITLE: Administration SOP Revision-04-05-07 Social Media 04-05-08 Digital Imagery Implementation Office of the Chief PURPOSE:
DEPARTMENT OF PUBLIC SAFETY DIVISION OF FIRE COLUMBUS, OHIO 17-007 SUBJECT: TITLE: Administration SOP Revision-04-05-07 Social Media 04-05-08 Digital Imagery Implementation Office of the Chief PURPOSE:
(1) Patents/Patentable means:
 3344-17-02 Patents policy. (A) (B) (C) Research is recognized as an integral part of the educational process to generate new knowledge; to encourage the spirit of inquiry; and to develop scientists, engineers,
3344-17-02 Patents policy. (A) (B) (C) Research is recognized as an integral part of the educational process to generate new knowledge; to encourage the spirit of inquiry; and to develop scientists, engineers,
Card Management. Chapter 7. 4/21/2014 Chapter 7 - Page 1. Copyright University of Pittsburgh. All rights reserved.
 Chapter 7 4/21/2014 Chapter 7 - Page 1 Section Objectives At the end of this section, you should be able to: Request cards Accept cards Assign cards Return cards Reclaim cards View card details 4/21/2014
Chapter 7 4/21/2014 Chapter 7 - Page 1 Section Objectives At the end of this section, you should be able to: Request cards Accept cards Assign cards Return cards Reclaim cards View card details 4/21/2014
UK Research and Innovation Conflicts of Interest Policy
 UK Research and Innovation Conflicts of Interest Policy Contents: Policy Statement 1. Introduction and Purpose. 2. Principles 3. Policy Review. 4. Definitions 5. Examples of Conflicts of Interest 6. Policy
UK Research and Innovation Conflicts of Interest Policy Contents: Policy Statement 1. Introduction and Purpose. 2. Principles 3. Policy Review. 4. Definitions 5. Examples of Conflicts of Interest 6. Policy
PREP Course 32: Intellectual Property (IP) in Research Kirk R. Manogue, PhD Vice President, Technology Transfer
 PREP Course 32: Intellectual Property (IP) in Research Kirk R. Manogue, PhD Vice President, Technology Transfer The Feinstein Institute for Medical Research North Shore-LIJ Health System CME Disclosure
PREP Course 32: Intellectual Property (IP) in Research Kirk R. Manogue, PhD Vice President, Technology Transfer The Feinstein Institute for Medical Research North Shore-LIJ Health System CME Disclosure
Essay Questions. Please review the following list of questions that are categorized by your area of certification. The six areas of certification are:
 Essay Questions Please review the following list of questions that are categorized by your area of certification. The six areas of certification are: Environmental Assessment Environmental Documentation
Essay Questions Please review the following list of questions that are categorized by your area of certification. The six areas of certification are: Environmental Assessment Environmental Documentation
MANAGEMENT DIRECTIVE CONTRACTOR ALERT REPORTING DATABASE (CARD)
 MANAGEMENT DIRECTIVE CONTRACTOR ALERT REPORTING DATABASE (CARD) Management Directive # MD 12-02 Date Issued: 03/01/12 New Policy Release Revision of existing Management Directive dated Cancels: POLICY/BACKGROUND
MANAGEMENT DIRECTIVE CONTRACTOR ALERT REPORTING DATABASE (CARD) Management Directive # MD 12-02 Date Issued: 03/01/12 New Policy Release Revision of existing Management Directive dated Cancels: POLICY/BACKGROUND
UW REGULATION Patents and Copyrights
 UW REGULATION 3-641 Patents and Copyrights I. GENERAL INFORMATION The Vice President for Research and Economic Development is the University of Wyoming officer responsible for articulating policy and procedures
UW REGULATION 3-641 Patents and Copyrights I. GENERAL INFORMATION The Vice President for Research and Economic Development is the University of Wyoming officer responsible for articulating policy and procedures
When Must a Non-UH Investigator Seek Review by the UH IRB? the Issue of Engagement
 University of Hawai i HRPP Standard Operating Procedures When Must a Non-UH Investigator Seek Review by the UH IRB? the Issue of Engagement Purpose and Scope SOP 103 Date: December 18, 2015 The question
University of Hawai i HRPP Standard Operating Procedures When Must a Non-UH Investigator Seek Review by the UH IRB? the Issue of Engagement Purpose and Scope SOP 103 Date: December 18, 2015 The question
Procedure for Obtaining Verification of a Stormwater Manufactured Treatment Device from New Jersey Corporation for Advanced Technology
 Procedure for Obtaining Verification of a Stormwater Manufactured Treatment Device from New Jersey Corporation for Advanced Technology For use in accordance with the Stormwater Management Rules, N.J.A.C.
Procedure for Obtaining Verification of a Stormwater Manufactured Treatment Device from New Jersey Corporation for Advanced Technology For use in accordance with the Stormwater Management Rules, N.J.A.C.
Invention and Technology Disclosure Instructions
 and Technology Disclosure Instructions General Information The purpose of disclosing an invention or other technology is to record what was invented or created and provide a basis for decisions on how
and Technology Disclosure Instructions General Information The purpose of disclosing an invention or other technology is to record what was invented or created and provide a basis for decisions on how
MORGAN STATE UNIVERSITY PROCEDURES ON PATENTS AND TECHNOLOGY TRANSFER APPROVED BY THE PRESIDENT NOVEMBER 2, 2015
 MORGAN STATE UNIVERSITY PROCEDURES ON PATENTS AND TECHNOLOGY TRANSFER APPROVED BY THE PRESIDENT NOVEMBER 2, 2015 I. Introduction The Morgan State University (hereinafter MSU or University) follows the
MORGAN STATE UNIVERSITY PROCEDURES ON PATENTS AND TECHNOLOGY TRANSFER APPROVED BY THE PRESIDENT NOVEMBER 2, 2015 I. Introduction The Morgan State University (hereinafter MSU or University) follows the
FIPPs Fair Information Practice Principles
 FIPPs Fair Information Practice Principles T H E G O L D S TA N DA R D F O R P R OT EC T I N G P E R S O N A L I N F O R M AT I O N Learning Objectives Recognize the Fair Information Practice Principles
FIPPs Fair Information Practice Principles T H E G O L D S TA N DA R D F O R P R OT EC T I N G P E R S O N A L I N F O R M AT I O N Learning Objectives Recognize the Fair Information Practice Principles
Medical Education Company ENDO 2019 Educational Activity Development Policy. TIMELINES Request for Proposal Timeline
 Medical Education Company ENDO 2019 Educational Activity Development Policy TIMELINES Request for Proposal Timeline August 31 October 1 October 9 no extensions Upon Proposal Approval from Endocrine Society
Medical Education Company ENDO 2019 Educational Activity Development Policy TIMELINES Request for Proposal Timeline August 31 October 1 October 9 no extensions Upon Proposal Approval from Endocrine Society
Public and Aboriginal engagement Public Information and Disclosure REGDOC-3.2.1
 Public and Aboriginal engagement Public Information and Disclosure REGDOC-3.2.1 August 2017 Public Information and Disclosure Regulatory document REGDOC-3.2.1 Canadian Nuclear Safety Commission (CNSC)
Public and Aboriginal engagement Public Information and Disclosure REGDOC-3.2.1 August 2017 Public Information and Disclosure Regulatory document REGDOC-3.2.1 Canadian Nuclear Safety Commission (CNSC)
Federalwide Assurance (FWA) for the Protection of Human Subjects
 FWA #: FWA00000068 Institution: U of California, San Francisco (UCSF) Expires: 07/10/2020 OMB No. 0990-0278 Approved for use through August 31, 2017 Federalwide Assurance (FWA) for the Protection of Human
FWA #: FWA00000068 Institution: U of California, San Francisco (UCSF) Expires: 07/10/2020 OMB No. 0990-0278 Approved for use through August 31, 2017 Federalwide Assurance (FWA) for the Protection of Human
HOW TO GET SPECIALTY CARE AND REFERRALS
 Insert for HARP Member Handbooks THE BELOW SECTIONS OF YOUR MEMBER HANDBOOK HAVE BEEN REVISED TO READ AS FOLLOWS HOW TO GET SPECIALTY CARE AND REFERRALS If you need care that your PCP cannot give, he or
Insert for HARP Member Handbooks THE BELOW SECTIONS OF YOUR MEMBER HANDBOOK HAVE BEEN REVISED TO READ AS FOLLOWS HOW TO GET SPECIALTY CARE AND REFERRALS If you need care that your PCP cannot give, he or
A Step by Step guide to making and maintaining a Universal Credit claim online
 A Step by Step guide to making and maintaining a Universal Credit claim online welfare changes Before you make a Universal Credit Claim To make a Universal Credit claim, you will need: Email address Your
A Step by Step guide to making and maintaining a Universal Credit claim online welfare changes Before you make a Universal Credit Claim To make a Universal Credit claim, you will need: Email address Your
How to apply for the Doctorate Extension Scheme. Guide for Students
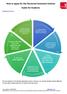 Standard process 7. Start working without restriction once your award is confirmed 1. Contact ISA for guidance when you hand in your thesis 6. Meet with ISA to check your application and submit your documents
Standard process 7. Start working without restriction once your award is confirmed 1. Contact ISA for guidance when you hand in your thesis 6. Meet with ISA to check your application and submit your documents
Essay Questions five
 Essay Questions Please review the following list of questions that are categorized by your area of certification. The five areas of certification are: Environmental Assessment Environmental Documentation
Essay Questions Please review the following list of questions that are categorized by your area of certification. The five areas of certification are: Environmental Assessment Environmental Documentation
A Guide To Writing And Submitting Your Written Testimonial.
 A Guide To Writing And Submitting Your Written Testimonial. This presentation will give you the necessary guidelines for writing and ultimately getting your written testimonial published on the Limbic
A Guide To Writing And Submitting Your Written Testimonial. This presentation will give you the necessary guidelines for writing and ultimately getting your written testimonial published on the Limbic
Applications of Professional Skepticism. CPA Ibrahim Muhumed. 8 th March 2018
 Applications of Professional Skepticism CPA Ibrahim Muhumed 8 th March 2018 Agenda 1. Definition 2. Renewed Focus on Professional Skepticism 3. When to Use Professional Skepticism 4. Main Areas 5. Elements
Applications of Professional Skepticism CPA Ibrahim Muhumed 8 th March 2018 Agenda 1. Definition 2. Renewed Focus on Professional Skepticism 3. When to Use Professional Skepticism 4. Main Areas 5. Elements
CENTER FOR DEVICES AND RADIOLOGICAL HEALTH. Notice to Industry Letters
 CENTER FOR DEVICES AND RADIOLOGICAL HEALTH Standard Operating Procedure for Notice to Industry Letters PURPOSE This document describes the Center for Devices and Radiological Health s (CDRH s, or Center
CENTER FOR DEVICES AND RADIOLOGICAL HEALTH Standard Operating Procedure for Notice to Industry Letters PURPOSE This document describes the Center for Devices and Radiological Health s (CDRH s, or Center
Diana Gordick, Ph.D. 150 E Ponce de Leon, Suite 350 Decatur, GA Health Insurance Portability and Accountability Act (HIPAA)
 Diana Gordick, Ph.D. 150 E Ponce de Leon, Suite 350 Decatur, GA 30030 Health Insurance Portability and Accountability Act (HIPAA) NOTICE OF PRIVACY PRACTICES I. COMMITMENT TO YOUR PRIVACY: DIANA GORDICK,
Diana Gordick, Ph.D. 150 E Ponce de Leon, Suite 350 Decatur, GA 30030 Health Insurance Portability and Accountability Act (HIPAA) NOTICE OF PRIVACY PRACTICES I. COMMITMENT TO YOUR PRIVACY: DIANA GORDICK,
Centralized Research Facility
 Centralized Research Facility at Drexel University User Handbook Table of Contents 1. Introduction 2. Organization 3. Contact Information 4. Advisory Committee 5. Access Requests 6. Safety Training 7.
Centralized Research Facility at Drexel University User Handbook Table of Contents 1. Introduction 2. Organization 3. Contact Information 4. Advisory Committee 5. Access Requests 6. Safety Training 7.
2. Applying for your CAS
 Prepare for your visa application, protect your immigration status Use our step-by-step check list to make sure you are prepared to apply for your Tier 4 visa from outside of the UK. See the following
Prepare for your visa application, protect your immigration status Use our step-by-step check list to make sure you are prepared to apply for your Tier 4 visa from outside of the UK. See the following
ONR Strategy 2015 to 2020
 Title of publication ONR Strategy 2015 to 2020 Office for Nuclear Regulation Page 1 of 5 Introduction Nick Baldwin, Chair The Energy Act 2013 provided for the creation of ONR as an independent, statutory
Title of publication ONR Strategy 2015 to 2020 Office for Nuclear Regulation Page 1 of 5 Introduction Nick Baldwin, Chair The Energy Act 2013 provided for the creation of ONR as an independent, statutory
Peer to Peer On-line Communication for Users with Complex Communication Needs
 Peer to Peer On-line Communication for Users with Complex Communication Needs Yih Lerh Huang & Jan Willem Kolkman Ability Spectrum Whether through physical or mental impairment, a person may not be able
Peer to Peer On-line Communication for Users with Complex Communication Needs Yih Lerh Huang & Jan Willem Kolkman Ability Spectrum Whether through physical or mental impairment, a person may not be able
Photography and Videos at School Policy
 Photography and Videos at School Policy Last updated: 25 May 2018 Contents: Statement of intent 1. Legal framework 2. Definitions 3. Roles and responsibilities 4. Parental consent 5. General procedures
Photography and Videos at School Policy Last updated: 25 May 2018 Contents: Statement of intent 1. Legal framework 2. Definitions 3. Roles and responsibilities 4. Parental consent 5. General procedures
Privacy by Design: Integrating Technology into Global Privacy Practices
 Privacy by Design: Integrating Technology into Global Privacy Practices Ann Cavoukian, Ph.D. Information and Privacy Commissioner Ontario, Canada Harvard Privacy Symposium August 23, 2007 Role of the IPC
Privacy by Design: Integrating Technology into Global Privacy Practices Ann Cavoukian, Ph.D. Information and Privacy Commissioner Ontario, Canada Harvard Privacy Symposium August 23, 2007 Role of the IPC
Privacy Policy SOP-031
 SOP-031 Version: 2.0 Effective Date: 18-Nov-2013 Table of Contents 1. DOCUMENT HISTORY...3 2. APPROVAL STATEMENT...3 3. PURPOSE...4 4. SCOPE...4 5. ABBREVIATIONS...5 6. PROCEDURES...5 6.1 COLLECTION OF
SOP-031 Version: 2.0 Effective Date: 18-Nov-2013 Table of Contents 1. DOCUMENT HISTORY...3 2. APPROVAL STATEMENT...3 3. PURPOSE...4 4. SCOPE...4 5. ABBREVIATIONS...5 6. PROCEDURES...5 6.1 COLLECTION OF
Intellectual Property
 Intellectual Property Policy Type: Board of Visitors Responsible Office: Office of Research and Innovation Initial Policy Approved: 05/15/2009 Current Revision Approved: 03/22/2018 Policy Statement and
Intellectual Property Policy Type: Board of Visitors Responsible Office: Office of Research and Innovation Initial Policy Approved: 05/15/2009 Current Revision Approved: 03/22/2018 Policy Statement and
Policy 7.6 Intellectual Property Policy
 Policy 7.6 Intellectual Property Policy Responsible Official: VP for Research Administration Administering Division/Department: Technology Transfer Effective Date: March 15, 2011 Last Revision: July 14,
Policy 7.6 Intellectual Property Policy Responsible Official: VP for Research Administration Administering Division/Department: Technology Transfer Effective Date: March 15, 2011 Last Revision: July 14,
GUIDE TO COMPLETING THE ONLINE TIER 4 VISA APPLICATION FROM OUTSIDE THE UK
 GUIDE TO COMPLETING THE ONLINE TIER 4 VISA APPLICATION FROM OUTSIDE THE UK You must apply online for a Tier 4 (General) student visa. It costs 348.00 to apply for this visa from outside the UK for the
GUIDE TO COMPLETING THE ONLINE TIER 4 VISA APPLICATION FROM OUTSIDE THE UK You must apply online for a Tier 4 (General) student visa. It costs 348.00 to apply for this visa from outside the UK for the
Pan-Canadian Trust Framework Overview
 Pan-Canadian Trust Framework Overview A collaborative approach to developing a Pan- Canadian Trust Framework Authors: DIACC Trust Framework Expert Committee August 2016 Abstract: The purpose of this document
Pan-Canadian Trust Framework Overview A collaborative approach to developing a Pan- Canadian Trust Framework Authors: DIACC Trust Framework Expert Committee August 2016 Abstract: The purpose of this document
INTELLECTUAL PROPERTY POLICY
 INTELLECTUAL PROPERTY POLICY Overview The University of Texas System (UT System) Board of Regents (Board) and the University of Texas Health Science Center at San Antonio (Health Science Center) encourage
INTELLECTUAL PROPERTY POLICY Overview The University of Texas System (UT System) Board of Regents (Board) and the University of Texas Health Science Center at San Antonio (Health Science Center) encourage
Intellectual Property. Rajkumar Lakshmanaswamy, PhD
 Intellectual Property Rajkumar Lakshmanaswamy, PhD Intellectual Property Patents Trademarks Copyrights Life & Duration Life of utility patent - 17 years from date of issue of Patent if application filed
Intellectual Property Rajkumar Lakshmanaswamy, PhD Intellectual Property Patents Trademarks Copyrights Life & Duration Life of utility patent - 17 years from date of issue of Patent if application filed
Certificate of Acceptance for Studies (CAS) Questionnaire
 Certificate of Acceptance for Studies (CAS) Questionnaire Please complete this form in block capitals and return it as soon as possible to the address listed at the end of the form; you should do this
Certificate of Acceptance for Studies (CAS) Questionnaire Please complete this form in block capitals and return it as soon as possible to the address listed at the end of the form; you should do this
NHS South Kent Coast. Clinical Commissioning Group. Complaints, Comments and Compliments Policy
 NHS South Kent Coast Clinical Commissioning Group Complaints, Comments and Compliments Policy Version: Version 1.6 Ratified by: Date ratified: Name of originator/author: Name of responsible committee/individual:
NHS South Kent Coast Clinical Commissioning Group Complaints, Comments and Compliments Policy Version: Version 1.6 Ratified by: Date ratified: Name of originator/author: Name of responsible committee/individual:
Best Interests Licensee Standards
 Best Interests Licensee Standards 22 October 2018 Best Interests Licensee Standards The Best Interests Licensee Standards are: Seven Safe Harbour Steps; Product Research and Replacement; Prioritising Clients
Best Interests Licensee Standards 22 October 2018 Best Interests Licensee Standards The Best Interests Licensee Standards are: Seven Safe Harbour Steps; Product Research and Replacement; Prioritising Clients
Guide to Tier 4 Print and Send Online Applications
 Guide to Tier 4 Print and Send Online Applications Information from the International Student Support Service April 2013 What is Print and Send? From 6 April 2013, applications for tier 4 and tier 4 dependant
Guide to Tier 4 Print and Send Online Applications Information from the International Student Support Service April 2013 What is Print and Send? From 6 April 2013, applications for tier 4 and tier 4 dependant
1. Go to the following website: https://visas-immigration.service.gov.uk/product/tier-4-student
 1. Go to the following website: https://visas-immigration.service.gov.uk/product/tier-4-student 2. Select Apply now 3. Select Save and continue at the bottom of each question. 4. If you have any other
1. Go to the following website: https://visas-immigration.service.gov.uk/product/tier-4-student 2. Select Apply now 3. Select Save and continue at the bottom of each question. 4. If you have any other
Product Navigator Submission Guidelines and FAQs
 Product Navigator Submission Guidelines and FAQs Thank you for your interest in submitting an article to the Product Navigator newsletter. Below please find a list of guidelines and answers to frequently
Product Navigator Submission Guidelines and FAQs Thank you for your interest in submitting an article to the Product Navigator newsletter. Below please find a list of guidelines and answers to frequently
Freedom of Information Act 2000 (FOIA) Decision notice
 Freedom of Information Act 2000 (FOIA) Decision notice Date: 21 June 2017 Public Authority: Address: NHS Guildford and Waverley Clinical Commissioning Group 3 rd Floor Dominion House Woodbridge Road Guildford
Freedom of Information Act 2000 (FOIA) Decision notice Date: 21 June 2017 Public Authority: Address: NHS Guildford and Waverley Clinical Commissioning Group 3 rd Floor Dominion House Woodbridge Road Guildford
NHS HMR CCG and NHS England Primary Care Joint Commissioning Committee 2015/16
 NHS HMR CCG and NHS England Primary Care Joint Commissioning Committee 2015/16 Date of Meeting: 15 September 2015 Agenda Item: 14 Subject: CCG Assurance New Operating Model Reporting Officer: Sandra Croasdale
NHS HMR CCG and NHS England Primary Care Joint Commissioning Committee 2015/16 Date of Meeting: 15 September 2015 Agenda Item: 14 Subject: CCG Assurance New Operating Model Reporting Officer: Sandra Croasdale
LESSONS LEARNED. Mr. Gianfranco Scipione, M.Sc., J.D./M.B.A. Manager, Research Integrity UHN Research
 Mr. Gianfranco Scipione, M.Sc., J.D./M.B.A. Manager, Research Integrity UHN Research Ms. Katie Roposa, BScN, MEd, RN, CMQ/OE Director, Research Quality Integration UHN Research LESSONS LEARNED Research
Mr. Gianfranco Scipione, M.Sc., J.D./M.B.A. Manager, Research Integrity UHN Research Ms. Katie Roposa, BScN, MEd, RN, CMQ/OE Director, Research Quality Integration UHN Research LESSONS LEARNED Research
Revolutionizing Informed Consent: Best Practices that Leverage Innovation and Technology
 Conference Agenda Thursday, July 23, 2015_v22 Bell Harbor International Conference Center 2211 Alaskan Way, Pier 66, Seattle, WA 98121 Revolutionizing Informed Consent: Best Practices that Leverage Innovation
Conference Agenda Thursday, July 23, 2015_v22 Bell Harbor International Conference Center 2211 Alaskan Way, Pier 66, Seattle, WA 98121 Revolutionizing Informed Consent: Best Practices that Leverage Innovation
Instructions for development of an assent script
 Human Research Protection Program Institutional Review Board Assent Script Template: Ages 7-11 Instructions for development of an assent script ASSENT OF MINOR (AGES 7-11) Children should be approached
Human Research Protection Program Institutional Review Board Assent Script Template: Ages 7-11 Instructions for development of an assent script ASSENT OF MINOR (AGES 7-11) Children should be approached
ATMS GUEST BLOGGER PROGRAM
 ATMS GUEST BLOGGER PROGRAM Guidelines / Terms and Conditions ATMS Guest Blogger Program The Australian Traditional-Medicine Society (ATMS) has a membership base of more than 10,000 practitioners across
ATMS GUEST BLOGGER PROGRAM Guidelines / Terms and Conditions ATMS Guest Blogger Program The Australian Traditional-Medicine Society (ATMS) has a membership base of more than 10,000 practitioners across
Important Plan Information
 Important Plan Information THE BELOW SECTIONS OF YOUR MEMBER HANDBOOK HAVE BEEN REVISED TO READ AS FOLLOWS HOW TO GET SPECIALTY CARE AND REFERRALS If you need care that your PCP cannot give, he or she
Important Plan Information THE BELOW SECTIONS OF YOUR MEMBER HANDBOOK HAVE BEEN REVISED TO READ AS FOLLOWS HOW TO GET SPECIALTY CARE AND REFERRALS If you need care that your PCP cannot give, he or she
Herts Valleys Clinical Commissioning Group. Review of NHS Herts Valleys CCG Constitution
 Herts Valleys Clinical Commissioning Group Review of NHS Herts Valleys CCG s constitution Agenda Item: 14 REPORT TO: HVCCG Board DATE of MEETING: 30 January 2014 SUBJECT: Review of NHS Herts Valleys CCG
Herts Valleys Clinical Commissioning Group Review of NHS Herts Valleys CCG s constitution Agenda Item: 14 REPORT TO: HVCCG Board DATE of MEETING: 30 January 2014 SUBJECT: Review of NHS Herts Valleys CCG
University of Southern California Guidelines for Assigning Authorship and for Attributing Contributions to Research Products and Creative Works
 University of Southern California Guidelines for Assigning Authorship and for Attributing Contributions to Research Products and Creative Works Drafted by the Joint Provost-Academic Senate University Research
University of Southern California Guidelines for Assigning Authorship and for Attributing Contributions to Research Products and Creative Works Drafted by the Joint Provost-Academic Senate University Research
UTAH COMMISSION ON AGING TOOL KIT FOR ADVANCE HEALTHCARE PLANNING
 UTAH COMMISSION ON AGING TOOL KIT FOR ADVANCE HEALTHCARE PLANNING Tool Kit printing has been sponsored by: VistaCare Hospice 1111 Brickyard Road, Suite 107 Salt Lake City, UT 84106-2590 801-467-7772 and
UTAH COMMISSION ON AGING TOOL KIT FOR ADVANCE HEALTHCARE PLANNING Tool Kit printing has been sponsored by: VistaCare Hospice 1111 Brickyard Road, Suite 107 Salt Lake City, UT 84106-2590 801-467-7772 and
General Manager Assurance and Risk Management in Oakton;
 AHSPO Conference C f Is it a Legal Catch Probity & Management Management 23 O October t b 2009 My Background Chartered Accountant and Certified Internal Auditor; General Manager Assurance and Risk Management
AHSPO Conference C f Is it a Legal Catch Probity & Management Management 23 O October t b 2009 My Background Chartered Accountant and Certified Internal Auditor; General Manager Assurance and Risk Management
Managing Conflicts of Interest in Research and Entrepreneurship A Shared Responsibility
 Managing Conflicts of Interest in Research and Entrepreneurship A Shared Responsibility Voichita M. Dadarlat, PhD Assistant Vice President for Research Office of the Executive Vice President for Research
Managing Conflicts of Interest in Research and Entrepreneurship A Shared Responsibility Voichita M. Dadarlat, PhD Assistant Vice President for Research Office of the Executive Vice President for Research
PEDIGREE Publication Policy & Procedures
 PEDIGREE Publication Policy & Procedures Table of Contents 1 PUBLICATION PRINCIPLES 2 2 PUBLICATION PROCEDURE 2 2.1 WRITING: PRELIMINARY NEGOTIATIONS 2 2.2 SUBMISSION OF MANUSCRIPT 2 2.3 ACKNOWLEDGMENTS
PEDIGREE Publication Policy & Procedures Table of Contents 1 PUBLICATION PRINCIPLES 2 2 PUBLICATION PROCEDURE 2 2.1 WRITING: PRELIMINARY NEGOTIATIONS 2 2.2 SUBMISSION OF MANUSCRIPT 2 2.3 ACKNOWLEDGMENTS
SPRING INTO AN INTERNSHIP. Career Services & Employer Relations
 SPRING INTO AN INTERNSHIP Career Services & Employer Relations TOP 10 SKILLS FOR JOB CANDIDATES (2013 NACE SURVEY) 1. Communication 2. Ability to work in a team environment 3. Problem solver, decision
SPRING INTO AN INTERNSHIP Career Services & Employer Relations TOP 10 SKILLS FOR JOB CANDIDATES (2013 NACE SURVEY) 1. Communication 2. Ability to work in a team environment 3. Problem solver, decision
A. Notice to Inventors
 Office of Technology Transfer (OTT) Office of the President () Plan for Carrying Out Licensing Decision Reviews c Provost King and Senior Vice President Mullinix, in a June 18, 2001 letter to Chancellors
Office of Technology Transfer (OTT) Office of the President () Plan for Carrying Out Licensing Decision Reviews c Provost King and Senior Vice President Mullinix, in a June 18, 2001 letter to Chancellors
NIHR / Wellcome Trust King s Clinical Research Facility. Guidance for Investigators
 NIHR / Wellcome Trust King s Clinical Research Facility Guidance for Investigators Introduction The NIHR Clinical Research Facilities provide dedicated facilities to support the delivery of externally-funded
NIHR / Wellcome Trust King s Clinical Research Facility Guidance for Investigators Introduction The NIHR Clinical Research Facilities provide dedicated facilities to support the delivery of externally-funded
To extend a Tier 4 visa or make a PBS Dependants visa application in the UK you must begin the process online. This guide will show you how.
 HomeOfficeCompliance@salford.ac.uk T: +44 (0)161 295 0023 To extend a Tier 4 visa or make a PBS Dependants visa application in the UK you must begin the process online. This guide will show you how. Before
HomeOfficeCompliance@salford.ac.uk T: +44 (0)161 295 0023 To extend a Tier 4 visa or make a PBS Dependants visa application in the UK you must begin the process online. This guide will show you how. Before
Use of a Central IRB (CIRB) for Minimal Risk Study Reviews. Steve Friedman Surveillance Research Program, NCI NAACCR Annual Conference
 Use of a Central IRB (CIRB) for Minimal Risk Study Reviews Steve Friedman Surveillance Research Program, NCI NAACCR Annual Conference June 12, 2018 Background Linkage of patient information from research
Use of a Central IRB (CIRB) for Minimal Risk Study Reviews Steve Friedman Surveillance Research Program, NCI NAACCR Annual Conference June 12, 2018 Background Linkage of patient information from research
Privacy Procedure SOP-031. Version: 04.01
 SOP-031 Version: 04.01 Effective Date: 01-Mar-2017 Table of Contents 1. DOCUMENT HISTORY... 3 2. APPROVAL STATEMENT... 3 3. PURPOSE... 4 4. SCOPE... 4 5. ABBREVIATIONS... 4 6. PROCEDURES... 5 6.1 COLLECTION
SOP-031 Version: 04.01 Effective Date: 01-Mar-2017 Table of Contents 1. DOCUMENT HISTORY... 3 2. APPROVAL STATEMENT... 3 3. PURPOSE... 4 4. SCOPE... 4 5. ABBREVIATIONS... 4 6. PROCEDURES... 5 6.1 COLLECTION
Guide to the Requirements for Public Information and Disclosure GD-99.3
 Guide to the Requirements for Public Information and Disclosure GD-99.3 November 2010 Guide to the Requirements for Public Information and Disclosure Guidance Document GD-99.3 Minister of Public Works
Guide to the Requirements for Public Information and Disclosure GD-99.3 November 2010 Guide to the Requirements for Public Information and Disclosure Guidance Document GD-99.3 Minister of Public Works
Office of Pharmaceutical Quality: Why, What, and How?
 Office of Pharmaceutical Quality: Why, What, and How? Lawrence Yu, Ph.D. Deputy Director, Office of Pharmaceutical Quality FDA Center for Drug Evaluation and Research NIPTE Annual Scientific Conference
Office of Pharmaceutical Quality: Why, What, and How? Lawrence Yu, Ph.D. Deputy Director, Office of Pharmaceutical Quality FDA Center for Drug Evaluation and Research NIPTE Annual Scientific Conference
STEP Code for Will Preparation in England & Wales
 STEP Code for Will Preparation in England & Wales Introduction The STEP Code for Will Preparation in England & Wales is a set of ethical principles that operate for the benefit of clients and demonstrate
STEP Code for Will Preparation in England & Wales Introduction The STEP Code for Will Preparation in England & Wales is a set of ethical principles that operate for the benefit of clients and demonstrate
Pickens Savings and Loan Association, F.A. Online Banking Agreement
 Pickens Savings and Loan Association, F.A. Online Banking Agreement INTERNET BANKING TERMS AND CONDITIONS AGREEMENT This Agreement describes your rights and obligations as a user of the Online Banking
Pickens Savings and Loan Association, F.A. Online Banking Agreement INTERNET BANKING TERMS AND CONDITIONS AGREEMENT This Agreement describes your rights and obligations as a user of the Online Banking
Policy No: TITLE: EFFECTIVE DATE: CANCELLATION: REVIEW DATE:
 Policy No: TITLE: AP-RE-03.2 Intellectual Property Policy EFFECTIVE DATE: February 14, 2014 ADMINISTERED BY: Office of Vice President for Research and Economic Development PURPOSE CANCELLATION: REVIEW
Policy No: TITLE: AP-RE-03.2 Intellectual Property Policy EFFECTIVE DATE: February 14, 2014 ADMINISTERED BY: Office of Vice President for Research and Economic Development PURPOSE CANCELLATION: REVIEW
H5ST 04 (SCDHSC0370) Support the Use of Technological Aids to Promote Independence 1
 H5ST 04 (SCDHSC0370) Support the Use of Technological Aids to Promote Independence Overview This standard identifies the requirements when you support individuals to use technological aids to promote their
H5ST 04 (SCDHSC0370) Support the Use of Technological Aids to Promote Independence Overview This standard identifies the requirements when you support individuals to use technological aids to promote their
ART COLLECTION POLICY
 Policies and Procedures GENERAL ART COLLECTION POLICY TABLE OF CONTENTS 1. Purpose and Principles 2. Care and Conservation 3. Acquisitions 4. Deaccessioning AUTHORITY: RESPONSIBILITY: EFFECTIVE DATE: Board
Policies and Procedures GENERAL ART COLLECTION POLICY TABLE OF CONTENTS 1. Purpose and Principles 2. Care and Conservation 3. Acquisitions 4. Deaccessioning AUTHORITY: RESPONSIBILITY: EFFECTIVE DATE: Board
CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017
 CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017 1. Introduction The goals of the CARRA Publication and Presentation Guidelines are to: a) Promote timely and high-quality presentation
CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017 1. Introduction The goals of the CARRA Publication and Presentation Guidelines are to: a) Promote timely and high-quality presentation
