Lurie Children s Panel #2
|
|
|
- Kristin Carroll
- 5 years ago
- Views:
Transcription
1 Lurie Children s Panel #2 Tricia E ifler, CIP Assistant Director Office of Research Integrity & Compliance teifler@luriechildrens.org Michelle Brown, CIP Biomedical IRB Manager Northwestern University Office of Research michelle.brown@northwestern.edu
2 What is Panel #2? Joint IRB Panel 50% Lurie Children s personnel; 50% NU personnel Monthly meetings Typical turnaround time: reviews or approvals issued within 4 days of IRB meeting Reviews studies involving Lurie Children s and Northwestern University (NU) or an affiliate (NMH, CDH, RIC, etc.) Panel #2 can serve as IRB of record for these studies NU IRB can accept reliance upon review in eirb+ Abbreviated application completed for eirb+ Applies to all types of studies (Full Board and Expedited)
3 The Institutional Authorization Agreement (IAA) Both institutions agreed that: NU may rely on Lurie Children s Panel #2 for review and continuing oversight of its human subject research in these situations: Any study that involves research activity at both Lurie Children s and NU; and/or Any study with funding through NU but research activity at LCH. Both institutions will make reasonable efforts to identify studies at the time of continuing review and transition them to Lurie Children s IRB Panel #2.
4 The Institutional Authorization Agreement (IAA) Lurie Children s Responsibilities: Serve as the IRB of record for studies involving both institutions Lurie Children s Panel #2 will review the entire study, including research activity that involves both adults and children taking place outside Lurie Children s IRB expertise is made up of both pediatric and adult specialties (i.e., maternal-fetal medicine, neonatology, dermatology, etc.) Lurie Children s Panel #2 will have oversight authority to correct non-compliance and/or suspend research
5 The Institutional Authorization Agreement (IAA) NU s Responsibilities: Ensure NU researchers comply with University policies and procedures regarding the conduct of human subject research. Conduct post-approval monitoring of research activities within NU or its affiliated entities. Report any non-compliance or conflict of interest to Lurie Children s IRB Panel #2. Attend Lurie Children s IRB Panel #2 meetings.
6 NU IRB Guidance Consideration #1: Location Which site(s) will be involved? My study will be conducted at NU (and NU affiliates) only. My study will be conducted at LCH only. My study will be conducted at NU and LCH. Consideration #2: Funding Determine where your grants and/or contracts are processed. My funding is processed through NU OSR. My funding is processed through LCH OSP.
7 NU IRB Guidance Consideration #3: Where to Submit If your research activity will involve only Lurie Children s and the funding is processed through the Lurie Children s OSP, contact the Lurie IRB to submit your study in Cayuse. No submission to NU is necessary. If your study will be conducted only at NU (or an NU affiliate) and the funding is processed through the NU OSR, contact the NU IRB to submit your study solely in eirb+. If your study involves research activity at both Lurie Children s and NU and/or has funding through NU but research activity at LCH, your study may be reviewed by the Lurie Children s Panel #2. This will require an abbreviated CIRB submission in NU eirb+ and a full submission in LCH Cayuse.
8 When is NU engaged? Examples of when NU is engaged: Participants will have study visits/activity at an NU site Identified data and/or samples will be sent to investigators at NU Non-Lurie Children s NU faculty or staff will be engaged in some aspect of the research Some or all of the funding will be coming through the NU Office of Sponsored Research Examples of when NU is involved, but not engaged: Data will be collected or stored using RedCAP Sharing de-identified data/specimens only Providing standard of care services only (e.g. radiation therapy that is only being done for standard of care) Performing commercial service (e.g. statistical analysis, lab analysis, etc.)
9 Cayuse IRB Questions
10 Lurie Children s Review Process Submission received via Cayuse IRB IRB staff conduct pre-review Identify major issues Note what type of review is required (convened, expedited) If convened, study is routed to IRB Chair for comments Submission returned to PI for response to pre-review comments Response to pre-review reviewed by same coordinator who conducted pre-review If convened, routed to Panel #2 for review If expedited, route to Vice Chair for review Issues remain, routed back to PI for response Approved, documents sent to PI
11 PI Submits Study, Modification or Renewal for Study involving Lurie Children s & NU or affiliate To Lurie Children s IRB Indicating Involvement with NU or affiliate Study Undergoes Pre- Review per Lurie Children s P &P Study Reviewed by Lurie Children s Panel #2 Once approved Principal Investigator provides Lurie Children s approval documents to NU IRB To NU IRB (abbreviated application) indicating Reliance on Lurie Children s NU or affiliates verifies Investigator/Staff are qualified and have met human subjects education requirement Confirm NU/affiliate has facilities to perform study/department approvals Forward any pertinent COI management plans to Lurie Children s Panel #2 NU determines if reliance is appropriate
12 Lurie Children s- Types of Submissions New studies Can request reliance between the institutions upon initial review Renewals (Continuing Reviews) Studies identified to have NU or an affiliate engagement in the research routed through Panel #2 for review PI can request NU s reliance on Lurie Children s upon submission of renewal to eirb+ (This requires a MOD/CR submission.) Modifications (Amendments) Modifications adding NU or an affiliate as a study site is routed through Panel #2 for review
13 Lurie Children s- Submission Requirements Submission requirements for studies reviewed under Panel #2 follow Lurie Children s policies and procedures Complete Cayuse IRB submission Typically completed prior to submitting study to eirb+ (unless NU involvement including funding, then simultaneous submissions are acceptable) Letters of Support and Organizational Approval for Lurie Children s only Joint Panel Informed Consent documents
14 NU - Types of Submissions & Requirements New studies Include the identifier (CIRB) at the beginning of the study title Indicate in Basic Information Question #7 that an external IRB will serve as the IRB of record Enter the Lurie Children s Panel #2 expiration date Updates (Continuing Reviews) Upload a copy of: the approval letter from Lurie Children s, the most recently approved protocol, and the most recently approved consent/assent documents (if applicable). Modifications (Amendments) Necessary only if the IRB responsibility needs to be changed Reportable New Information (RNIs) Anytime participants at NU are involved, follow guidance on IRB website.
15
16
17 NU Post Review Acknowledgment Why do I get a letter sometimes and not others?! Still working out converting active studies The submission fields were not an exact match in eirb+ This is an evolving process
18 Case #1 Dr. Knee, a Lurie Children s physician, has a greater than minimal risk study being conducted at the Lurie Children s pediatric orthopaedic clinic. Dr. Knee also has privileges at NU, and his funding for this study was received through the NU Office of Sponsored Research. No research procedures are occurring at NU. Which panel at Lurie Children s should review this study? Is NU IRB review required?
19 Case #1 Answers Which panel at Lurie Children s should review this study? Lurie Children s Panel #2 Is NU IRB reliance required? Yes
20 Case #2 Dr. Baby is conducting a study in the Prentice NICU and Lurie Children s PICU. Both Lurie Children s and NU staff are research personnel, and will perform study procedures. The study population includes mothers and their newborn infants being seen in the NICU. Which panel at Lurie Children s should review this study? Is NU IRB reliance required?
21 Case #2 Answers Which panel at Lurie Children s should review this study? Lurie Children s Panel #2 Is NU IRB reliance required? Yes
22 Case #3 Dr. Head is a neurologist at Lurie Children s, and has a study where all the research activity is occurring at Lurie Children s. NU s involvement is limited to data being stored in REDCap. Which panel at Lurie Children s should review this study? Is NU IRB review required?
23 Case #3 Answers Which panel at Lurie Children s should review this study? Lurie Children s Panel #1 Is NU IRB reliance required? No
24 Cayuse IRB Access To request Cayuse IRB access: Complete the Cayuse Backbone form Available on the ORIC website or from ORIC staff Complete Human Subjects Protection Education CITI or NIH course acceptable Send both to
25 eirb+ Access NU Biomedical IRB Information: You may either call 312/ or send an to: Instructions on how to submit your study can be found on the NU IRB website:
26 Panel #2 Meeting Schedule Panel #2 For studies with NU involvement and/or submissions that have completed the pre-review process by noon 10 days prior to the meeting as the agenda permits. August 31, 2015 October 5, 2015 November 2, 2015 December 7, 2015
27 Questions?
PREP Course 10: Electronic External Interests Review & The HRPP Review Process Presented by: Hallie Kassan Scott Beardsley Gerry Kassoff
 PREP Course 10: Electronic External Interests Review & The HRPP Review Process Presented by: Hallie Kassan Scott Beardsley Gerry Kassoff January 18, 2018 1 CME Disclosure Statement The Northwell Health
PREP Course 10: Electronic External Interests Review & The HRPP Review Process Presented by: Hallie Kassan Scott Beardsley Gerry Kassoff January 18, 2018 1 CME Disclosure Statement The Northwell Health
4.1. Accurate: The information is a true reflection of the original observation.
 SOP #: DOC-101 Page: 1 of 7 Effective Date: 1. POLICY STATEMENT: The Principal Investigator and research team members are required to prepare and maintain adequate and accurate case histories designed
SOP #: DOC-101 Page: 1 of 7 Effective Date: 1. POLICY STATEMENT: The Principal Investigator and research team members are required to prepare and maintain adequate and accurate case histories designed
IRB APPLICATION PROCESS. Claire V. Murphy, PharmD, BCPS
 IRB APPLICATION PROCESS Claire V. Murphy, PharmD, BCPS What is the IRB? Institutional Review Board responsible for the risks to research participants Mission is to protect the rights, dignity, welfare
IRB APPLICATION PROCESS Claire V. Murphy, PharmD, BCPS What is the IRB? Institutional Review Board responsible for the risks to research participants Mission is to protect the rights, dignity, welfare
Scope of Services. Project management tasks are estimated based upon anticipated project duration of 52 weeks.
 Scope of Services Project Understanding The City of Sparks (City) has also requested the HDR Engineering, Inc. (HDR) provided limited shop drawing review and periodic observation during construction and
Scope of Services Project Understanding The City of Sparks (City) has also requested the HDR Engineering, Inc. (HDR) provided limited shop drawing review and periodic observation during construction and
Frequently Asked Questions
 Table of Contents Who should an Investigator contact to submit an ERP proposal? What type of information is needed for Medtronic to review? Is a protocol required? Why is so much information necessary?
Table of Contents Who should an Investigator contact to submit an ERP proposal? What type of information is needed for Medtronic to review? Is a protocol required? Why is so much information necessary?
SECTION ADMINISTRATIVE REQUIREMENTS
 PART 1 GENERAL 1.01 SECTION INCLUDES A. Electronic document submittal service. B. Preconstruction meeting. C. Progress meetings. D. Construction progress schedule. E. Coordination drawings. SECTION 013000
PART 1 GENERAL 1.01 SECTION INCLUDES A. Electronic document submittal service. B. Preconstruction meeting. C. Progress meetings. D. Construction progress schedule. E. Coordination drawings. SECTION 013000
NIHR / Wellcome Trust King s Clinical Research Facility. Guidance for Investigators
 NIHR / Wellcome Trust King s Clinical Research Facility Guidance for Investigators Introduction The NIHR Clinical Research Facilities provide dedicated facilities to support the delivery of externally-funded
NIHR / Wellcome Trust King s Clinical Research Facility Guidance for Investigators Introduction The NIHR Clinical Research Facilities provide dedicated facilities to support the delivery of externally-funded
 IRBNet: https://training.irbnet.org/training/study/wizard.do 1 of 1 01/10/2014 11:17 AM IRBNet ID: 52832-1 My Reminders (26) UC Davis - Project Information Worksheet - [52832-1] Smart Form Screen Shots
IRBNet: https://training.irbnet.org/training/study/wizard.do 1 of 1 01/10/2014 11:17 AM IRBNet ID: 52832-1 My Reminders (26) UC Davis - Project Information Worksheet - [52832-1] Smart Form Screen Shots
Use of a Central IRB (CIRB) for Minimal Risk Study Reviews. Steve Friedman Surveillance Research Program, NCI NAACCR Annual Conference
 Use of a Central IRB (CIRB) for Minimal Risk Study Reviews Steve Friedman Surveillance Research Program, NCI NAACCR Annual Conference June 12, 2018 Background Linkage of patient information from research
Use of a Central IRB (CIRB) for Minimal Risk Study Reviews Steve Friedman Surveillance Research Program, NCI NAACCR Annual Conference June 12, 2018 Background Linkage of patient information from research
Clinical Safety & Effectiveness Cohort # 11
 Clinical Safety & Effectiveness Cohort # 11 Compliance in the Documentation of Verbal Disclosure of Relevant Financial Relationships with Commercial Interests from Presenters DATE Educating for Quality
Clinical Safety & Effectiveness Cohort # 11 Compliance in the Documentation of Verbal Disclosure of Relevant Financial Relationships with Commercial Interests from Presenters DATE Educating for Quality
WANT TO PARTICIPATE IN RESEARCH? THERE S AN APP FOR THAT!
 WANT TO PARTICIPATE IN RESEARCH? THERE S AN APP FOR THAT! SECURITY AND PRIVACY ISSUES WITH THE INCREASED USE OF CONNECTED DEVICES, APPS, AND SOCIAL MEDIA IN RESEARCH. PRESENTED BY: DAVID MATA, SENIOR ASSOCIATE,
WANT TO PARTICIPATE IN RESEARCH? THERE S AN APP FOR THAT! SECURITY AND PRIVACY ISSUES WITH THE INCREASED USE OF CONNECTED DEVICES, APPS, AND SOCIAL MEDIA IN RESEARCH. PRESENTED BY: DAVID MATA, SENIOR ASSOCIATE,
About the Course. Benefits of Exhibiting & Supporting
 Structural Heart Intervention and Imaging 2012: A Practical Approach February 2-3, 2012 San Diego Marriott La Jolla San Diego, California Exhibitor Prospectus About the Course Transcatheter structural
Structural Heart Intervention and Imaging 2012: A Practical Approach February 2-3, 2012 San Diego Marriott La Jolla San Diego, California Exhibitor Prospectus About the Course Transcatheter structural
Rulemaking Hearing Rules of the Tennessee Department of Health Bureau of Health Licensure and Regulation Division of Emergency Medical Services
 Rulemaking Hearing Rules of the Tennessee Department of Health Bureau of Health Licensure and Regulation Division of Emergency Medical Services Chapter 1200-12-01 General Rules Amendments of Rules Subparagraph
Rulemaking Hearing Rules of the Tennessee Department of Health Bureau of Health Licensure and Regulation Division of Emergency Medical Services Chapter 1200-12-01 General Rules Amendments of Rules Subparagraph
STATEMENT OF WORK Environmental Assessment for the Red Cliffs/Long Valley Land Exchange in Washington County, Utah
 I. Introduction STATEMENT OF WORK Environmental Assessment for the Red Cliffs/Long Valley Land Exchange in Washington County, Utah The Bureau of Land Management s (BLM) St. George Field Office (SGFO) requires
I. Introduction STATEMENT OF WORK Environmental Assessment for the Red Cliffs/Long Valley Land Exchange in Washington County, Utah The Bureau of Land Management s (BLM) St. George Field Office (SGFO) requires
Academic Medical Center Security and Privacy Conference (AMCSP)
 Academic Medical Center Security and Privacy Conference (AMCSP) econsent Guidance and Use Cases JUNE 27, 2016 Colleen Lawrence, PhD, CCRP, Research Services Consultant, Vanderbilt University Medical Center
Academic Medical Center Security and Privacy Conference (AMCSP) econsent Guidance and Use Cases JUNE 27, 2016 Colleen Lawrence, PhD, CCRP, Research Services Consultant, Vanderbilt University Medical Center
PEDIGREE Publication Policy & Procedures
 PEDIGREE Publication Policy & Procedures Table of Contents 1 PUBLICATION PRINCIPLES 2 2 PUBLICATION PROCEDURE 2 2.1 WRITING: PRELIMINARY NEGOTIATIONS 2 2.2 SUBMISSION OF MANUSCRIPT 2 2.3 ACKNOWLEDGMENTS
PEDIGREE Publication Policy & Procedures Table of Contents 1 PUBLICATION PRINCIPLES 2 2 PUBLICATION PROCEDURE 2 2.1 WRITING: PRELIMINARY NEGOTIATIONS 2 2.2 SUBMISSION OF MANUSCRIPT 2 2.3 ACKNOWLEDGMENTS
Procedure for Obtaining Verification of a Stormwater Manufactured Treatment Device from New Jersey Corporation for Advanced Technology
 Procedure for Obtaining Verification of a Stormwater Manufactured Treatment Device from New Jersey Corporation for Advanced Technology For use in accordance with the Stormwater Management Rules, N.J.A.C.
Procedure for Obtaining Verification of a Stormwater Manufactured Treatment Device from New Jersey Corporation for Advanced Technology For use in accordance with the Stormwater Management Rules, N.J.A.C.
MANAGEMENT DIRECTIVE CONTRACTOR ALERT REPORTING DATABASE (CARD)
 MANAGEMENT DIRECTIVE CONTRACTOR ALERT REPORTING DATABASE (CARD) Management Directive # MD 12-02 Date Issued: 03/01/12 New Policy Release Revision of existing Management Directive dated Cancels: POLICY/BACKGROUND
MANAGEMENT DIRECTIVE CONTRACTOR ALERT REPORTING DATABASE (CARD) Management Directive # MD 12-02 Date Issued: 03/01/12 New Policy Release Revision of existing Management Directive dated Cancels: POLICY/BACKGROUND
ESEA Flexibility. Guidance for Renewal Process. November 13, 2014
 ESEA Flexibility Guidance for Renewal Process November 13, 2014 INTRODUCTION In September 2011, the U.S. Department of Education (Department) offered each State educational agency (SEA) the opportunity
ESEA Flexibility Guidance for Renewal Process November 13, 2014 INTRODUCTION In September 2011, the U.S. Department of Education (Department) offered each State educational agency (SEA) the opportunity
Centralized Research Facility
 Centralized Research Facility at Drexel University User Handbook Table of Contents 1. Introduction 2. Organization 3. Contact Information 4. Advisory Committee 5. Access Requests 6. Safety Training 7.
Centralized Research Facility at Drexel University User Handbook Table of Contents 1. Introduction 2. Organization 3. Contact Information 4. Advisory Committee 5. Access Requests 6. Safety Training 7.
Item 3.0 BOARD OF DIRECTORS OPEN MEETING. MINUTES LOCATION: Providence Boardroom TIME/DATE: September 12, 2017 at 6:00 p.m.
 BOARD OF DIRECTORS OPEN MEETING Item 3.0 MINUTES LOCATION: Providence Boardroom TIME/DATE: September 12, 2017 at 6:00 p.m. ATTENDEES: REGRETS: Governance Advisor: Minutes Recorded By: Brian Devlin - Chair,
BOARD OF DIRECTORS OPEN MEETING Item 3.0 MINUTES LOCATION: Providence Boardroom TIME/DATE: September 12, 2017 at 6:00 p.m. ATTENDEES: REGRETS: Governance Advisor: Minutes Recorded By: Brian Devlin - Chair,
CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017
 CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017 1. Introduction The goals of the CARRA Publication and Presentation Guidelines are to: a) Promote timely and high-quality presentation
CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017 1. Introduction The goals of the CARRA Publication and Presentation Guidelines are to: a) Promote timely and high-quality presentation
ENGINEERING DRAWINGS MANAGEMENT POLICY (IFC/AS BUILTS)
 Approval Amendment Record Approval Date Version Description 15/10/2015 1 This policy takes precedence over L1-NAM-PRO-003 Infrastructure As Built Drawing Management due to business restructures. New Content
Approval Amendment Record Approval Date Version Description 15/10/2015 1 This policy takes precedence over L1-NAM-PRO-003 Infrastructure As Built Drawing Management due to business restructures. New Content
Lewis-Clark State College No Date 2/87 Rev. Policy and Procedures Manual Page 1 of 7
 Policy and Procedures Manual Page 1 of 7 1.0 Policy Statement 1.1 As a state supported public institution, Lewis-Clark State College's primary mission is teaching, research, and public service. The College
Policy and Procedures Manual Page 1 of 7 1.0 Policy Statement 1.1 As a state supported public institution, Lewis-Clark State College's primary mission is teaching, research, and public service. The College
BOARD OF TRUSTEES OPEN MEETING MINUTES Montgomery College, Rockville, Maryland September 21, 2015
 1. CALL TO ORDER BOARD OF TRUSTEES OPEN MEETING MINUTES Montgomery College, Rockville, Maryland September 21, 2015 The Board of Trustees of Montgomery College met in open session on September 21, 2015,
1. CALL TO ORDER BOARD OF TRUSTEES OPEN MEETING MINUTES Montgomery College, Rockville, Maryland September 21, 2015 The Board of Trustees of Montgomery College met in open session on September 21, 2015,
Policy on Patents (CA)
 RESEARCH Effective Date: Date Revised: N/A Supersedes: N/A Related Policies: Policy on Copyright (CA) Responsible Office/Department: Center for Research Innovation (CRI) Keywords: Patent, Intellectual
RESEARCH Effective Date: Date Revised: N/A Supersedes: N/A Related Policies: Policy on Copyright (CA) Responsible Office/Department: Center for Research Innovation (CRI) Keywords: Patent, Intellectual
State of California Health and Human Services Agency California Department of Public Health
 State of California Health and Human Services Agency California Department of Public Health KAREN L. SMITH, MD, MPH Director and State Health Officer EDMUND G. BROWN JR. Governor June 11, 2015 15-07 TO:
State of California Health and Human Services Agency California Department of Public Health KAREN L. SMITH, MD, MPH Director and State Health Officer EDMUND G. BROWN JR. Governor June 11, 2015 15-07 TO:
MORGAN STATE UNIVERSITY PROCEDURES ON PATENTS AND TECHNOLOGY TRANSFER APPROVED BY THE PRESIDENT NOVEMBER 2, 2015
 MORGAN STATE UNIVERSITY PROCEDURES ON PATENTS AND TECHNOLOGY TRANSFER APPROVED BY THE PRESIDENT NOVEMBER 2, 2015 I. Introduction The Morgan State University (hereinafter MSU or University) follows the
MORGAN STATE UNIVERSITY PROCEDURES ON PATENTS AND TECHNOLOGY TRANSFER APPROVED BY THE PRESIDENT NOVEMBER 2, 2015 I. Introduction The Morgan State University (hereinafter MSU or University) follows the
Licensing Procedure for Wireless Broadband Services (WBS) in the Frequency Band MHz
 Issue 1 February 2010 Spectrum Management and Telecommunications Client Procedures Circular Licensing Procedure for Wireless Broadband Services (WBS) in the Frequency Band 3650-3700 MHz Note: Section 6.5
Issue 1 February 2010 Spectrum Management and Telecommunications Client Procedures Circular Licensing Procedure for Wireless Broadband Services (WBS) in the Frequency Band 3650-3700 MHz Note: Section 6.5
Appeals Policy Council for the Accreditation of Educator Preparation th Street, N.W., Suite 400 Washington, D.C
 Appeals Policy Council for the Accreditation of Educator Preparation 1140 19th Street, N.W., Suite 400 Washington, D.C. 20036 Website: caepnet.org Phone: 202.223.0077 July 2017 Document Version Control
Appeals Policy Council for the Accreditation of Educator Preparation 1140 19th Street, N.W., Suite 400 Washington, D.C. 20036 Website: caepnet.org Phone: 202.223.0077 July 2017 Document Version Control
Brad Luke. Director Peddle Thorp Auckland
 Brad Luke Director Peddle Thorp Auckland Site Observation and Practical Completion Preparation PEDDLE THORP Introduction Architects Agreement for Services. Observation Work Plans. Auckland Council Quality
Brad Luke Director Peddle Thorp Auckland Site Observation and Practical Completion Preparation PEDDLE THORP Introduction Architects Agreement for Services. Observation Work Plans. Auckland Council Quality
Revolutionizing Informed Consent: Best Practices that Leverage Innovation and Technology
 Conference Agenda Thursday, July 23, 2015_v22 Bell Harbor International Conference Center 2211 Alaskan Way, Pier 66, Seattle, WA 98121 Revolutionizing Informed Consent: Best Practices that Leverage Innovation
Conference Agenda Thursday, July 23, 2015_v22 Bell Harbor International Conference Center 2211 Alaskan Way, Pier 66, Seattle, WA 98121 Revolutionizing Informed Consent: Best Practices that Leverage Innovation
CASE REPORT FORM DESIGN AND IMPLEMENTATION
 CASE REPORT FORM DESIGN AND IMPLEMENTATION DOCUMENT NO.: CR013 v1.0 AUTHOR: Elizabeth Craig ISSUE DATE: 25 October 2016 EFFECTIVE DATE: 08 November 2016 1 INTRODUCTION 1.1 The Academic & Clinical Central
CASE REPORT FORM DESIGN AND IMPLEMENTATION DOCUMENT NO.: CR013 v1.0 AUTHOR: Elizabeth Craig ISSUE DATE: 25 October 2016 EFFECTIVE DATE: 08 November 2016 1 INTRODUCTION 1.1 The Academic & Clinical Central
Loyola University Maryland Provisional Policies and Procedures for Intellectual Property, Copyrights, and Patents
 Loyola University Maryland Provisional Policies and Procedures for Intellectual Property, Copyrights, and Patents Approved by Loyola Conference on May 2, 2006 Introduction In the course of fulfilling the
Loyola University Maryland Provisional Policies and Procedures for Intellectual Property, Copyrights, and Patents Approved by Loyola Conference on May 2, 2006 Introduction In the course of fulfilling the
Agricultural Data Verification Protocol for the Chesapeake Bay Program Partnership
 Agricultural Data Verification Protocol for the Chesapeake Bay Program Partnership December 3, 2012 Summary In response to an independent program evaluation by the National Academy of Sciences, and the
Agricultural Data Verification Protocol for the Chesapeake Bay Program Partnership December 3, 2012 Summary In response to an independent program evaluation by the National Academy of Sciences, and the
Instructions for development of an assent script
 Human Research Protection Program Institutional Review Board Assent Script Template: Ages 7-11 Instructions for development of an assent script ASSENT OF MINOR (AGES 7-11) Children should be approached
Human Research Protection Program Institutional Review Board Assent Script Template: Ages 7-11 Instructions for development of an assent script ASSENT OF MINOR (AGES 7-11) Children should be approached
New Approaches to Safety and Risk Management
 New Approaches to Safety and Risk Management 15 18 May 2011 The 3rd DIA China Annual Meeting, Bejjin, China Ayman Ayoub MD MSC (med) Safety Surveillance & Risk Management Pfizer Disclaimer The views/opinions
New Approaches to Safety and Risk Management 15 18 May 2011 The 3rd DIA China Annual Meeting, Bejjin, China Ayman Ayoub MD MSC (med) Safety Surveillance & Risk Management Pfizer Disclaimer The views/opinions
NHS HMR CCG and NHS England Primary Care Joint Commissioning Committee 2015/16
 NHS HMR CCG and NHS England Primary Care Joint Commissioning Committee 2015/16 Date of Meeting: 15 September 2015 Agenda Item: 14 Subject: CCG Assurance New Operating Model Reporting Officer: Sandra Croasdale
NHS HMR CCG and NHS England Primary Care Joint Commissioning Committee 2015/16 Date of Meeting: 15 September 2015 Agenda Item: 14 Subject: CCG Assurance New Operating Model Reporting Officer: Sandra Croasdale
DATE OF REVISION March 15, ADMINISTRATIVE OFFICE Office of Research
 ADMINISTRATIVE DIVISION RSCH Office of Research POLICY TITLE Data Access, Retention, and Ownership SCOPE OF POLICY USC System RESPONSIBLE OFFICER Vice President for Research DATE OF REVISION March 15,
ADMINISTRATIVE DIVISION RSCH Office of Research POLICY TITLE Data Access, Retention, and Ownership SCOPE OF POLICY USC System RESPONSIBLE OFFICER Vice President for Research DATE OF REVISION March 15,
New Mexico Lottery Authority MY REWARDS PROGRAM DRAWING PROCEDURES v1.0
 New Mexico Lottery Authority MY REWARDS PROGRAM DRAWING PROCEDURES v1.0 1.0 PURPOSE This document establishes the MY REWARDS Program (the Program ) Drawing Procedures utilized by the NEW MEXICO LOTTERY
New Mexico Lottery Authority MY REWARDS PROGRAM DRAWING PROCEDURES v1.0 1.0 PURPOSE This document establishes the MY REWARDS Program (the Program ) Drawing Procedures utilized by the NEW MEXICO LOTTERY
Examples of Mentoring Agreements
 Examples of Mentoring Agreements Adapted from the W.H. Freeman Entering Mentoring Series, 2017 1 Mentor/Mentee Expectations Fall 2017 Stephanie Robert The relationships between doctoral students and their
Examples of Mentoring Agreements Adapted from the W.H. Freeman Entering Mentoring Series, 2017 1 Mentor/Mentee Expectations Fall 2017 Stephanie Robert The relationships between doctoral students and their
A Step by Step guide to making and maintaining a Universal Credit claim online
 A Step by Step guide to making and maintaining a Universal Credit claim online welfare changes Before you make a Universal Credit Claim To make a Universal Credit claim, you will need: Email address Your
A Step by Step guide to making and maintaining a Universal Credit claim online welfare changes Before you make a Universal Credit Claim To make a Universal Credit claim, you will need: Email address Your
Human Biological Material Collection, Storage and Use
 Avenue E. Mounier 83/11 1200 Brussels Belgium Tel: +32 2 774 1611 Email: eortc@eortc.be www.eortc.org Human Biological Material Collection, Storage and Use POL020 Version 2.1 ALWAYS REFER TO THE INTERNET
Avenue E. Mounier 83/11 1200 Brussels Belgium Tel: +32 2 774 1611 Email: eortc@eortc.be www.eortc.org Human Biological Material Collection, Storage and Use POL020 Version 2.1 ALWAYS REFER TO THE INTERNET
Federalwide Assurance (FWA) for the Protection of Human Subjects
 FWA #: FWA00000068 Institution: U of California, San Francisco (UCSF) Expires: 07/10/2020 OMB No. 0990-0278 Approved for use through August 31, 2017 Federalwide Assurance (FWA) for the Protection of Human
FWA #: FWA00000068 Institution: U of California, San Francisco (UCSF) Expires: 07/10/2020 OMB No. 0990-0278 Approved for use through August 31, 2017 Federalwide Assurance (FWA) for the Protection of Human
Before the FEDERAL COMMUNICATIONS COMMISSION Washington, D.C. ) ) ) ) )
 Before the FEDERAL COMMUNICATIONS COMMISSION Washington, D.C. In the Matter of Amendment of Part 90 of the Commission s Rules ) ) ) ) ) WP Docket No. 07-100 To: The Commission COMMENTS OF THE AMERICAN
Before the FEDERAL COMMUNICATIONS COMMISSION Washington, D.C. In the Matter of Amendment of Part 90 of the Commission s Rules ) ) ) ) ) WP Docket No. 07-100 To: The Commission COMMENTS OF THE AMERICAN
A. Notice to Inventors
 Office of Technology Transfer (OTT) Office of the President () Plan for Carrying Out Licensing Decision Reviews c Provost King and Senior Vice President Mullinix, in a June 18, 2001 letter to Chancellors
Office of Technology Transfer (OTT) Office of the President () Plan for Carrying Out Licensing Decision Reviews c Provost King and Senior Vice President Mullinix, in a June 18, 2001 letter to Chancellors
18th Annual Primary Care in Paradise March 25-28, 2013 Wailea Beach Marriott Resort & Spa Wailea Maui, Hawaii. Exhibitor Prospectus
 18th Annual Primary Care in Paradise March 25-28, 2013 Wailea Beach Marriott Resort & Spa Wailea Maui, Hawaii Exhibitor Prospectus About the Course Scripps Clinic s Primary Care in Paradise conference
18th Annual Primary Care in Paradise March 25-28, 2013 Wailea Beach Marriott Resort & Spa Wailea Maui, Hawaii Exhibitor Prospectus About the Course Scripps Clinic s Primary Care in Paradise conference
SECTION ADMINISTRATIVE REQUIREMENTS SECTION ADMINISTRATIVE REQUIREMENTS
 PART 1 GENERAL 1.01 SECTION INCLUDES A. Project Coordination. B. Preconstruction meeting. C. Progress meetings. D. Preinstallation conferences. E. Requests for information (RFI). F. Coordination drawings.
PART 1 GENERAL 1.01 SECTION INCLUDES A. Project Coordination. B. Preconstruction meeting. C. Progress meetings. D. Preinstallation conferences. E. Requests for information (RFI). F. Coordination drawings.
CALGARY BOARD OF EDUCATION
 CALGARY BOARD OF EDUCATION Minutes of the Regular Meeting of the Board of Trustees (the Board ) held in the Multipurpose Room, Education Centre, 1221 8 Street SW, Calgary, Alberta on Tuesday, January 20,
CALGARY BOARD OF EDUCATION Minutes of the Regular Meeting of the Board of Trustees (the Board ) held in the Multipurpose Room, Education Centre, 1221 8 Street SW, Calgary, Alberta on Tuesday, January 20,
An interpretation of NHS England s Primary Care Co-commissioning: Regional Roadshows questions and answers Rachel Lea, Beds & Herts LMC Ltd
 An interpretation of NHS England s Primary Care Co-commissioning: Regional Roadshows questions and answers Rachel Lea, Beds & Herts LMC Ltd 2. Joint Commissioning Arrangements 2.1 One option for CCGs is
An interpretation of NHS England s Primary Care Co-commissioning: Regional Roadshows questions and answers Rachel Lea, Beds & Herts LMC Ltd 2. Joint Commissioning Arrangements 2.1 One option for CCGs is
National Provider Identifier (NPI) Frequently Asked Questions
 National Provider Identifier (NPI) Frequently Asked Questions I. TRANSITION PERIOD OVERVIEW & PROVIDER REQUIREMENTS II. GETTING, SHARING, AND USING NPI GENERAL QUESTIONS III. TYPE 1 (INDIVIDUAL) VS TYPE
National Provider Identifier (NPI) Frequently Asked Questions I. TRANSITION PERIOD OVERVIEW & PROVIDER REQUIREMENTS II. GETTING, SHARING, AND USING NPI GENERAL QUESTIONS III. TYPE 1 (INDIVIDUAL) VS TYPE
E5 Implementation Working Group Questions & Answers (R1) Current version dated June 2, 2006
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE E5 Implementation Working Group & (R1) Current version dated June 2, 2006 ICH Secretariat,
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE E5 Implementation Working Group & (R1) Current version dated June 2, 2006 ICH Secretariat,
CBD Request to WIPO on the Interrelation of Access to Genetic Resources and Disclosure Requirements
 CBD Request to WIPO on the Interrelation of Access to Genetic Resources and Disclosure Requirements Establishing an adequate framework for a WIPO Response 1 Table of Contents I. Introduction... 1 II. Supporting
CBD Request to WIPO on the Interrelation of Access to Genetic Resources and Disclosure Requirements Establishing an adequate framework for a WIPO Response 1 Table of Contents I. Introduction... 1 II. Supporting
ATMS GUEST BLOGGER PROGRAM
 ATMS GUEST BLOGGER PROGRAM Guidelines / Terms and Conditions ATMS Guest Blogger Program The Australian Traditional-Medicine Society (ATMS) has a membership base of more than 10,000 practitioners across
ATMS GUEST BLOGGER PROGRAM Guidelines / Terms and Conditions ATMS Guest Blogger Program The Australian Traditional-Medicine Society (ATMS) has a membership base of more than 10,000 practitioners across
RESEARCH DATA MANAGEMENT PROCEDURES 2015
 RESEARCH DATA MANAGEMENT PROCEDURES 2015 Issued by: Deputy Vice Chancellor (Research) Date: 1 December 2014 Last amended: 8 June 2017 (administrative amendments only) Signature: Name: Professor Jill Trewhella
RESEARCH DATA MANAGEMENT PROCEDURES 2015 Issued by: Deputy Vice Chancellor (Research) Date: 1 December 2014 Last amended: 8 June 2017 (administrative amendments only) Signature: Name: Professor Jill Trewhella
Air Monitoring Directive Chapter 9: Reporting
 Air Monitoring Directive Chapter 9: Reporting Version Dec 16, 2016 Amends the original Air Monitoring Directive published June, 1989 Title: Air Monitoring Directive Chapter 9: Reporting Number: Program
Air Monitoring Directive Chapter 9: Reporting Version Dec 16, 2016 Amends the original Air Monitoring Directive published June, 1989 Title: Air Monitoring Directive Chapter 9: Reporting Number: Program
How to Complete a PCR
 How to Complete a PCR Information you will need In order to complete your Project Completion Report (PCR), you will need to know the following: Actual Project Outcomes Achieved How these Outcomes have
How to Complete a PCR Information you will need In order to complete your Project Completion Report (PCR), you will need to know the following: Actual Project Outcomes Achieved How these Outcomes have
Merton Clinical Commissioning Group Constitution. [29 May] 2012
![Merton Clinical Commissioning Group Constitution. [29 May] 2012 Merton Clinical Commissioning Group Constitution. [29 May] 2012](/thumbs/73/68756658.jpg) Merton Clinical Commissioning Group Constitution [29 May] 2012 Merton Clinical Commissioning Group Constitution Introduction Dear Members CHAIR S STATEMENT Merton Clinical Commissioning Group has been
Merton Clinical Commissioning Group Constitution [29 May] 2012 Merton Clinical Commissioning Group Constitution Introduction Dear Members CHAIR S STATEMENT Merton Clinical Commissioning Group has been
Ballast Water Collaborative IMO Latest Updates
 Ballast Water Collaborative IMO Latest Updates Silver Springs March 3 2014 Chris Wiley Chair IMO Ballast Water Groups International Maritime Organization The common sense and experience of seafarers coupled
Ballast Water Collaborative IMO Latest Updates Silver Springs March 3 2014 Chris Wiley Chair IMO Ballast Water Groups International Maritime Organization The common sense and experience of seafarers coupled
PREP Course #11: Interactions with Industry What Most Docs Ask About
 PREP Course #11: Interactions with Industry What Most Docs Ask About Presented by: Lou DiGiovanni, Corporate Compliance & Emmelyn Kim, Research Compliance January 23, 2018 1 CME Disclosure Statement The
PREP Course #11: Interactions with Industry What Most Docs Ask About Presented by: Lou DiGiovanni, Corporate Compliance & Emmelyn Kim, Research Compliance January 23, 2018 1 CME Disclosure Statement The
NEVADA DEPARTMENT OF TRANSPORTATION Addendum 3 to RFP July 28, 2017
 NEVADA DEPARTMENT OF TRANSPORTATION Addendum 3 to RFP 697-16-016 July 28, 2017 Reference is made to the Request for Proposal (RFP) to Service Providers for Nevada Shared Radio Replacement Project, upon
NEVADA DEPARTMENT OF TRANSPORTATION Addendum 3 to RFP 697-16-016 July 28, 2017 Reference is made to the Request for Proposal (RFP) to Service Providers for Nevada Shared Radio Replacement Project, upon
To extend a Tier 4 visa or make a PBS Dependants visa application in the UK you must begin the process online. This guide will show you how.
 HomeOfficeCompliance@salford.ac.uk T: +44 (0)161 295 0023 To extend a Tier 4 visa or make a PBS Dependants visa application in the UK you must begin the process online. This guide will show you how. Before
HomeOfficeCompliance@salford.ac.uk T: +44 (0)161 295 0023 To extend a Tier 4 visa or make a PBS Dependants visa application in the UK you must begin the process online. This guide will show you how. Before
What We Heard Report Inspection Modernization: The Case for Change Consultation from June 1 to July 31, 2012
 What We Heard Report Inspection Modernization: The Case for Change Consultation from June 1 to July 31, 2012 What We Heard Report: The Case for Change 1 Report of What We Heard: The Case for Change Consultation
What We Heard Report Inspection Modernization: The Case for Change Consultation from June 1 to July 31, 2012 What We Heard Report: The Case for Change 1 Report of What We Heard: The Case for Change Consultation
Definition of Bulk Electric System Phase 2
 Definition of Bulk Electric System Phase 2 NERC Industry Webinar Peter Heidrich, FRCC, Standard Drafting Team Chair June 26, 2013 Topics Phase 2 - Definition of Bulk Electric System (BES) Project Order
Definition of Bulk Electric System Phase 2 NERC Industry Webinar Peter Heidrich, FRCC, Standard Drafting Team Chair June 26, 2013 Topics Phase 2 - Definition of Bulk Electric System (BES) Project Order
MARCH 21 24, 2018 COLORADO CONVENTION CENTER DENVER, CO
 Guidelines for Commercially Supported Symposia (CME and NCE) HOPA 14TH ANNUAL CONFERENCE MARCH 21 24, 2018 COLORADO CONVENTION CENTER DENVER, CO Industry Partner, We invite you to participate in the Hematology/Oncology
Guidelines for Commercially Supported Symposia (CME and NCE) HOPA 14TH ANNUAL CONFERENCE MARCH 21 24, 2018 COLORADO CONVENTION CENTER DENVER, CO Industry Partner, We invite you to participate in the Hematology/Oncology
Common Ground Food Co-op Board Meeting Minutes November 14, :15 pm to 8:15 pm Lincoln Square Conference Center, Room 154D
 Common Ground Food Co-op Board Meeting Minutes November 14, 2016 6:15 pm to 8:15 pm Lincoln Square Conference Center, Room 154D Present: Magdalena Casper-Shipp, Charles Delman, Tom Howley, Margaret Johnstone,
Common Ground Food Co-op Board Meeting Minutes November 14, 2016 6:15 pm to 8:15 pm Lincoln Square Conference Center, Room 154D Present: Magdalena Casper-Shipp, Charles Delman, Tom Howley, Margaret Johnstone,
Connecticut Department of Social Services Medical Assistance Program Provider Bulletin. National Provider Identifier (NPI), UB-04 and CMS-1500 Updates
 Connecticut Department of Social Services Medical Assistance Program Provider Bulletin PB 2007-14 March 2007 TO: SUBJECT: All Providers (non-pharmacy) National Provider Identifier (NPI), UB-04 and CMS-1500
Connecticut Department of Social Services Medical Assistance Program Provider Bulletin PB 2007-14 March 2007 TO: SUBJECT: All Providers (non-pharmacy) National Provider Identifier (NPI), UB-04 and CMS-1500
Establishment of Electrical Safety Regulations Governing Generation, Transmission and Distribution of Electricity in Ontario
 August 7, 2001 See Distribution List RE: Establishment of Electrical Safety Regulations Governing Generation, Transmission and Distribution of Electricity in Ontario Dear Sir/Madam: The Electrical Safety
August 7, 2001 See Distribution List RE: Establishment of Electrical Safety Regulations Governing Generation, Transmission and Distribution of Electricity in Ontario Dear Sir/Madam: The Electrical Safety
DESIGN PRACTICE NOTE DESIGN/REVIEW REQUIREMENTS FOR TRACTION BONDING PLAN
 Approval Amendment Record Approval Date Version Description 01/04/2014 1 Initial Issue under MTM PRINTOUT MAY NOT BE UP-TO-DATE; REFER TO METRO INTRANET FOR THE LATEST VERSION Page 1 of 7 Table of Contents
Approval Amendment Record Approval Date Version Description 01/04/2014 1 Initial Issue under MTM PRINTOUT MAY NOT BE UP-TO-DATE; REFER TO METRO INTRANET FOR THE LATEST VERSION Page 1 of 7 Table of Contents
Lambeth Clinical Commissioning Group
 Lambeth Clinical Commissioning Group Constitution V003 18 June 2012 Lambeth Clinical Commissioning Group Introduction [DN: Introductory wording to be provided by CCG] Doc no. CLS/067266.5912242 i Lambeth
Lambeth Clinical Commissioning Group Constitution V003 18 June 2012 Lambeth Clinical Commissioning Group Introduction [DN: Introductory wording to be provided by CCG] Doc no. CLS/067266.5912242 i Lambeth
PROGRAM ANNOUNCEMENT. New Jersey Institute of Technology. MSPhM Systems Engineering. Newark. Fall 2008
 PROGRAM ANNOUNCEMENT November 2007 Institution: New Program Title: Degree Designation: Degree Abbreviation: CIP Code and Nomenclature (if possible): Campus(es) where the program will be offered: Date when
PROGRAM ANNOUNCEMENT November 2007 Institution: New Program Title: Degree Designation: Degree Abbreviation: CIP Code and Nomenclature (if possible): Campus(es) where the program will be offered: Date when
Northwestern Intellectual Property Policies. OSR-Evanston Quarterly Network Monday, April 13 th Ben Frey, J.D., Senior Contracts Manager
 Northwestern Intellectual Property Policies OSR-Evanston Quarterly Network Monday, April 13 th Ben Frey, J.D., Senior Contracts Manager Overview Define baseline terms (IP, patents, inventions, copyright)
Northwestern Intellectual Property Policies OSR-Evanston Quarterly Network Monday, April 13 th Ben Frey, J.D., Senior Contracts Manager Overview Define baseline terms (IP, patents, inventions, copyright)
DNVGL-CG-0214 Edition September 2016
 CLASS GUIDELINE DNVGL-CG-0214 Edition September 2016 The content of this service document is the subject of intellectual property rights reserved by ("DNV GL"). The user accepts that it is prohibited by
CLASS GUIDELINE DNVGL-CG-0214 Edition September 2016 The content of this service document is the subject of intellectual property rights reserved by ("DNV GL"). The user accepts that it is prohibited by
Electronic Plans Management Training. Upload Drawings
 Electronic Plans Management Training Upload Drawings Upload Drawings The Electronic Plan Management system (EPM) is an online tool designed to allow architects, engineers, and designers the ability to
Electronic Plans Management Training Upload Drawings Upload Drawings The Electronic Plan Management system (EPM) is an online tool designed to allow architects, engineers, and designers the ability to
AGENDA COMMITTEE ON UNIVERSITY AND FACULTY PERSONNEL
 AGENDA COMMITTEE ON UNIVERSITY AND FACULTY PERSONNEL Meeting: 8:30 a.m., Wednesday, May 24, 2017 Glenn S. Dumke Auditorium Hugo N. Morales, Chair Thelma Meléndez de Santa Ana, Vice Chair Silas H. Abrego
AGENDA COMMITTEE ON UNIVERSITY AND FACULTY PERSONNEL Meeting: 8:30 a.m., Wednesday, May 24, 2017 Glenn S. Dumke Auditorium Hugo N. Morales, Chair Thelma Meléndez de Santa Ana, Vice Chair Silas H. Abrego
AGREEMENT on UnifiedPrinciples and Rules of Technical Regulation in the Republic of Belarus, Republic of Kazakhstan and the Russian Federation
 AGREEMENT on UnifiedPrinciples and Rules of Technical Regulation in the Republic of Belarus, Republic of Kazakhstan and the Russian Federation The Republic of Belarus, Republic of Kazakhstan and the Russian
AGREEMENT on UnifiedPrinciples and Rules of Technical Regulation in the Republic of Belarus, Republic of Kazakhstan and the Russian Federation The Republic of Belarus, Republic of Kazakhstan and the Russian
Appendix 6.1 Data Source Described in Detail Vital Records
 Appendix 6.1 Data Source Described in Detail Vital Records Appendix 6.1 Data Source Described in Detail Vital Records Source or Site Birth certificates Fetal death certificates Elective termination reports
Appendix 6.1 Data Source Described in Detail Vital Records Appendix 6.1 Data Source Described in Detail Vital Records Source or Site Birth certificates Fetal death certificates Elective termination reports
Chair Puig welcomed all Trustees and University faculty, staff, and students.
 FLORIDA INTERNATIONAL UNIVERSITY BOARD OF TRUSTEES FULL BOARD MEETING FEBRUARY 27, 2018 MINUTES 1. Call to Order and Chair s Remarks Board Chair Claudia Puig convened the meeting of the Florida International
FLORIDA INTERNATIONAL UNIVERSITY BOARD OF TRUSTEES FULL BOARD MEETING FEBRUARY 27, 2018 MINUTES 1. Call to Order and Chair s Remarks Board Chair Claudia Puig convened the meeting of the Florida International
DNVGL-CP-0338 Edition October 2015
 CLASS PROGRAMME DNVGL-CP-0338 Edition October 2015 The electronic pdf version of this document, available free of charge from http://www.dnvgl.com, is the officially binding version. FOREWORD DNV GL class
CLASS PROGRAMME DNVGL-CP-0338 Edition October 2015 The electronic pdf version of this document, available free of charge from http://www.dnvgl.com, is the officially binding version. FOREWORD DNV GL class
NZ China EEEMRA. Topics:
 NZ China EEEMRA Topics: Mutual Recognition Agreements (MRAs) China Compulsory Certification (CCC) system EEEMRA Advantages of the EEEMRA Beyond the EEEMRA Peter Morfee Principal Technical Advisor EnergySafety
NZ China EEEMRA Topics: Mutual Recognition Agreements (MRAs) China Compulsory Certification (CCC) system EEEMRA Advantages of the EEEMRA Beyond the EEEMRA Peter Morfee Principal Technical Advisor EnergySafety
You may review a blank copy of the application form by clicking on this pdf link. *Last Name *First Name Middle *Position Title.
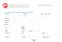 *Last Name *First Name Middle *Position Title *Institution *Department *Address 1: Address 2: *City: Postal Code: State / Province / Region: Country Please Select *Telephone # Fax # *E Mail: *Username:
*Last Name *First Name Middle *Position Title *Institution *Department *Address 1: Address 2: *City: Postal Code: State / Province / Region: Country Please Select *Telephone # Fax # *E Mail: *Username:
Registration of Innovative Medical Devices in China
 Registration of Innovative Medical Devices in China July 2017 Ⅰ Policies on Registration of Innovative Medical Devices Ⅱ Registration of Innovative Medical Devices Ⅲ Registration of Medical Devices with
Registration of Innovative Medical Devices in China July 2017 Ⅰ Policies on Registration of Innovative Medical Devices Ⅱ Registration of Innovative Medical Devices Ⅲ Registration of Medical Devices with
Research with Digital Health Methods 2.0 version date: 03/06/18
 Research with Digital Health Methods 2.0 version date: 03/06/18 Important Caveat: This is intended to be general guidance to assist investigators in navigating a complex area; it is a points to consider
Research with Digital Health Methods 2.0 version date: 03/06/18 Important Caveat: This is intended to be general guidance to assist investigators in navigating a complex area; it is a points to consider
National BIM Standard - United States Version 3
 National BIM Standard - United States Version 3 5 Practice Documents 5.5 Mechanical, Electrical, Plumbing, And Fire Protection Systems (MEP) Spatial Coordination Requirements for Construction Installation
National BIM Standard - United States Version 3 5 Practice Documents 5.5 Mechanical, Electrical, Plumbing, And Fire Protection Systems (MEP) Spatial Coordination Requirements for Construction Installation
Integrative and Holistic Nursing Conference Saturday, April 30 Sunday, May 1, 2016 Paradise Point Resort San Diego, CA Exhibitor Prospectus
 ABOUT THIS CONFERENCE This course is a unique experience that includes an evidence-based overview of holistic and integrative nursing. The latest research on nutrition, nutriceuticals and mind-body-spirit
ABOUT THIS CONFERENCE This course is a unique experience that includes an evidence-based overview of holistic and integrative nursing. The latest research on nutrition, nutriceuticals and mind-body-spirit
UK Shared Business Services Ltd
 SBS 001-18 UK Shared Business Services Ltd Minutes of the 76 th UK SBS Board Meeting held at Polaris House, Swindon on Wednesday 6 th December 2017 commencing at 10.00am. Attending: Board Members: UK SBS
SBS 001-18 UK Shared Business Services Ltd Minutes of the 76 th UK SBS Board Meeting held at Polaris House, Swindon on Wednesday 6 th December 2017 commencing at 10.00am. Attending: Board Members: UK SBS
Welcome to the 25th Annual AMAT Conference
 Welcome to the 25th Annual AMAT Conference As the Association for Multicultural Affairs in Transplantation (AMAT) celebrates its 25 th Annual Conference, we have continuously been a leader for professionals
Welcome to the 25th Annual AMAT Conference As the Association for Multicultural Affairs in Transplantation (AMAT) celebrates its 25 th Annual Conference, we have continuously been a leader for professionals
NEW MEXICO GAMING CONTROL BOARD One-Day Regular Board Meeting July 20, 2011 MINUTES
 NEW MEXICO GAMING CONTROL BOARD One-Day Regular Board Meeting July 20, 2011 MINUTES The Board of Directors of the New Mexico Gaming Control Board (Board) conducted a one-day Regular Board meeting at Gaming
NEW MEXICO GAMING CONTROL BOARD One-Day Regular Board Meeting July 20, 2011 MINUTES The Board of Directors of the New Mexico Gaming Control Board (Board) conducted a one-day Regular Board meeting at Gaming
Agency Information Collection Activities; Proposed Collection; Comment Request; Good
 This document is scheduled to be published in the Federal Register on 06/12/2014 and available online at http://federalregister.gov/a/2014-13787, and on FDsys.gov DEPARTMENT OF HEALTH AND HUMAN SERVICES
This document is scheduled to be published in the Federal Register on 06/12/2014 and available online at http://federalregister.gov/a/2014-13787, and on FDsys.gov DEPARTMENT OF HEALTH AND HUMAN SERVICES
Charter of the Regional Technical Forum Policy Advisory Committee
 Phil Rockefeller Chair Washington Tom Karier Washington Henry Lorenzen Oregon Bill Bradbury Oregon W. Bill Booth Vice Chair Idaho James Yost Idaho Pat Smith Montana Jennifer Anders Montana Charter of the
Phil Rockefeller Chair Washington Tom Karier Washington Henry Lorenzen Oregon Bill Bradbury Oregon W. Bill Booth Vice Chair Idaho James Yost Idaho Pat Smith Montana Jennifer Anders Montana Charter of the
Minutes from the Health and Well-Being Board Financial Planning Group Tuesday 15 December North London Business Park, F13 1pm 3pm
 Minutes from the Health and Well-Being Board Financial Planning Group Tuesday 15 December North London Business Park, F13 1pm 3pm Present: (AC) Andrew Charlwood, Head of Governance, LBB (AD) Anisa Darr,
Minutes from the Health and Well-Being Board Financial Planning Group Tuesday 15 December North London Business Park, F13 1pm 3pm Present: (AC) Andrew Charlwood, Head of Governance, LBB (AD) Anisa Darr,
DRAFT. Cardiac Safety Research Consortium CSRC. Membership Committee Charter. 12September2018. Table of Contents
 DRAFT Cardiac Safety Research Consortium CSRC Membership Committee Charter 12September2018 Table of Contents Purpose of the CSRC Membership Charter and CSRC Membership Committee Composition, Responsibilities
DRAFT Cardiac Safety Research Consortium CSRC Membership Committee Charter 12September2018 Table of Contents Purpose of the CSRC Membership Charter and CSRC Membership Committee Composition, Responsibilities
CRICUT ACCESS TERMS OF USE
 CRICUT ACCESS TERMS OF USE THIS IS AN AGREEMENT BETWEEN YOU AND PROVO CRAFT & NOVELTY, INC., (DOING BUSINESS AS CRICUT ) WITH ITS AFFILIATES, ( PROVO CRAFT OR WE ). PLEASE READ THESE TERMS OF USE, ALL
CRICUT ACCESS TERMS OF USE THIS IS AN AGREEMENT BETWEEN YOU AND PROVO CRAFT & NOVELTY, INC., (DOING BUSINESS AS CRICUT ) WITH ITS AFFILIATES, ( PROVO CRAFT OR WE ). PLEASE READ THESE TERMS OF USE, ALL
Current State of Physician - System - Corporate Joint Ventures
 University of Wisconsin-Madison From the SelectedWorks of Vikas Singh May, 2007 Current State of Physician - System - Corporate Joint Ventures Vikas Singh, University of Arkansas for Medical Sciences Available
University of Wisconsin-Madison From the SelectedWorks of Vikas Singh May, 2007 Current State of Physician - System - Corporate Joint Ventures Vikas Singh, University of Arkansas for Medical Sciences Available
MISSISSIPPI STATE UNIVERSITY Office of Planning Design and Construction Administration
 SECTION 01 340 - SHOP DRAWINGS, PRODUCT DATA AND SAMPLES PART 1 - GENERAL 1.1 RELATED DOCUMENTS A. Drawings and general provisions of the Contract, including General and Supplementary Conditions and other
SECTION 01 340 - SHOP DRAWINGS, PRODUCT DATA AND SAMPLES PART 1 - GENERAL 1.1 RELATED DOCUMENTS A. Drawings and general provisions of the Contract, including General and Supplementary Conditions and other
Franklin Lakes Public Library - Technology Lab Policy
 Franklin Lakes Public Library - Technology Lab Policy Introduction The Franklin Lakes Public Library Technology Lab offers state-of-the-art equipment to facilitate the training of members of the public
Franklin Lakes Public Library - Technology Lab Policy Introduction The Franklin Lakes Public Library Technology Lab offers state-of-the-art equipment to facilitate the training of members of the public
POOLE HOSPITAL NHS FOUNDATION TRUST BOARD OF DIRECTORS
 A POOLE HOSPITAL NHS FOUNDATION TRUST BOARD OF DIRECTORS Part 1 Minutes of the meeting of the Board of Directors of Poole Hospital NHS Foundation Trust held at 9.00 am on Wednesday 29 February 2012 in
A POOLE HOSPITAL NHS FOUNDATION TRUST BOARD OF DIRECTORS Part 1 Minutes of the meeting of the Board of Directors of Poole Hospital NHS Foundation Trust held at 9.00 am on Wednesday 29 February 2012 in
When Must a Non-UH Investigator Seek Review by the UH IRB? the Issue of Engagement
 University of Hawai i HRPP Standard Operating Procedures When Must a Non-UH Investigator Seek Review by the UH IRB? the Issue of Engagement Purpose and Scope SOP 103 Date: December 18, 2015 The question
University of Hawai i HRPP Standard Operating Procedures When Must a Non-UH Investigator Seek Review by the UH IRB? the Issue of Engagement Purpose and Scope SOP 103 Date: December 18, 2015 The question
Privacy Policy SOP-031
 SOP-031 Version: 2.0 Effective Date: 18-Nov-2013 Table of Contents 1. DOCUMENT HISTORY...3 2. APPROVAL STATEMENT...3 3. PURPOSE...4 4. SCOPE...4 5. ABBREVIATIONS...5 6. PROCEDURES...5 6.1 COLLECTION OF
SOP-031 Version: 2.0 Effective Date: 18-Nov-2013 Table of Contents 1. DOCUMENT HISTORY...3 2. APPROVAL STATEMENT...3 3. PURPOSE...4 4. SCOPE...4 5. ABBREVIATIONS...5 6. PROCEDURES...5 6.1 COLLECTION OF
Issues in Emerging Health Technologies Bulletin Process
 Issues in Emerging Health Technologies Bulletin Process Updated: April 2015 Version 1.0 REVISION HISTORY Periodically, this document will be revised as part of ongoing process improvement activities. The
Issues in Emerging Health Technologies Bulletin Process Updated: April 2015 Version 1.0 REVISION HISTORY Periodically, this document will be revised as part of ongoing process improvement activities. The
