Pre-clinical Development Support Services
|
|
|
- Conrad Lawrence
- 5 years ago
- Views:
Transcription
1 Pre-clinical Development Support Services Enabling device development with unique understanding
2 Our Company - Vascular Flow Technologies Vascular Flow Technologies (VFT) has evolved from a tiny surgeon led start-up to a young, innovative Medical Devices Company. VFT developed and patented its novel Spiral Laminar Flow (SLF) technology which restores the natural pattern of blood flow. Through its use in vascular implants, SLF technology has been shown to extend the effective life of implants, help prevent disease progression and generally improve patient outcomes. The company is currently developing new products using the SLF technology platform and has accumulated considerable experience and understanding of preclinical development and testing processes during the journey. Having taken products from concept to regulatory approval, VFT is well placed to offer this service to others embarking on a similar path. VFT has evolved into an ISO Medical Device Development company with unique insight and capabilities in pre-clinical development and testing Our Vision Enabling device development with unique understanding We now work in partnership with a number of medical device companies, Universities and SMEs offering a tailored service to assist their development process If you would like further information on our services and how we can assist you in your projects, please contact us: Vascular Flow Technologies Ltd Prospect Business Centre, Gemini Crescent, Dundee DD2 1TYUK T: +44 (0) M: +44 (0) E: admin@vascular-flow.com W: vascular-flow.com
3 Our History Product development capability has evolved over a number of years 2000 SLFTM observed and patentprotected by three Scottish Clinicians - [Professor Peter Stonebridge, Professor Graeme Houston & John Dick] 2008 First product approved with CE mark for Peripheral Vascular Grafts. Arteriovenous Access Grafts followed in June New investment Business strategy developed to prove commercial concept and build new product pipeline 2013 Clinical Advisory Board appointed. New products launched in Europe and USA. Clinical Evidence Programme (CEP) delivers early strong data Clinical evidence development in Europe and the USA. Direct pilot sales model in USA and new product launches Commercial partnerships with Vascular device companies 2016 VFT focus on Stent,Catheter and Heart valve product development, divest Graft business & establish a niche product development partnership centred on fluid dynamic modelling and testing Our Team We understand Medical Device development
4 Our Capabilities - how we can help Computational Fluid Dynamics At Vascular Flow we are able to provide off-the-shelf or totally bespoke services that enable quicker and more cost-effective progression of projects, ranging from new product development to research initiatives. We offer an outsourced R&D service that can enhance your internal capabilities and reduce cost within your business Finding Product development partners or outsourcing parts of your R&D programme can enable you to react more quickly you can call upon a pool of resource with the skills to deliver your requirements speedily, cost effectively and without increase in headcount. You will also have access to the latest technology without the related capital investment. Our background in product development ensures we have the relevant experience to understand your needs and offer innovative ways forward for your project whilst always keeping a clear eye on budget. Working with us can also mitigate risk. We work in close collaboration all the way and dovetail into your own Project Management structure as part of the team. We operate to ISO13485 certification We offer an outsourced R&D service that can enhance your internal capabilities and reduce cost within your business Computer simulations are used to speedup key design phases of vascular devices. VFT has proficiency at investigating haemodynamic flows with steady and pulsatile characteristics in parametric and physiological representative models. Utilising industry leading CFD software packages, numerical models are used to design/conduct experiments and optimize the outcome of medical devices before prototyping. VFT has proficiency at investigating haemodynamic flows with steady and pulsatile characteristics in parametric and physiological representative models. In Vitro Flow (Bench Models) In vitro tests with a bespoke, computer controlled flow rig are used to perform verification of the outcomes of a medical device in a controlled environment. VFT has the capability of producing complex geometries and pulsatile flow to represent devices in blood vessels. We measure flow rates with pressure and flow sensors, and complex flow structures are measured with Doppler ultrasound. Blood viscosity and density are reproduced with blood mimic fluid. VFT has the capability of producing complex geometries and pulsatile flow to represent devices in blood vessels
5 In Vitro Flow Testing & Deployment Prototyping Cadaveric models are the most realistic anatomical models to test vascular devices prior to pre-clinical trials. We have a partnership with the Institute of Medical Sciences & Technology to test the deployment and flow outcome of vascular devices in cadaveric anatomical model. The cadaveric bodies using the Thiel soft-fix method, which preserves them with life-like flexibility and tissue quality. Flow and anatomy are visualized with ultrasound, fluoroscopy, CT or MRI. "Cadaveric models are the most realistic anatomical models to test vascular devices prior to pre-clinical trials." 3D Printing We have a state of the art Big Box 3D printer which can be utilised to print prototype products or can be used to print flow rig components like anatomical models. The options are endless. Injection Moulding We have two Injection moulding machines and a wealth of injection moulding experience to enhance the prototyping process. Heat Setting/Electropolishing We have experience of working with shape memory alloys and have strong knowledge of product design. We have the capability to shape set with bespoke tooling design and can carry out vital surface finishing using electro polishing techniques. VFT has a broad range of services and equipment available to enhance product development Computer Aided Design Project Management & Delivery VFT utilises the latest Solidworks 3D CAD software to compliment its R&D offering. It is used to create and improve designs and reduce costs and is key to good communication of new products and documenting design history. VFT has experience in parametric design processes and non-linear/complex curvature model creation. VFT has experience in parametric design processes and non-linear/complex curvature model creation. VFT s highly experienced team has a proven track record of taking products from initial design concept to market. We have developed a robust project management office to deliver ISO13485 compliant Project Management & Design History File generation. We can work in collaborative partnerships or provide full turn-key solutions, offering flexible design, development & verification costing models. VFT s highly experienced team has a proven track record of taking products from initial design concept to market.
6 ISO Standard Performance testing VFT has invested heavily in new equipment and enhanced its laboratory to capitalise on the in house experience gained through previous projects and taking products to market. This expertise allows us to offer product performance testing to ISO 10555, ISO 25539, ISO 7198 and ISO Working within the constraints of our quality system ensures customers always receive comprehensive reports and test results. Our expertise allows us to offer product performance testing to ISO 10555, ISO 25539, ISO 7198 and ISO Work with us VFT would love to assist you in your projects. You will find us a flexible and responsive partner, eager to help at all times. If you would like further information on our services and how we can assist you in your projects, please contact us: Vascular Flow Technologies Ltd Prospect Business Centre Gemini Crescent Dundee, DD2 1TYUK T +44 (0) M +44 (0) E info@vascular-flow.com W Regulatory Support VFT has extensive Class IIa, IIb, III device submission experience with both CE and 510k submissions and approvals We can support full ISO design history and technical file generation & ISO14971 Risk Assessment Our experienced team can undertake full Clinical Literature Reviews on your behalf to support the early stage feasibility processes. We can support full ISO design history and technical file generation & ISO14971 Risk Assessment
7 vascular-flow.com
PRECISION ENGINEERED PRODUCTS FOR THE MEDICAL DEVICES SECTOR
 PRECISION ENGINEERED PRODUCTS FOR THE MEDICAL DEVICES SECTOR Introduction Caragh Precision provide precision machined metal and plastic products for the medical devices industry. We work in partnership
PRECISION ENGINEERED PRODUCTS FOR THE MEDICAL DEVICES SECTOR Introduction Caragh Precision provide precision machined metal and plastic products for the medical devices industry. We work in partnership
USTGlobal. 3D Printing. Changing the Face of Healthcare
 USTGlobal 3D Printing Changing the Face of Healthcare UST Global Inc, November 2017 Table of Contents Introduction 3 Challenges 3 Impact of 3D Printing 4 IT Solutions for 3D Printing 5 How UST Global Can
USTGlobal 3D Printing Changing the Face of Healthcare UST Global Inc, November 2017 Table of Contents Introduction 3 Challenges 3 Impact of 3D Printing 4 IT Solutions for 3D Printing 5 How UST Global Can
Cellexus International Limited
 MAIN CONFERENCE SPONSOR Medtronic As a global leader in medical technology, services and solutions, Medtronic improves the health and lives of millions of people each year. We believe our deep clinical,
MAIN CONFERENCE SPONSOR Medtronic As a global leader in medical technology, services and solutions, Medtronic improves the health and lives of millions of people each year. We believe our deep clinical,
emblation microwave INDUSTRIAL, SCIENTIFIC AND MEDICAL
 emblation microwave INDUSTRIAL, SCIENTIFIC AND MEDICAL The Next Generation of Microwave Systems Cold Plasma Microwave Ablation Microwave ablation is a rapidly growing field of medicine. As products and
emblation microwave INDUSTRIAL, SCIENTIFIC AND MEDICAL The Next Generation of Microwave Systems Cold Plasma Microwave Ablation Microwave ablation is a rapidly growing field of medicine. As products and
Technology and Innovation in the NHS Scottish Health Innovations Ltd
 Technology and Innovation in the NHS Scottish Health Innovations Ltd Introduction Scottish Health Innovations Ltd (SHIL) has, since 2002, worked in partnership with NHS Scotland to identify, protect, develop
Technology and Innovation in the NHS Scottish Health Innovations Ltd Introduction Scottish Health Innovations Ltd (SHIL) has, since 2002, worked in partnership with NHS Scotland to identify, protect, develop
Leveraging Med Device Expertise to Develop Combination Products
 Leveraging Med Device Expertise to Develop Combination Products 20 th Annual Drug Delivery Partnership Conference January 20, 2016 Dirk Smith VP of Technology Solutions Minnetronix, Inc. 250 employees,
Leveraging Med Device Expertise to Develop Combination Products 20 th Annual Drug Delivery Partnership Conference January 20, 2016 Dirk Smith VP of Technology Solutions Minnetronix, Inc. 250 employees,
YOUR PARTNER FOR CHALLENGING PROJECTS
 YOUR PARTNER FOR CHALLENGING PROJECTS OUR CLAIM FOR PERFECTION in plastics processing FOR QUALITY thanks to complete process capability Injection moulding, extrusion and assembly are our core competencies.
YOUR PARTNER FOR CHALLENGING PROJECTS OUR CLAIM FOR PERFECTION in plastics processing FOR QUALITY thanks to complete process capability Injection moulding, extrusion and assembly are our core competencies.
Your partner for challenging projects
 Your partner for challenging projects Our claim for PERFECTION in plastics processing for QUALITY thanks to complete process capability Injection moulding, extrusion and assembly are our core competencies.
Your partner for challenging projects Our claim for PERFECTION in plastics processing for QUALITY thanks to complete process capability Injection moulding, extrusion and assembly are our core competencies.
TGA Discussion Paper 3D Printing Technology in the Medical Device Field Australian Regulatory Considerations
 TGA Discussion Paper 3D Printing Technology in the Medical Device Field Australian Regulatory Considerations MTAA Response - October 2017 October 2017 Australian Regulatory Considerations Page 1 of 7 Level
TGA Discussion Paper 3D Printing Technology in the Medical Device Field Australian Regulatory Considerations MTAA Response - October 2017 October 2017 Australian Regulatory Considerations Page 1 of 7 Level
Navigating the Healthcare Innovation Cycle
 Navigating the Healthcare Innovation Cycle Introduction: CIMIT s 20 + years of experience in facilitating more than 600 projects is that innovation in Healthcare is a learnable, teachable process, which
Navigating the Healthcare Innovation Cycle Introduction: CIMIT s 20 + years of experience in facilitating more than 600 projects is that innovation in Healthcare is a learnable, teachable process, which
Response to the Western Australian Government Sustainable Health Review
 Response to the Western Australian Government Sustainable Health Review On behalf of Australia s digital health community, HISA commends this submission to the Sustainable Health Review Panel, and wish
Response to the Western Australian Government Sustainable Health Review On behalf of Australia s digital health community, HISA commends this submission to the Sustainable Health Review Panel, and wish
Key Insight Business Briefing
 Promoting physics, supporting physicists Key Insight Business Briefing The role of government in promoting innovation Chairman: Peter Saraga The Institute of Physics 28 April 2008 www.iop.org Promoting
Promoting physics, supporting physicists Key Insight Business Briefing The role of government in promoting innovation Chairman: Peter Saraga The Institute of Physics 28 April 2008 www.iop.org Promoting
December Eucomed HTA Position Paper UK support from ABHI
 December 2008 Eucomed HTA Position Paper UK support from ABHI The Eucomed position paper on Health Technology Assessment presents the views of the Medical Devices Industry of the challenges of performing
December 2008 Eucomed HTA Position Paper UK support from ABHI The Eucomed position paper on Health Technology Assessment presents the views of the Medical Devices Industry of the challenges of performing
Medical Images Analysis and Processing
 Medical Images Analysis and Processing - 25642 Emad Course Introduction Course Information: Type: Graduated Credits: 3 Prerequisites: Digital Image Processing Course Introduction Reference(s): Insight
Medical Images Analysis and Processing - 25642 Emad Course Introduction Course Information: Type: Graduated Credits: 3 Prerequisites: Digital Image Processing Course Introduction Reference(s): Insight
peace of mind For from development to commercial supply
 For peace of mind from development to commercial supply aesica-pharma.com weshouldtalk@aesica-pharma.com @aesica CorporateBroEng_v2 0814 @ Aesica 2014 Your full service CDMO Our vision: To be the number
For peace of mind from development to commercial supply aesica-pharma.com weshouldtalk@aesica-pharma.com @aesica CorporateBroEng_v2 0814 @ Aesica 2014 Your full service CDMO Our vision: To be the number
Helping your business grow in the UK health system
 Helping your business grow in the UK health system OVERVIEW PROVIDING INSIGHT AND HELPING YOU DELIVER INNOVATIVE PRODUCTS AND SERVICES TO THE NHS AND BEYOND. The health system is a complex and uncertain
Helping your business grow in the UK health system OVERVIEW PROVIDING INSIGHT AND HELPING YOU DELIVER INNOVATIVE PRODUCTS AND SERVICES TO THE NHS AND BEYOND. The health system is a complex and uncertain
Health & Social Care Industrial Innovation
 Health & Social Care Industrial Innovation Mr Andrew Fowlie Scottish Government Health Innovations Team SHINE North Sea Region Program 2014 2020 Scotland s Medical Technologies Landscape Imaging Non Imaging
Health & Social Care Industrial Innovation Mr Andrew Fowlie Scottish Government Health Innovations Team SHINE North Sea Region Program 2014 2020 Scotland s Medical Technologies Landscape Imaging Non Imaging
SHTG primary submission process
 Meeting date: 24 April 2014 Agenda item: 8 Paper number: SHTG 14-16 Title: Purpose: SHTG primary submission process FOR INFORMATION Background The purpose of this paper is to update SHTG members on developments
Meeting date: 24 April 2014 Agenda item: 8 Paper number: SHTG 14-16 Title: Purpose: SHTG primary submission process FOR INFORMATION Background The purpose of this paper is to update SHTG members on developments
Driving Innovation. Connect and Catalyse. Medicines and Healthcare the Global perspective (+10 years) Zahid Latif
 Connect and Catalyse Medicines and Healthcare the Global perspective (+10 years) Zahid Latif % GDP spent on health OECD countries spent nearly $3.5trn in 2005 ($4.6 trillion in 2008) on healthcare services
Connect and Catalyse Medicines and Healthcare the Global perspective (+10 years) Zahid Latif % GDP spent on health OECD countries spent nearly $3.5trn in 2005 ($4.6 trillion in 2008) on healthcare services
Medical Technology Association of NZ. Proposed European Union/New Zealand Free Trade Agreement. Submission to Ministry of Foreign Affairs & Trade
 Medical Technology Association of NZ Proposed European Union/New Zealand Free Trade Agreement Submission to Ministry of Foreign Affairs & Trade February 2016 1 Introduction The Medical Technology Association
Medical Technology Association of NZ Proposed European Union/New Zealand Free Trade Agreement Submission to Ministry of Foreign Affairs & Trade February 2016 1 Introduction The Medical Technology Association
Leader in Pharmaceutical Films
 TSX-V: IGX OTCQX: IGXT Leader in Pharmaceutical Films Your Specialist in Contract Development & Manufacturing WE MAKE APPROVED DRUGS BETTER Our Mission We make approved drugs better, with next generation
TSX-V: IGX OTCQX: IGXT Leader in Pharmaceutical Films Your Specialist in Contract Development & Manufacturing WE MAKE APPROVED DRUGS BETTER Our Mission We make approved drugs better, with next generation
ECSEL Smart Health Project Portfolio and Strategic Agenda. Patrick Vandenberghe Programme Officer
 ECSEL Smart Health Project Portfolio and Strategic Agenda Patrick Vandenberghe Programme Officer ECSEL-IMI Joint Workshop July 5th 2017 1 Multi-Annual Strategic Plan MASP 2017 Strategic Thrusts Instruments
ECSEL Smart Health Project Portfolio and Strategic Agenda Patrick Vandenberghe Programme Officer ECSEL-IMI Joint Workshop July 5th 2017 1 Multi-Annual Strategic Plan MASP 2017 Strategic Thrusts Instruments
Taking a broader view
 Taking a broader view A brief introduction to DNV GL 1 SAFER, SMARTER, GREENER We are a global classification, certification, technical assurance and advisory company 2 In a challenging world we make businesses
Taking a broader view A brief introduction to DNV GL 1 SAFER, SMARTER, GREENER We are a global classification, certification, technical assurance and advisory company 2 In a challenging world we make businesses
scinnovation-global.com Innovative Engineering Robust Solutions
 scinnovation-global.com Innovative Engineering Robust Solutions SC INNOVATION IS A SPECIALIST ENGINEERING COMPANY, EXPERIENCED IN PROVIDING PRODUCTS AND SERVICES FOR DEMANDING ENVIRONMENTS WHERE QUALITY
scinnovation-global.com Innovative Engineering Robust Solutions SC INNOVATION IS A SPECIALIST ENGINEERING COMPANY, EXPERIENCED IN PROVIDING PRODUCTS AND SERVICES FOR DEMANDING ENVIRONMENTS WHERE QUALITY
ESG. Staging Temporary Structures Event Overlay Project Management
 ESG Staging Temporary Structures Event Overlay Project Management COMPANY PROFILE ES GLOBAL PROVIDES INNOVATIVE AND CREATIVE SOLUTIONS FOR MUSIC, SPORTING, CORPORATE AND HOSPITALITY EVENTS Creating the
ESG Staging Temporary Structures Event Overlay Project Management COMPANY PROFILE ES GLOBAL PROVIDES INNOVATIVE AND CREATIVE SOLUTIONS FOR MUSIC, SPORTING, CORPORATE AND HOSPITALITY EVENTS Creating the
Job Description. Commitment: Must be available to work full-time hours, M-F for weeks beginning Summer of 2018.
 Research Intern Director of Research We are seeking a summer intern to support the team to develop prototype 3D sensing systems based on state-of-the-art sensing technologies along with computer vision
Research Intern Director of Research We are seeking a summer intern to support the team to develop prototype 3D sensing systems based on state-of-the-art sensing technologies along with computer vision
Translational scientist competency profile
 C-COMEND Competency profile for Translational Scientists C-COMEND is a two-year European training project supported by the Erasmus plus programme, which started on November 1st 2015. The overall objective
C-COMEND Competency profile for Translational Scientists C-COMEND is a two-year European training project supported by the Erasmus plus programme, which started on November 1st 2015. The overall objective
HealthTech: What does it mean for compliance?
 HealthTech: What does it mean for compliance? May 2018 Agenda 11:15 AM 11:30 AM Introduction Kathleen Meriwether, Americas Leader - Life Sciences Fraud Investigation & Dispute Services, EY 11:30 AM 12:15
HealthTech: What does it mean for compliance? May 2018 Agenda 11:15 AM 11:30 AM Introduction Kathleen Meriwether, Americas Leader - Life Sciences Fraud Investigation & Dispute Services, EY 11:30 AM 12:15
HTA Position Paper. The International Network of Agencies for Health Technology Assessment (INAHTA) defines HTA as:
 HTA Position Paper The Global Medical Technology Alliance (GMTA) represents medical technology associations whose members supply over 85 percent of the medical devices and diagnostics purchased annually
HTA Position Paper The Global Medical Technology Alliance (GMTA) represents medical technology associations whose members supply over 85 percent of the medical devices and diagnostics purchased annually
Higher Education Contribution to Health Science Innovation
 Scottish University of the Year 2017 Higher Education Contribution to Health Science Innovation Professor Sir Pete Downes Principal, University of Dundee Lead Member for Health, Universities Scotland 28
Scottish University of the Year 2017 Higher Education Contribution to Health Science Innovation Professor Sir Pete Downes Principal, University of Dundee Lead Member for Health, Universities Scotland 28
Offshore Renewable Energy Catapult
 Offshore Renewable Energy 7 s s: A long-term vision for innovation & growth The centres have been set up to make real changes to the way innovation happens in the UK to make things faster, less risky and
Offshore Renewable Energy 7 s s: A long-term vision for innovation & growth The centres have been set up to make real changes to the way innovation happens in the UK to make things faster, less risky and
AT THE SERVICE OF HEALTH AN ORIGINAL ENGINEERING OFFICE CLINICAL AFFAIRS R&D DEPARTMENT & DESIGN TECHNICAL ASSISTANCE
 REGULATORY ASSISTANCE QUALITY ASSISTANCE R&D DEPARTMENT & DESIGN TECHNICAL ASSISTANCE CLINICAL AFFAIRS AN ORIGINAL ENGINEERING OFFICE AT THE SERVICE OF HEALTH HTTP://EN.BIOTIKA.UNIV-FCOMTE.FR MESSAGE FROM
REGULATORY ASSISTANCE QUALITY ASSISTANCE R&D DEPARTMENT & DESIGN TECHNICAL ASSISTANCE CLINICAL AFFAIRS AN ORIGINAL ENGINEERING OFFICE AT THE SERVICE OF HEALTH HTTP://EN.BIOTIKA.UNIV-FCOMTE.FR MESSAGE FROM
Chemionix Solutions. Outsourcing. Engineering. Drafting
 Chemionix Solutions. Outsourcing. Engineering. Drafting Chemionix Your Outsourcing Partner Companies have been outsourcing since time began. It is only recently that business processes can be outsourced
Chemionix Solutions. Outsourcing. Engineering. Drafting Chemionix Your Outsourcing Partner Companies have been outsourcing since time began. It is only recently that business processes can be outsourced
Parkinson s World A transformational project by The Cure Parkinson s Trust
 Parkinson s World A transformational project by The Cure Parkinson s Trust Executive Summary During my time working in the Parkinson s field, I have come to realise there is a patent lack of communication
Parkinson s World A transformational project by The Cure Parkinson s Trust Executive Summary During my time working in the Parkinson s field, I have come to realise there is a patent lack of communication
Surrey Knowledge Transfer Account
 Surrey Knowledge Transfer Account Innovation Powered. Innovation Powered Innovation is vital if the UK is to remain competitive on the world stage. The University of Surrey has a track record of successful
Surrey Knowledge Transfer Account Innovation Powered. Innovation Powered Innovation is vital if the UK is to remain competitive on the world stage. The University of Surrey has a track record of successful
Written Submission for the Pre-Budget Consultations in Advance of the 2019 Budget By: The Danish Life Sciences Forum
 Written Submission for the Pre-Budget Consultations in Advance of the 2019 Budget By: The Danish Life Sciences Forum List of recommendations: Recommendation 1: That the government creates a Life Sciences
Written Submission for the Pre-Budget Consultations in Advance of the 2019 Budget By: The Danish Life Sciences Forum List of recommendations: Recommendation 1: That the government creates a Life Sciences
Horizon Societal Challenge 1: Health, demographic change and wellbeing. Jeremy Bray DG Research & Innovation European Commission
 Horizon 2020 Societal Challenge 1: Health, demographic change and wellbeing Jeremy Bray DG Research & Innovation European Commission National NHS day Leeds, UK, 29 January 2014 What is Horizon 2020? The
Horizon 2020 Societal Challenge 1: Health, demographic change and wellbeing Jeremy Bray DG Research & Innovation European Commission National NHS day Leeds, UK, 29 January 2014 What is Horizon 2020? The
GALILEO Research and Development Activities. Second Call. Area 3. Statement of Work
 GALILEO Research and Development Activities Second Call Area 3 Innovation by Small and Medium Enterprises Statement of Work Rue du Luxembourg, 3 B 1000 Brussels Tel +32 2 507 80 00 Fax +32 2 507 80 01
GALILEO Research and Development Activities Second Call Area 3 Innovation by Small and Medium Enterprises Statement of Work Rue du Luxembourg, 3 B 1000 Brussels Tel +32 2 507 80 00 Fax +32 2 507 80 01
Nepean Rubber & Plastics
 Nepean Rubber & Plastics I n n o v a t i v e I d e a s a n d S o l u t i o n s C u s t o m i s e d T o Y o u r N e e d s History and Growth of Nepean Rubber & Plastics Our business has grown strongly over
Nepean Rubber & Plastics I n n o v a t i v e I d e a s a n d S o l u t i o n s C u s t o m i s e d T o Y o u r N e e d s History and Growth of Nepean Rubber & Plastics Our business has grown strongly over
MedTech Europe position on future EU cooperation on Health Technology Assessment (21 March 2017)
 MedTech Europe position on future EU cooperation on Health Technology Assessment (21 March 2017) Table of Contents Executive Summary...3 The need for healthcare reform...4 The medical technology industry
MedTech Europe position on future EU cooperation on Health Technology Assessment (21 March 2017) Table of Contents Executive Summary...3 The need for healthcare reform...4 The medical technology industry
INSPIRING TECHNICAL EXCELLENCE
 INSPIRING TECHNICAL EXCELLENCE A new world-class training facility for Oman Introducing TPO Takatuf Petrofac Oman (TPO) is a new joint venture founded by Takatuf, the Human Capital solutions provider,
INSPIRING TECHNICAL EXCELLENCE A new world-class training facility for Oman Introducing TPO Takatuf Petrofac Oman (TPO) is a new joint venture founded by Takatuf, the Human Capital solutions provider,
Horizon Work Programme Leadership in enabling and industrial technologies - Introduction
 EN Horizon 2020 Work Programme 2018-2020 5. Leadership in enabling and industrial technologies - Introduction Important notice on the Horizon 2020 Work Programme This Work Programme covers 2018, 2019 and
EN Horizon 2020 Work Programme 2018-2020 5. Leadership in enabling and industrial technologies - Introduction Important notice on the Horizon 2020 Work Programme This Work Programme covers 2018, 2019 and
IMPROVING PATIENTS SELF-INJECTION EXPERIENCE
 IMPROVING PATIENTS SELF-INJECTION EXPERIENCE Jeff Lettman, Senior Research & Design Engineer, and Josh Hopkins, Engineering Manager,, explain how they worked with a client to develop a product that would
IMPROVING PATIENTS SELF-INJECTION EXPERIENCE Jeff Lettman, Senior Research & Design Engineer, and Josh Hopkins, Engineering Manager,, explain how they worked with a client to develop a product that would
MISSISSIPPI POLYMER INSTITUTE: Real World Solutions to Your Technical Challenges
 MISSISSIPPI POLYMER INSTITUTE: Real World Solutions to Your Technical Challenges MPI is an ISO/IEC 17025 accredited, full-service resource for polymer and material science-related industries. Established
MISSISSIPPI POLYMER INSTITUTE: Real World Solutions to Your Technical Challenges MPI is an ISO/IEC 17025 accredited, full-service resource for polymer and material science-related industries. Established
GE Healthcare. Vivid S5. Cardiovascular ultrasound system
 GE Healthcare Vivid S5 Cardiovascular ultrasound system Versatility. It s a new design concept one that leverages our miniaturization expertise gained from the Vivid i and our performance expertise of
GE Healthcare Vivid S5 Cardiovascular ultrasound system Versatility. It s a new design concept one that leverages our miniaturization expertise gained from the Vivid i and our performance expertise of
Innovation Integrator
 Innovation Integrator Innovation Integrator The Centre for Process Innovation Making successful innovation happen The Centre for Process Innovation From innovation to commercialisation The High Value Manufacturing
Innovation Integrator Innovation Integrator The Centre for Process Innovation Making successful innovation happen The Centre for Process Innovation From innovation to commercialisation The High Value Manufacturing
Offshore Renewables Institute
 Offshore Renewables Institute Introducing the Offshore Renewables Institute (ORI) About Us We bring together experts and expertise from across a broad university partnership: the University of Dundee;
Offshore Renewables Institute Introducing the Offshore Renewables Institute (ORI) About Us We bring together experts and expertise from across a broad university partnership: the University of Dundee;
Medical Device Usability Engineering. Product and Service Design Innovation Consultancy
 Medical Device Usability Engineering Product and Service Design Innovation Consultancy Delivering Meaningful Innovation PDD services Our integration of multiple disciplines means we offer a comprehensive
Medical Device Usability Engineering Product and Service Design Innovation Consultancy Delivering Meaningful Innovation PDD services Our integration of multiple disciplines means we offer a comprehensive
WAVE & TIDAL SERVICES WAVE & TIDAL
 WAVE & TIDAL SERVICES WAVE & TIDAL ore.catapult.org.uk 3 The UK s flagship technology innovation and research centre for offshore wind, wave and tidal energy 1 The Offshore Renewable Energy (ORE) Catapult
WAVE & TIDAL SERVICES WAVE & TIDAL ore.catapult.org.uk 3 The UK s flagship technology innovation and research centre for offshore wind, wave and tidal energy 1 The Offshore Renewable Energy (ORE) Catapult
Infrared Screening. with TotalVision anatomy software
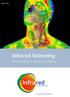 Infrared Screening with TotalVision anatomy software Unlimited possibilities with our high-quality infrared screening systems Energetic Health Systems leads the fi eld in infrared screening and is the
Infrared Screening with TotalVision anatomy software Unlimited possibilities with our high-quality infrared screening systems Energetic Health Systems leads the fi eld in infrared screening and is the
International Forensic Services
 International Forensic Services Right People. Delivering Results. Experienced scientists delivering forensic effectiveness, unquestionable integrity, focused customer service and value for money. Strengthening
International Forensic Services Right People. Delivering Results. Experienced scientists delivering forensic effectiveness, unquestionable integrity, focused customer service and value for money. Strengthening
September 18, 2017 Special Called Meeting of the U. T. System Board of Regents - Meeting of the Board
 AGENDA SPECIAL CALLED TELEPHONE MEETING of THE UNIVERSITY OF TEXAS SYSTEM BOARD OF REGENTS September 18, 2017 Austin, Texas Page CONVENE THE BOARD IN OPEN SESSION TO RECESS TO EXECUTIVE SESSION PURSUANT
AGENDA SPECIAL CALLED TELEPHONE MEETING of THE UNIVERSITY OF TEXAS SYSTEM BOARD OF REGENTS September 18, 2017 Austin, Texas Page CONVENE THE BOARD IN OPEN SESSION TO RECESS TO EXECUTIVE SESSION PURSUANT
Phantoms in Medical Physics (RT) U. Oelfke. Division of Radiotherapy & Imaging
 in partnership with Phantoms in Medical Physics (RT) U. Oelfke Division of Radiotherapy & Imaging uwe.oelfke@icr.ac.uk Making the discoveries that defeat cancer 1. Introduction What is a phantom? Wiki:
in partnership with Phantoms in Medical Physics (RT) U. Oelfke Division of Radiotherapy & Imaging uwe.oelfke@icr.ac.uk Making the discoveries that defeat cancer 1. Introduction What is a phantom? Wiki:
A Vision for Better Prototyping
 1 The external case of the prototype Gonioscope was built on a 3D printer. A Vision for Better Prototyping Nidek Technologies Accelerates Clinical Trial Evaluations And Time-To-Market With 3D Printing
1 The external case of the prototype Gonioscope was built on a 3D printer. A Vision for Better Prototyping Nidek Technologies Accelerates Clinical Trial Evaluations And Time-To-Market With 3D Printing
AUTO INJECTORS & PEN INJECTORS: A USER-CENTRIC DESIGN APPROACH
 AUTO INJECTORS & PEN INJECTORS: A USER-CENTRIC DESIGN APPROACH In this article, SHL Group provides insights into their injection device design processes and culture, emphasising their focus on the patient
AUTO INJECTORS & PEN INJECTORS: A USER-CENTRIC DESIGN APPROACH In this article, SHL Group provides insights into their injection device design processes and culture, emphasising their focus on the patient
HDR UK & Digital Innovation Hubs Introduction. 22 nd November 2018
 HDR UK & Digital Innovation Hubs Introduction 22 nd November 2018 Health Data Research UK s vision To create a thriving, high-energy UK-wide network of inter-disciplinary research expertise that will:
HDR UK & Digital Innovation Hubs Introduction 22 nd November 2018 Health Data Research UK s vision To create a thriving, high-energy UK-wide network of inter-disciplinary research expertise that will:
Tianjin Onehealth Medical Instruments. Onehealth Your surgical solutions partner
 Tianjin Onehealth Medical Instruments Introduction Tianjin Onehealth Medical Instruments Co Ltd is a part of KXBD Group which was established in 2006. Onehealth has a long term and massive investment strategy
Tianjin Onehealth Medical Instruments Introduction Tianjin Onehealth Medical Instruments Co Ltd is a part of KXBD Group which was established in 2006. Onehealth has a long term and massive investment strategy
INTRODUCTION TO PORTICOS. Product design for the way we re designed
 INTRODUCTION TO PORTICOS Product design for the way we re designed 2013 General Sales Presentation ABOUT PORTICOS EXECUTIVE SUMMARY Porticos is a full service product design and development firm located
INTRODUCTION TO PORTICOS Product design for the way we re designed 2013 General Sales Presentation ABOUT PORTICOS EXECUTIVE SUMMARY Porticos is a full service product design and development firm located
The petroleum industry, internationalisation, 11 and technology development. Industry development and internationalisation
 The petroleum industry, internationalisation, employment 11 and technology development Industry development and internationalisation Employment in the petroleum sector The significance of technology development
The petroleum industry, internationalisation, employment 11 and technology development Industry development and internationalisation Employment in the petroleum sector The significance of technology development
EU Support for SME Innovation: The SME Instrument
 Audit preview Information on an upcoming audit EU Support for SME Innovation: The SME Instrument April 2019 2 Traditionally, start-ups and small and medium-sized enterprises (SMEs) in the EU have faced
Audit preview Information on an upcoming audit EU Support for SME Innovation: The SME Instrument April 2019 2 Traditionally, start-ups and small and medium-sized enterprises (SMEs) in the EU have faced
Effective engineering solutions
 Effective engineering solutions As one of the UK s leading engineering companies, Strata Technology designs, manufactures, commissions, inspects and maintains bespoke laboratory bench top instruments,
Effective engineering solutions As one of the UK s leading engineering companies, Strata Technology designs, manufactures, commissions, inspects and maintains bespoke laboratory bench top instruments,
THE BIOMEDICAL ENGINEERING TEACHING & INNOVATION CENTER. at Boston University s College of Engineering
 THE BIOMEDICAL ENGINEERING TEACHING & INNOVATION CENTER at Boston University s College of Engineering The vision At Boston University s College of Engineering, we intend to create an exciting new resource
THE BIOMEDICAL ENGINEERING TEACHING & INNOVATION CENTER at Boston University s College of Engineering The vision At Boston University s College of Engineering, we intend to create an exciting new resource
Before I talk through the strategy itself, I want to tell you more about why
 This presentation was given by Dr Annette Bramley, the Healthcare Technologies Theme leader. She thanked Professor Nelson and Professor Taylor for their presentations, and thanked everyone at the meeting
This presentation was given by Dr Annette Bramley, the Healthcare Technologies Theme leader. She thanked Professor Nelson and Professor Taylor for their presentations, and thanked everyone at the meeting
Model Based Design Of Medical Devices
 Model Based Design Of Medical Devices A Tata Elxsi Perspective Tata Elxsi s Solutions - Medical Electronics Abstract Modeling and Simulation (M&S) is an important tool that may be employed in the end-to-end
Model Based Design Of Medical Devices A Tata Elxsi Perspective Tata Elxsi s Solutions - Medical Electronics Abstract Modeling and Simulation (M&S) is an important tool that may be employed in the end-to-end
Digital Reality TM changes everything
 F E B R U A R Y 2 0 1 8 Digital Reality TM changes everything Step into the future What are we talking about? Virtual Reality VR is an entirely digital world that completely immerses the user in an environment
F E B R U A R Y 2 0 1 8 Digital Reality TM changes everything Step into the future What are we talking about? Virtual Reality VR is an entirely digital world that completely immerses the user in an environment
The Centre for Process Innovation
 The Centre for Process Innovation The Centre for Process Innovation From innovation to commercialisation The future inspired... The High Value Manufacturing Catapult is a partnership between industry and
The Centre for Process Innovation The Centre for Process Innovation From innovation to commercialisation The future inspired... The High Value Manufacturing Catapult is a partnership between industry and
Convergence and Differentiation within the Framework of European Scientific and Technical Cooperation on HTA
 EUnetHTA European network for Health Technology Assessment Convergence and Differentiation within the Framework of European Scientific and Technical Cooperation on HTA University of Tokyo, October 24,
EUnetHTA European network for Health Technology Assessment Convergence and Differentiation within the Framework of European Scientific and Technical Cooperation on HTA University of Tokyo, October 24,
Radiate Engineering & Design
 Radiate Engineering & Design Advanced development of lightweight products and disruptive innovation Radiate Engineering & Design is a vertically-integrated composite expert, product developer, and leading
Radiate Engineering & Design Advanced development of lightweight products and disruptive innovation Radiate Engineering & Design is a vertically-integrated composite expert, product developer, and leading
Acquisition of MST Medical Surgery Technologies Ltd:
 Acquisition of MST Medical Surgery Technologies Ltd: Meaningfully Bolsters Senhance Platform Innovation to Further Advance Digital Laparoscopy September 24, 2018 2 FORWARD LOOKING STATEMENTS This presentation
Acquisition of MST Medical Surgery Technologies Ltd: Meaningfully Bolsters Senhance Platform Innovation to Further Advance Digital Laparoscopy September 24, 2018 2 FORWARD LOOKING STATEMENTS This presentation
Lifting Engineering. Capability & Experience
 Lifting Engineering Capability & Experience Results for our customers The WorleyParsons consulting group is a collaborative, multidisciplinary team with a broad range of strategic, technical and project
Lifting Engineering Capability & Experience Results for our customers The WorleyParsons consulting group is a collaborative, multidisciplinary team with a broad range of strategic, technical and project
Additive Manufacturing: A New Frontier for Simulation
 BEST PRACTICES Additive Manufacturing: A New Frontier for Simulation ADDITIVE MANUFACTURING popularly known as 3D printing is poised to revolutionize both engineering and production. With its capability
BEST PRACTICES Additive Manufacturing: A New Frontier for Simulation ADDITIVE MANUFACTURING popularly known as 3D printing is poised to revolutionize both engineering and production. With its capability
UGEO H60. Performance in Style. Features
 UGEO H60 Performance in Style The UGEO H60 implements superior performance with new design principles of simplicity and lightness. Its 10.1" touchscreen improves usability while its 18.5" LED monitor enhances
UGEO H60 Performance in Style The UGEO H60 implements superior performance with new design principles of simplicity and lightness. Its 10.1" touchscreen improves usability while its 18.5" LED monitor enhances
WHY ACCOUNTANCY & SOCIAL DESIGN
 OPEN DESIGN STUDIO WHY ACCOUNTANCY & SOCIAL DESIGN Last year, we launched a ground-breaking partnership with the Royal Society of Art, which explored the future of our society and outlined a vision for
OPEN DESIGN STUDIO WHY ACCOUNTANCY & SOCIAL DESIGN Last year, we launched a ground-breaking partnership with the Royal Society of Art, which explored the future of our society and outlined a vision for
CORPORATE OVERVIEW BREAKING THERMAL BARRIERS
 CORPORATE OVERVIEW BREAKING THERMAL BARRIERS Breaking Thermal Barriers Higher performance and greater electronic densities don t have to mean higher temperatures when you have Thermacore technologies and
CORPORATE OVERVIEW BREAKING THERMAL BARRIERS Breaking Thermal Barriers Higher performance and greater electronic densities don t have to mean higher temperatures when you have Thermacore technologies and
We turn good ideas into great products
 Introduction to Porticos www.porticos.net We turn good ideas into great products Executive Summary Porticos is a full service Mechanical Engineering and Design Firm located in Research Triangle Park, NC,
Introduction to Porticos www.porticos.net We turn good ideas into great products Executive Summary Porticos is a full service Mechanical Engineering and Design Firm located in Research Triangle Park, NC,
From late-life reservoir management through to final permanent abandonment, we create bespoke solutions to meet your specific well requirements.
 From late-life reservoir management through to final permanent abandonment, we create bespoke solutions to meet your specific well requirements. LATE-LIFE RESERVOIR MANAGEMENT All oil and gas fields will
From late-life reservoir management through to final permanent abandonment, we create bespoke solutions to meet your specific well requirements. LATE-LIFE RESERVOIR MANAGEMENT All oil and gas fields will
Keywords: Synairgen plc, Southampton, collaboration, spin-out, asthma, COPD
 Professor Stephen Holgate is a member of the Infection, Inflammation and Repair Division in the University of Southampton School of Medicine. He is a co-founder and non-executive director of Synairgen
Professor Stephen Holgate is a member of the Infection, Inflammation and Repair Division in the University of Southampton School of Medicine. He is a co-founder and non-executive director of Synairgen
Engineering Services. When it s critical
 Engineering Services When it s critical When it s critical Ferno Engineering - when it s critical From small beginnings as a supplier of ambulance stretchers 36 years ago, Ferno Australia now develops,
Engineering Services When it s critical When it s critical Ferno Engineering - when it s critical From small beginnings as a supplier of ambulance stretchers 36 years ago, Ferno Australia now develops,
What you should know about us... robot systems complete robot cells temperature control units complete cooling units
 What you should know about us... robot systems complete robot cells temperature control units complete cooling units Expertise by tradition For decades REMAK has developing and manufacturing been handling
What you should know about us... robot systems complete robot cells temperature control units complete cooling units Expertise by tradition For decades REMAK has developing and manufacturing been handling
Advanced Technology Solutions. Microwave Materials
 Advanced Technology Solutions Microwave Materials BAE SYSTEMS is an international company engaged in the development, delivery and support of advanced defence systems in the air, on land, at sea and in
Advanced Technology Solutions Microwave Materials BAE SYSTEMS is an international company engaged in the development, delivery and support of advanced defence systems in the air, on land, at sea and in
Conclusions on the future of information and communication technologies research, innovation and infrastructures
 COUNCIL OF THE EUROPEAN UNION Conclusions on the future of information and communication technologies research, innovation and infrastructures 2982nd COMPETITIVESS (Internal market, Industry and Research)
COUNCIL OF THE EUROPEAN UNION Conclusions on the future of information and communication technologies research, innovation and infrastructures 2982nd COMPETITIVESS (Internal market, Industry and Research)
Defence and security engineering
 Defence and security engineering 2018-2019 Symposia Symposia at Shrivenham provides a forum to Government agencies, military and civilian, industry and research establishments for the exploration and exchange
Defence and security engineering 2018-2019 Symposia Symposia at Shrivenham provides a forum to Government agencies, military and civilian, industry and research establishments for the exploration and exchange
University-Industry Collaboration Is It a Real Opportunity? Ömer Rüştü Ergen
 Ömer Rüştü Ergen R&D at Ford Otosan Developing automotive products, services and technologies >1500 engineers 3 R&D Centers Advanced laboratories and state of the art infrastructure 464 Mil. TL annual
Ömer Rüştü Ergen R&D at Ford Otosan Developing automotive products, services and technologies >1500 engineers 3 R&D Centers Advanced laboratories and state of the art infrastructure 464 Mil. TL annual
Climate Change Innovation and Technology Framework 2017
 Climate Change Innovation and Technology Framework 2017 Advancing Alberta s environmental performance and diversification through investments in innovation and technology Table of Contents 2 Message from
Climate Change Innovation and Technology Framework 2017 Advancing Alberta s environmental performance and diversification through investments in innovation and technology Table of Contents 2 Message from
Regulatory Science and Innovation: FDA s Role in Transformation of the MCM Enterprise
 Regulatory Science and Innovation: FDA s Role in Transformation of the MCM Enterprise Jesse L. Goodman, MD, MPH Chief Scientist and Deputy Commissioner for Science and Public Health (Acting), presentation
Regulatory Science and Innovation: FDA s Role in Transformation of the MCM Enterprise Jesse L. Goodman, MD, MPH Chief Scientist and Deputy Commissioner for Science and Public Health (Acting), presentation
Evolution of Research Consortia Increasing Clinical Focus
 1 July 11, 2015 Evolution of Research Consortia Increasing Clinical Focus Elekta Synergy Research Partnerships Lead to development of Synergy Systems Served as model for other technology research groups
1 July 11, 2015 Evolution of Research Consortia Increasing Clinical Focus Elekta Synergy Research Partnerships Lead to development of Synergy Systems Served as model for other technology research groups
GE Healthcare LOGIQ P3. Staying ahead of the curve
 GE Healthcare LOGIQ P3 Staying ahead of the curve Ultrasound Expertise. Enhanced. GE is a trusted partner of many healthcare providers around the world. GE s TruScan technology, incorporating SmartScan,
GE Healthcare LOGIQ P3 Staying ahead of the curve Ultrasound Expertise. Enhanced. GE is a trusted partner of many healthcare providers around the world. GE s TruScan technology, incorporating SmartScan,
Medical Devices cyber risks and threats
 Medical Devices cyber risks and threats David Grainger Senior Medical Device Specialist MHRA The challenges of software medical device regulation. david.grainger@mhra.gov.uk Current framework 1998 In Vitro
Medical Devices cyber risks and threats David Grainger Senior Medical Device Specialist MHRA The challenges of software medical device regulation. david.grainger@mhra.gov.uk Current framework 1998 In Vitro
MENTORS REGULATORY AFFAIRS. Christophe AMIEL Head of Medical Device & Digital, Voisin Consulting Life Sciences STRATEGY/BUSINESS
 Hosted by : INNOLABS IDEAS CONTEST 21 st -22 nd June, Paris MENTORS REGULATORY AFFAIRS Christophe Amiel is heading the design and operational implementation of international regulatory strategies as Senior
Hosted by : INNOLABS IDEAS CONTEST 21 st -22 nd June, Paris MENTORS REGULATORY AFFAIRS Christophe Amiel is heading the design and operational implementation of international regulatory strategies as Senior
Technology Leadership Course Descriptions
 ENG BE 700 A1 Advanced Biomedical Design and Development (two semesters, eight credits) Significant advances in medical technology require a profound understanding of clinical needs, the engineering skills
ENG BE 700 A1 Advanced Biomedical Design and Development (two semesters, eight credits) Significant advances in medical technology require a profound understanding of clinical needs, the engineering skills
Industrial Innovation Information Days Brussels 3-4 October 2017
 Industrial Innovation Information Days Brussels 3-4 October 2017 NMBP Programme Parallel Sessions OPEN INNOVATION TEST BEDS Calls 2018/2019 Helene CHRAYE, HoU Unit D3 DG Research & Innovation A joint presentation
Industrial Innovation Information Days Brussels 3-4 October 2017 NMBP Programme Parallel Sessions OPEN INNOVATION TEST BEDS Calls 2018/2019 Helene CHRAYE, HoU Unit D3 DG Research & Innovation A joint presentation
Making a healthier world
 Medical Components With 200 years of expertise in platinum group metals, Johnson Matthey offers medical device manufacturers the security of supply to source, manage, fabricate, and recycle materials for
Medical Components With 200 years of expertise in platinum group metals, Johnson Matthey offers medical device manufacturers the security of supply to source, manage, fabricate, and recycle materials for
Innovation for success
 Innovation for success Success in the medical sector Thin film substrates for medical implants Retinal implants for Retina Implant AG, Germany Our mission: To restore sight to blind people and thus increase
Innovation for success Success in the medical sector Thin film substrates for medical implants Retinal implants for Retina Implant AG, Germany Our mission: To restore sight to blind people and thus increase
Perspectives of Innovative Small Companies on the Industry s Prospects for 2012 and Beyond
 Perspectives of Innovative Small Companies on the Industry s Prospects for 2012 and Beyond ACI Forum on Structuring, Negotiating and Managing Life Sciences Collaborations and Acquisitions February 28,
Perspectives of Innovative Small Companies on the Industry s Prospects for 2012 and Beyond ACI Forum on Structuring, Negotiating and Managing Life Sciences Collaborations and Acquisitions February 28,
Global Source Ventures, LLC Introduction. Antonius Schuh Managing Partner Stephen Zaniboni Managing Partner
 Global Source Ventures, LLC Introduction Antonius Schuh Managing Partner Stephen Zaniboni Managing Partner Introduction to Global Source Ventures GSV provides seed capital to emerging companies with emphasis
Global Source Ventures, LLC Introduction Antonius Schuh Managing Partner Stephen Zaniboni Managing Partner Introduction to Global Source Ventures GSV provides seed capital to emerging companies with emphasis
Innovations in Health: Approaches from the Regional Innovation Funds
 The Young Foundation March 2010 Innovations in Health: Approaches from the Regional Innovation Funds Geoff Mulgan, Young Foundation with the Regional Innovation Funds Advisory Service (RIFAS) team March
The Young Foundation March 2010 Innovations in Health: Approaches from the Regional Innovation Funds Geoff Mulgan, Young Foundation with the Regional Innovation Funds Advisory Service (RIFAS) team March
Draft executive summaries to target groups on industrial energy efficiency and material substitution in carbonintensive
 Technology Executive Committee 29 August 2017 Fifteenth meeting Bonn, Germany, 12 15 September 2017 Draft executive summaries to target groups on industrial energy efficiency and material substitution
Technology Executive Committee 29 August 2017 Fifteenth meeting Bonn, Germany, 12 15 September 2017 Draft executive summaries to target groups on industrial energy efficiency and material substitution
Rick Legleiter Appointed Chief Executive Officer, Chairman Succession and Board Renewal Process
 Universal Biosensors, Inc. ARBN 121 559 993 1 Corporate Avenue Rowville Victoria 3178 Australia Telephone +61 3 9213 9000 Facsimile +61 3 9213 9099 Email info@universalbiosensors.com www.universalbiosensors.com
Universal Biosensors, Inc. ARBN 121 559 993 1 Corporate Avenue Rowville Victoria 3178 Australia Telephone +61 3 9213 9000 Facsimile +61 3 9213 9099 Email info@universalbiosensors.com www.universalbiosensors.com
Regulatory Science For Innovation
 Regulatory Science For Innovation Fergal Donnelly European Commission Directorate-General for Research & Innovation Directorate 'Industrial Technologies' Unit 'Advanced Materials and Nanotechnology' The
Regulatory Science For Innovation Fergal Donnelly European Commission Directorate-General for Research & Innovation Directorate 'Industrial Technologies' Unit 'Advanced Materials and Nanotechnology' The
IoT in Health and Social Care
 IoT in Health and Social Care Preserving Privacy: Good Practice Brief NOVEMBER 2017 Produced by Contents Introduction... 3 The DASH Project... 4 Why the Need for Guidelines?... 5 The Guidelines... 6 DASH
IoT in Health and Social Care Preserving Privacy: Good Practice Brief NOVEMBER 2017 Produced by Contents Introduction... 3 The DASH Project... 4 Why the Need for Guidelines?... 5 The Guidelines... 6 DASH
