SAMPLE AUTO13-A2. February This document identifies important factors that designers
|
|
|
- Lilian Curtis
- 5 years ago
- Views:
Transcription
1 February 2003 Laboratory Instruments and Data Management Systems: Design of Software User Interfaces and End-User Software Systems Validation, Operation, and Monitoring; Approved Guideline Second Edition This document identifies important factors that designers and laboratory managers should consider when developing new software-driven systems and selecting software user interfaces. Also included are simple rules to help prepare validation protocols for assessing the functionality and dependability of software. A guideline for global application developed through the Clinical and Laboratory Standards Institute consensus process.
2 Clinical and Laboratory Standards Institute Setting the standard for quality in clinical laboratory testing around the world. The Clinical and Laboratory Standards Institute (CLSI) is a not-for-profit membership organization that brings together the varied perspectives and expertise of the worldwide laboratory community for the advancement of a common cause: to foster excellence in laboratory medicine by developing and implementing clinical laboratory standards and guidelines that help laboratories fulfill their responsibilities with efficiency, effectiveness, and global applicability. Consensus Process Consensus the substantial agreement by materially affected, competent, and interested parties is core to the development of all CLSI documents. It does not always connote unanimous agreement, but does mean that the participants in the development of a consensus document have considered and resolved all relevant objections and accept the resulting agreement. Commenting on Documents CLSI documents undergo periodic evaluation and modification to keep pace with advancements in technologies, procedures, methods, and protocols affecting the laboratory or health care. CLSI s consensus process depends on experts who volunteer to serve as contributing authors and/or as participants in the reviewing and commenting process. At the end of each comment period, the committee that developed the document is obligated to review all comments, respond in writing to all substantive comments, and revise the draft document as appropriate. Comments on published CLSI documents are equally essential, and may be submitted by anyone, at any time, on any document. All comments are addressed according to the consensus process by a committee of experts. Appeals Process If it is believed that an objection has not been adequately addressed, the process for appeals is documented in the CLSI Standards Development Policies and Process document. All comments and responses submitted on draft and published documents are retained on file at CLSI and are available upon request. Get Involved Volunteer! Do you use CLSI documents in your workplace? Do you see room for improvement? Would you like to get involved in the revision process? Or maybe you see a need to develop a new document for an emerging technology? CLSI wants to hear from you. We are always looking for volunteers. By donating your time and talents to improve the standards that affect your own work, you will play an active role in improving public health across the globe. For further information on committee participation or to submit comments, contact CLSI. Clinical and Laboratory Standards Institute 950 West Valley Road, Suite 2500 Wayne, PA USA P: F: standard@clsi.org
3 ISBN Vol. 23 No. 4 Formerly GP19-A2 ISSN Vol. 23 No. 4 Laboratory Instruments and Data Management Systems: Design of Software User Interfaces and End-User Software Systems Validation, Operation, and Monitoring; Approved Guideline Second Edition Volume 23 Number 4 Paul J. Mountain, M.Sc., M.T. (ASCP) Andrzej J. Knafel, Ph.D. Suzanne H. Butch, M.A., M.T. (ASCP), SBB Louis Dunka, Jr., Ph.D. Rodney S. Markin, M.D., Ph.D. David O Bryan, Ph.D. Abstract Clinical and Laboratory Standards Institute document Laboratory Instruments and Data Management Systems: Design of Software User Interfaces and End-User Software Systems Validation, Operation, and Monitoring; Approved Guideline Second Edition provides user interface design recommendations that make new software programs easier for laboratory personnel to learn and use. Additionally, the guideline addresses the preparation and execution of validation plans for software purchased from manufacturers; custom software commissioned by the laboratory; or in-house applications developed to collect, interpret, or report laboratory, patient, or quality control information. Clinical and Laboratory Standards Institute (CLSI). Laboratory Instruments and Data Management Systems: Design of Software User Interfaces and End-User Software Systems Validation, Operation, and Monitoring; Approved Guideline Second Edition. CLSI document (ISBN ). Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania USA, The Clinical and Laboratory Standards Institute consensus process, which is the mechanism for moving a document through two or more levels of review by the health care community, is an ongoing process. Users should expect revised editions of any given document. Because rapid changes in technology may affect the procedures, methods, and protocols in a standard or guideline, users should replace outdated editions with the current editions of CLSI documents. Current editions are listed in the CLSI catalog and posted on our website at If your organization is not a member and would like to become one, and to request a copy of the catalog, contact us at: Telephone: ; Fax: ; customerservice@clsi.org; Website:
4 Number 4 Copyright 2003 Clinical and Laboratory Standards Institute. Except as stated below, any reproduction of content from a CLSI copyrighted standard, guideline, companion product, or other material requires express written consent from CLSI. All rights reserved. Interested parties may send permission requests to permissions@clsi.org. CLSI hereby grants permission to each individual member or purchaser to make a single reproduction of this publication for use in its laboratory procedure manual at a single site. To request permission to use this publication in any other manner, permissions@clsi.org. Suggested Citation CLSI. Laboratory Instruments and Data Management Systems: Design of Software User Interfaces and End-User Software Systems Validation, Operation, and Monitoring; Approved Guideline Second Edition. CLSI document. Wayne, PA: Clinical and Laboratory Standards Institute; Proposed Guideline November 1994 Approved Guideline December 1995 Approved Guideline Second Edition February 2003 ISBN ISSN ii
5 Volume 23 Contents Abstract... i Committee Membership... iii The Quality System Approach... viii Foreword... ix 1 Scope Introduction... 2 Part I: Design of User Interfaces Overview Scope Intended Audience Design and Evaluation Process Task Automation Definition: What is Task Automation? Discussion Task Analysis Design Guidelines for Task Analysis and Task Allocation Guidelines for Evaluating Task Automation in Laboratory Systems User Control Consistency Definition: What is Consistency? Discussion Design Guidelines That Promote Consistency Guidelines for Evaluating Consistency in Laboratory Systems Reserved Keys Navigation Definition Discussion Guidelines for Assessing Navigation Screens and Reports Definition Discussion Error Handling: Recognition, Prevention, and Recovery Definition Discussion Perceptual Errors Cognitive Errors Motor Errors Error Recognition v
6 Number 4 Contents (Continued) 9.7 Guidelines for Error Prevention and Recovery Security Part II: Systems Validation, Operation, and Monitoring Overview Scope Intended Audience Definitions Resource Requirements Planning Project Management Assess How the Project Will Affect the Operation of the Institution Specifications and Documentation Testing Strategy Approach (Component vs. System) Environment (Active vs. Static Tests) Sign-Off and Approvals System Access Points Data Collection Methods Acceptance Criteria Test Plan Error Analysis and Handling Evaluation of Results Operation and Monitoring Security Records Change Requests Training Audits References Additional References Appendix A. Format of a Software Specification Document Appendix B. Data Entry Validation Form Appendix C. Sample Test Case Report Format Appendix D. Sample Test Case #1 Expired Crossmatch Appendix E. Sample Test Case #2 - ABO/Rh Compatibility Table Review Appendix F. Sample Test Case #3 ABO/Rh Compatibility Test vi
7 Volume 23 Contents (Continued) Appendix G. Sample Test Case #4 Patient s Historical Blood Type Appendix H. Hospital Information System (HIS) Interface Validation Appendix I. HIS Interface Quality Assurance Documentation Appendix J. Worksheet Calculations Validation Appendix K. Example of Validation of Worksheet Calculations Summary of Comments and Area Committee Responses Related NCCLS Publications vii
8 Number 4 The Quality System Approach NCCLS subscribes to a quality system approach in the development of standards and guidelines, which facilitates project management; defines a document structure via a template; and provides a process to identify needed documents through a gap analysis. The approach is based on the model presented in the most current edition of NCCLS document HS1 A Quality System Model for Health Care. The quality system approach applies a core set of quality system essentials (QSEs), basic to any organization, to all operations in any healthcare service s path of workflow. The QSEs provide the framework for delivery of any type of product or service, serving as a manager s guide. The quality system essentials (QSEs) are: Documents & Records Equipment Information Management Process Improvement Organization Purchasing & Inventory Occurrence Management Service & Satisfaction Personnel Process Control Assessment Facilities & Safety addresses the quality system essentials (QSEs) indicated by an "X." For a description of the other NCCLS documents listed in the grid, please refer to the Related NCCLS Publications section at the end of the document. Documents & Records X GP2-A4 Organization X Personnel X GP21-A Equipment Purchasing & Inventory Process Control Information Management Occurrence Management Assessment Process Improvement Service & Satisfaction Facilities & Safety X X X X X X X X GP18-A Adapted from NCCLS document HS1 A Quality System Model for Health Care. viii
9 Volume 23 Foreword Many in vitro diagnostic instruments and specimen processing devices in the hospital laboratory are now computer controlled and actuated. Instrument manufacturers design and develop the embedded software that provides the functionality for these systems. The computer software presents an interface to the user that can make operation of the instrument a reasonable task to learn and perform. In addition to other computerized information management systems, laboratorians in the modern clinical laboratory use a host of different computer controlled instruments, each with its own (sometimes unique) user interface. This multitude of different user interfaces affects learning, training, productivity, and potentially patient outcomes in the laboratory. Part I (Sections 3 through 10) provides guidelines for the design of software user interfaces that offer developers a common, consistent design direction for laboratory device applications. Whether they purchase software from third-party vendors or develop the software themselves, laboratory personnel are responsible for the validation of this software. Part II (Sections 11 through 14) contains recommendations for preparation and execution of validation plans for software packages. Key Words Data management systems, laboratory instrumentation, process control, software design, software validation, user interface ix
10 Volume 23 Laboratory Instruments and Data Management Systems: Design of Software User Interfaces and End-User Software Systems Validation, Operation, and Monitoring; Approved Guideline Second Edition 1 Scope The scope of this document is limited to issues that affect ease of learning and the ease of use of software user 3,4 interfaces. Although there is a need to improve the hardware interface between operators and instruments (e.g., keyboard, mouse, touch screen, printer, reports, voice, and light pens used when adding or removing patient samples, reagents, and waste), these topics are not within the scope of this guideline. is not intended as a tool to be used in the selection, recommendation, or judgment of the suitability of specific input/output technologies, since these may change rapidly. Since it is described elsewhere, 1,2 the transfer of electronic information between information and/or automation systems (such as between a laboratory information system or laboratory automation system software and an instrument) is not the subject of this document. This document identifies the most important factors that designers and laboratory managers should consider during the development of a new software-actuated system and when selecting a software user interface intended to improve the ease of learning and use within the clinical laboratory. Without attempting to provide a comprehensive or exhaustive discussion of software user interfaces or trying to define an identical appearance for user interfaces by describing a single, detailed design solution, this document addresses some common design elements. This discussion is intended to encourage manufacturers of laboratory instruments and specimen handling devices to develop more uniform software user interfaces within their product lines. The primary focus of this document is the software user interface within the centralized laboratory environment. The guidelines presented in this document are not directly constructed for point-of-care, physician-office, or over-the-counter devices, although many of the principles discussed apply to these devices as well. The primary focus of this document is software user interfaces on instruments, although the guidelines also apply to interfaces on laboratory systems and other associated information systems used in the laboratory. These design guidelines and examples are not, however, universally applicable to all laboratory systems. Implementation of a specific design depends on the size, complexity, and cost of a device, as justified by its intended use. This document provides some simple rules to help laboratory personnel prepare validation protocols that fulfill the laboratory's obligation to test and verify the functionality and dependability of its software. This document does not advocate relieving software developers of their duty to validate the software products that they develop; AUTO13 offers assistance to the purchaser when no other means of validation is available. Developers should refer to sources such as IEEE sources for specific guidelines for software system validation. Clinical and Laboratory Standards Institute. All rights reserved. 1
11 Number 4 2 Introduction This document provides guidelines for the design of software user interfaces (also referred to as humancomputer interaction ) that focus on two related areas. First, it identifies elements of a user interface that are most likely to facilitate ease of learning and use. Second, it provides some direction as to commonalities in look and feel that further the development of an easier-to-use software interface, as well as improve its design. The subcommittee believes that will initiate a dialogue between users (e.g., laboratorians) and developers of software. This dialogue is intended to bring laboratorians and manufacturers closer to an agreement as to what elements should be consistent among software user interfaces if the goals of ease of learning and use are to be accomplished. In the modern clinical laboratory, it is necessary for clinical laboratory personnel to learn and use various user interfaces for many computer-actuated devices, including clinical instruments and laboratory information systems. The user interfaces of computer-driven devices help to manage the workflow of laboratory personnel; for this reason, they have a major effect on productivity and the effectiveness of laboratory processes. Because these interfaces have such an effect on productivity, it is important to ensure that they are easy to learn and use. This is especially true at a time when laboratorians are frequently asked to perform more tasks, as well as new tasks (e.g., CLIA compliance), with fewer or less highly skilled staff members. In concept, easy-to-learn and easy-to-use software user interfaces have the potential to improve laboratory productivity. The reality is that user software interfaces are often difficult to learn and use. First, software user interfaces are different from one manufacturer to another; frequently, they are also different from one product to another from the same manufacturer. This makes it difficult for technicians to learn one interface and apply that learning to another interface. Second, many interfaces are difficult to use, because current knowledge in software user interface design is not applied during the development of the products. Furthermore, many products are not tested by their designers for ease of learning and use. This document will help designers of computer-actuated diagnostic instrument systems identify those areas of a software user interface where commonalties in design, rather than product differentiation, can provide the greatest benefit to the laboratorians. also provides guidelines for purchasers of computer-actuated diagnostic instrument systems that establish common benchmarks by which to judge the suitability of a potential acquisition; these guidelines move laboratorians closer to an important goal a decrease in the total cost of laboratory operations. also offers help to laboratorians in the preparation and execution of end-user validation plans for software developed to collect, interpret, or report laboratory, patient, or quality control information from various sources, including: software purchased from vendors; custom software commissioned by the laboratory; and applications constructed in-house. There was some discussion about splitting this document into two independent guidelines, because the audience for each part was different: product software designers for Part 1 and end users for Part 2. While there is some logic to that approach, it was felt that the two sections should be kept in one document to promote active discussion and interaction between both target audiences and to maximize the acceptance and utility of any software interface to laboratorians. Software validation is a requirement of laboratories before it can be implemented, and user interface designers must understand the laboratory's need for effective, easy-to-use and easy-to-validate software 2 Clinical and Laboratory Standards Institute. All rights reserved.
12 Volume 23 Related NCCLS Publications * GP2-A4 Clinical Laboratory Technical Procedure Manuals; Approved Guideline Fourth Edition (2002). This document provides guidance on development, review, approval, management, and use of policy, process, and procedure documents in the laboratory testing community. GP18-A Laboratory Design; Approved Guideline (1998). This guideline provides a foundation of information about laboratory design elements that can be used to help define the issues being considered when designing a laboratory. GP21-A Training Verification for Laboratory Personnel; Approved Guideline (1995). This document provides background and recommends an infrastructure for developing a training verification program that meets quality/regulatory objectives. * Proposed- and tentative-level documents are being advanced through the NCCLS consensus process; therefore, readers should refer to the most recent editions. Clinical and Laboratory Standards Institute. All rights reserved. 69
13 PL As we continue to set the global standard for quality in laboratory testing, we re adding initiatives to bring even more value to our members and customers. E Explore the Latest Offerings from CLSI! Power Forward with this Official Interactive Guide Fundamentals for implementing a quality management system in the clinical laboratory. The value of a CLSI membership begins with significant discounts up to 70% off on our trusted clinical laboratory standards and guidelines, but the benefits extend far beyond cost savings: Benefits to Industry Contribute to Standards that Streamline Product Review Processes Access a Deep Network of Customers, Peers, Regulators, and Industry Leaders Raise Your Organization s Profile in the Clinical Laboratory Community Benefits to Laboratories Directly Influence CLSI Standards to Ensure they are Practical and Achievable Access Globally Recognized Standards for Accreditation Preparedness Help Drive Higher Levels of Patient Care Quality All Over the World Benefits to Government Aid in the Development of Consensus Standards that can Impact Legislation Connect with Over 2,000 Influential Organizations Across the Global Laboratory Community Help Laboratories Provide Safe and Effective Care of the Highest Quality and Value About CLSI M The Clinical and Laboratory Standards Institute Visit the CLSI U Education Center SA Where we provide the convenient and cost-effective education resources that laboratories need to put CLSI standards into practice, including webinars, workshops, and more. Shop Our Online Products e CLIPSE TM Ultimate Access Including eclipse Ultimate Access, CLSI s cloud-based, online portal that makes it easy to access our standards and guidelines anytime, anywhere. Introducing CLSI s New Membership Opportunities (CLSI) is a not-for-profit membership organization that brings together the varied perspectives and expertise of the worldwide laboratory community for the advancement of a common cause: to foster excellence in laboratory medicine by developing and implementing clinical standards and guidelines 950 West Valley Road, Suite 2500, Wayne, PA P: Toll Free (US): F: E: membership@clsi.org that help laboratories fulfill their responsibilities with efficiency, effectiveness, and global applicability. More Options. More Benefits. More Value. Join in Our Mission to Improve Health Care Outcomes We ve made it even easier for your organization to take full advantage of the standards resources and networking opportunities available through membership with CLSI. Find Membership Opportunities See the options that make it even easier for your organization to take full advantage of CLSI benefits and our unique membership value. For more information, visit today.
14 950 West Valley Road, Suite 2500, Wayne, PA USA ISBN P: Toll Free (US): F: E:
SURGERY STRATEGIC CLINICAL NETWORK EVIDENCE DECISION SUPPORT PROGRAM. New ideas & Improvements
 SURGERY STRATEGIC CLINICAL NETWORK EVIDENCE DECISION SUPPORT PROGRAM 2014 Revision (v3) New ideas & Improvements Department of Surgery Evidence Decision Support Program Resource Tool Box Regional Clinical
SURGERY STRATEGIC CLINICAL NETWORK EVIDENCE DECISION SUPPORT PROGRAM 2014 Revision (v3) New ideas & Improvements Department of Surgery Evidence Decision Support Program Resource Tool Box Regional Clinical
This document is a preview generated by EVS
 TECHNICAL REPORT ISO/TR 28380-2 First edition 2014-02-15 Health informatics IHE global standards adoption Part 2: Integration and content profiles Informatique de santé Adoption des normes globales IHE
TECHNICAL REPORT ISO/TR 28380-2 First edition 2014-02-15 Health informatics IHE global standards adoption Part 2: Integration and content profiles Informatique de santé Adoption des normes globales IHE
Brief to the. Senate Standing Committee on Social Affairs, Science and Technology. Dr. Eliot A. Phillipson President and CEO
 Brief to the Senate Standing Committee on Social Affairs, Science and Technology Dr. Eliot A. Phillipson President and CEO June 14, 2010 Table of Contents Role of the Canada Foundation for Innovation (CFI)...1
Brief to the Senate Standing Committee on Social Affairs, Science and Technology Dr. Eliot A. Phillipson President and CEO June 14, 2010 Table of Contents Role of the Canada Foundation for Innovation (CFI)...1
Guidance for Industry and FDA Staff Use of Symbols on Labels and in Labeling of In Vitro Diagnostic Devices Intended for Professional Use
 Guidance for Industry and FDA Staff Use of Symbols on Labels and in Labeling of In Vitro Diagnostic Devices Intended for Professional Use Document issued on: November 30, 2004 The draft of this document
Guidance for Industry and FDA Staff Use of Symbols on Labels and in Labeling of In Vitro Diagnostic Devices Intended for Professional Use Document issued on: November 30, 2004 The draft of this document
Pan-Canadian Trust Framework Overview
 Pan-Canadian Trust Framework Overview A collaborative approach to developing a Pan- Canadian Trust Framework Authors: DIACC Trust Framework Expert Committee August 2016 Abstract: The purpose of this document
Pan-Canadian Trust Framework Overview A collaborative approach to developing a Pan- Canadian Trust Framework Authors: DIACC Trust Framework Expert Committee August 2016 Abstract: The purpose of this document
Canadian Technology Accreditation Criteria (CTAC) ELECTROMECHANICAL ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC)
 Canadian Technology Accreditation Criteria (CTAC) ELECTROMECHANICAL ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC) Preamble These CTAC are applicable to programs having titles
Canadian Technology Accreditation Criteria (CTAC) ELECTROMECHANICAL ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC) Preamble These CTAC are applicable to programs having titles
IEEE-SA Overview. Don Wright IEEE Standards Association Treasurer. CCSA/IEEE-SA Internet of Things Workshop 5 June 2012 Beijing, China
 IEEE-SA Overview Don Wright IEEE Standards Association Treasurer CCSA/IEEE-SA Internet of Things Workshop 5 June 2012 Beijing, China IEEE Today The world s largest professional association advancing technology
IEEE-SA Overview Don Wright IEEE Standards Association Treasurer CCSA/IEEE-SA Internet of Things Workshop 5 June 2012 Beijing, China IEEE Today The world s largest professional association advancing technology
National Standard of the People s Republic of China
 ICS 01.120 A 00 National Standard of the People s Republic of China GB/T XXXXX.1 201X Association standardization Part 1: Guidelines for good practice Click here to add logos consistent with international
ICS 01.120 A 00 National Standard of the People s Republic of China GB/T XXXXX.1 201X Association standardization Part 1: Guidelines for good practice Click here to add logos consistent with international
American Nuclear Society
 American Nuclear Society 1 Unraveling the Mystery of Consensus Standards Presented by: The American Nuclear Society Standards Committee January 31, 2017 Copyright 2017 by American Nuclear Society Purpose
American Nuclear Society 1 Unraveling the Mystery of Consensus Standards Presented by: The American Nuclear Society Standards Committee January 31, 2017 Copyright 2017 by American Nuclear Society Purpose
Enpr EMA. Enpr-EMA. European Network of Paediatric Research at the European Medicines Agency
 Enpr EMA European Network of Paediatric Research at the European Medicines Agency Enpr-EMA European Network of Paediatric Research at the European Medicines Agency An agency of the European Union Enpr
Enpr EMA European Network of Paediatric Research at the European Medicines Agency Enpr-EMA European Network of Paediatric Research at the European Medicines Agency An agency of the European Union Enpr
WIPO Development Agenda
 WIPO Development Agenda 2 The WIPO Development Agenda aims to ensure that development considerations form an integral part of WIPO s work. As such, it is a cross-cutting issue which touches upon all sectors
WIPO Development Agenda 2 The WIPO Development Agenda aims to ensure that development considerations form an integral part of WIPO s work. As such, it is a cross-cutting issue which touches upon all sectors
Canadian Technology Accreditation Criteria (CTAC) POWER SYSTEMS ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC)
 Canadian Technology Accreditation Criteria (CTAC) POWER SYSTEMS ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC) Preamble These CTAC are applicable to programs having titles involving
Canadian Technology Accreditation Criteria (CTAC) POWER SYSTEMS ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC) Preamble These CTAC are applicable to programs having titles involving
ASME NQA-1 Quality Assurance Requirements for Nuclear Facility Applications. Prague, CR July 7 8, 2014
 ASME NQA-1 Quality Assurance Requirements for Nuclear Facility Applications Prague, CR July 7 8, 2014 ASME NQA-1 Due to the expansion of the global supply chain and advances in technology, many codes and
ASME NQA-1 Quality Assurance Requirements for Nuclear Facility Applications Prague, CR July 7 8, 2014 ASME NQA-1 Due to the expansion of the global supply chain and advances in technology, many codes and
NEMA XR X-ray Equipment for Interventional Procedures User Quality Control Mode
 NEMA XR 27-2012 X-ray Equipment for Interventional Procedures User Quality Control Mode Published by: National Electrical Manufacturers Association 1300 North 17th Street, Suite 1752 Rosslyn, Virginia
NEMA XR 27-2012 X-ray Equipment for Interventional Procedures User Quality Control Mode Published by: National Electrical Manufacturers Association 1300 North 17th Street, Suite 1752 Rosslyn, Virginia
NHS England CCG Authorisation
 NHS England CCG Authorisation Post Authorisation December 2013 Review Conditions Report CCG name: Vale of York CCG Wave: 3 Regional Operations Director Jon Develing Date report generated: 22/01/2014 Vale
NHS England CCG Authorisation Post Authorisation December 2013 Review Conditions Report CCG name: Vale of York CCG Wave: 3 Regional Operations Director Jon Develing Date report generated: 22/01/2014 Vale
Imagine your future lab. Designed using Virtual Reality and Computer Simulation
 Imagine your future lab Designed using Virtual Reality and Computer Simulation Bio At Roche Healthcare Consulting our talented professionals are committed to optimising patient care. Our diverse range
Imagine your future lab Designed using Virtual Reality and Computer Simulation Bio At Roche Healthcare Consulting our talented professionals are committed to optimising patient care. Our diverse range
CO-ORDINATION MECHANISMS FOR DIGITISATION POLICIES AND PROGRAMMES:
 CO-ORDINATION MECHANISMS FOR DIGITISATION POLICIES AND PROGRAMMES: NATIONAL REPRESENTATIVES GROUP (NRG) SUMMARY REPORT AND CONCLUSIONS OF THE MEETING OF 10 DECEMBER 2002 The third meeting of the NRG was
CO-ORDINATION MECHANISMS FOR DIGITISATION POLICIES AND PROGRAMMES: NATIONAL REPRESENTATIVES GROUP (NRG) SUMMARY REPORT AND CONCLUSIONS OF THE MEETING OF 10 DECEMBER 2002 The third meeting of the NRG was
Parenteral Nutrition Down Under Inc. (PNDU) Working with Pharmaceutical Companies Policy (Policy)
 Parenteral Nutrition Down Under Inc. (PNDU) Working with Pharmaceutical Companies Policy (Policy) BACKGROUND (Reason or Purpose) The purpose of this Policy is to provide clear principles and guidance about
Parenteral Nutrition Down Under Inc. (PNDU) Working with Pharmaceutical Companies Policy (Policy) BACKGROUND (Reason or Purpose) The purpose of this Policy is to provide clear principles and guidance about
NHS South Kent Coast. Clinical Commissioning Group. Complaints, Comments and Compliments Policy
 NHS South Kent Coast Clinical Commissioning Group Complaints, Comments and Compliments Policy Version: Version 1.6 Ratified by: Date ratified: Name of originator/author: Name of responsible committee/individual:
NHS South Kent Coast Clinical Commissioning Group Complaints, Comments and Compliments Policy Version: Version 1.6 Ratified by: Date ratified: Name of originator/author: Name of responsible committee/individual:
Convergence and Differentiation within the Framework of European Scientific and Technical Cooperation on HTA
 EUnetHTA European network for Health Technology Assessment Convergence and Differentiation within the Framework of European Scientific and Technical Cooperation on HTA University of Tokyo, October 24,
EUnetHTA European network for Health Technology Assessment Convergence and Differentiation within the Framework of European Scientific and Technical Cooperation on HTA University of Tokyo, October 24,
Fiscal 2007 Environmental Technology Verification Pilot Program Implementation Guidelines
 Fifth Edition Fiscal 2007 Environmental Technology Verification Pilot Program Implementation Guidelines April 2007 Ministry of the Environment, Japan First Edition: June 2003 Second Edition: May 2004 Third
Fifth Edition Fiscal 2007 Environmental Technology Verification Pilot Program Implementation Guidelines April 2007 Ministry of the Environment, Japan First Edition: June 2003 Second Edition: May 2004 Third
HP Laboratories. US Labor Rates for Directed Research Activities. Researcher Qualifications and Descriptions. HP Labs US Labor Rates
 HP Laboratories US Labor Rates for Directed Research Activities This note provides: Information about the job categories and job descriptions that apply to HP Laboratories (HP Labs) research, managerial
HP Laboratories US Labor Rates for Directed Research Activities This note provides: Information about the job categories and job descriptions that apply to HP Laboratories (HP Labs) research, managerial
Digitisation Plan
 Digitisation Plan 2016-2020 University of Sydney Library University of Sydney Library Digitisation Plan 2016-2020 Mission The University of Sydney Library Digitisation Plan 2016-20 sets out the aim and
Digitisation Plan 2016-2020 University of Sydney Library University of Sydney Library Digitisation Plan 2016-2020 Mission The University of Sydney Library Digitisation Plan 2016-20 sets out the aim and
MedTech Europe position on future EU cooperation on Health Technology Assessment (21 March 2017)
 MedTech Europe position on future EU cooperation on Health Technology Assessment (21 March 2017) Table of Contents Executive Summary...3 The need for healthcare reform...4 The medical technology industry
MedTech Europe position on future EU cooperation on Health Technology Assessment (21 March 2017) Table of Contents Executive Summary...3 The need for healthcare reform...4 The medical technology industry
Guidelines for the Stage of Implementation - Self-Assessment Activity
 GUIDELINES FOR PRIVACY AND INFORMATION MANAGEMENT (PIM) PROGRAM SELF-ASSESSMENT ACTIVITY Guidelines for the Stage of Implementation - Self-Assessment Activity PURPOSE This tool is for the use of school
GUIDELINES FOR PRIVACY AND INFORMATION MANAGEMENT (PIM) PROGRAM SELF-ASSESSMENT ACTIVITY Guidelines for the Stage of Implementation - Self-Assessment Activity PURPOSE This tool is for the use of school
Herts Valleys Clinical Commissioning Group. Review of NHS Herts Valleys CCG Constitution
 Herts Valleys Clinical Commissioning Group Review of NHS Herts Valleys CCG s constitution Agenda Item: 14 REPORT TO: HVCCG Board DATE of MEETING: 30 January 2014 SUBJECT: Review of NHS Herts Valleys CCG
Herts Valleys Clinical Commissioning Group Review of NHS Herts Valleys CCG s constitution Agenda Item: 14 REPORT TO: HVCCG Board DATE of MEETING: 30 January 2014 SUBJECT: Review of NHS Herts Valleys CCG
The 45 Adopted Recommendations under the WIPO Development Agenda
 The 45 Adopted Recommendations under the WIPO Development Agenda * Recommendations with an asterisk were identified by the 2007 General Assembly for immediate implementation Cluster A: Technical Assistance
The 45 Adopted Recommendations under the WIPO Development Agenda * Recommendations with an asterisk were identified by the 2007 General Assembly for immediate implementation Cluster A: Technical Assistance
Operational Objectives Outcomes Indicators
 UNEP/CBD/BS/COP-MOP/5/17 Page 106 ELEMENTS OF STRATEGIC PLAN FOR THE CARTAGENA PROTOCOL ON BIOSAFETY VISION Biological diversity is adequately protected from any adverse effects of living modified organisms
UNEP/CBD/BS/COP-MOP/5/17 Page 106 ELEMENTS OF STRATEGIC PLAN FOR THE CARTAGENA PROTOCOL ON BIOSAFETY VISION Biological diversity is adequately protected from any adverse effects of living modified organisms
Air Monitoring Directive Chapter 9: Reporting
 Air Monitoring Directive Chapter 9: Reporting Version Dec 16, 2016 Amends the original Air Monitoring Directive published June, 1989 Title: Air Monitoring Directive Chapter 9: Reporting Number: Program
Air Monitoring Directive Chapter 9: Reporting Version Dec 16, 2016 Amends the original Air Monitoring Directive published June, 1989 Title: Air Monitoring Directive Chapter 9: Reporting Number: Program
Type Approval JANUARY The electronic pdf version of this document found through is the officially binding version
 STANDARD FOR CERTIFICATION No. 1.2 Type Approval JANUARY 2013 The electronic pdf version of this document found through http://www.dnv.com is the officially binding version The content of this service
STANDARD FOR CERTIFICATION No. 1.2 Type Approval JANUARY 2013 The electronic pdf version of this document found through http://www.dnv.com is the officially binding version The content of this service
Australian/New Zealand Standard
 Australian/New Zealand Standard Quality management and quality assurance Vocabulary This Joint Australian/New Zealand Standard was prepared by Joint Technical Committee QR/7, Quality Terminology. It was
Australian/New Zealand Standard Quality management and quality assurance Vocabulary This Joint Australian/New Zealand Standard was prepared by Joint Technical Committee QR/7, Quality Terminology. It was
EXPLORATION DEVELOPMENT OPERATION CLOSURE
 i ABOUT THE INFOGRAPHIC THE MINERAL DEVELOPMENT CYCLE This is an interactive infographic that highlights key findings regarding risks and opportunities for building public confidence through the mineral
i ABOUT THE INFOGRAPHIC THE MINERAL DEVELOPMENT CYCLE This is an interactive infographic that highlights key findings regarding risks and opportunities for building public confidence through the mineral
This version has been archived. Find the current version at on the Current Documents page. Scientific Working Groups on.
 Scientific Working Groups on Digital Evidence and Imaging Technology SWGDE/SWGIT Guidelines & Recommendations for Training in Digital & Multimedia Evidence Disclaimer: As a condition to the use of this
Scientific Working Groups on Digital Evidence and Imaging Technology SWGDE/SWGIT Guidelines & Recommendations for Training in Digital & Multimedia Evidence Disclaimer: As a condition to the use of this
Our position. ICDPPC declaration on ethics and data protection in artificial intelligence
 ICDPPC declaration on ethics and data protection in artificial intelligence AmCham EU speaks for American companies committed to Europe on trade, investment and competitiveness issues. It aims to ensure
ICDPPC declaration on ethics and data protection in artificial intelligence AmCham EU speaks for American companies committed to Europe on trade, investment and competitiveness issues. It aims to ensure
Global citizenship at HP. Corporate accountability and governance. Overarching message
 Global citizenship at HP Overarching message With HP s global reach comes global responsibility. We take our role seriously by being an economic, intellectual and social asset to the communities in which
Global citizenship at HP Overarching message With HP s global reach comes global responsibility. We take our role seriously by being an economic, intellectual and social asset to the communities in which
What We Heard Report Inspection Modernization: The Case for Change Consultation from June 1 to July 31, 2012
 What We Heard Report Inspection Modernization: The Case for Change Consultation from June 1 to July 31, 2012 What We Heard Report: The Case for Change 1 Report of What We Heard: The Case for Change Consultation
What We Heard Report Inspection Modernization: The Case for Change Consultation from June 1 to July 31, 2012 What We Heard Report: The Case for Change 1 Report of What We Heard: The Case for Change Consultation
Issues in Emerging Health Technologies Bulletin Process
 Issues in Emerging Health Technologies Bulletin Process Updated: April 2015 Version 1.0 REVISION HISTORY Periodically, this document will be revised as part of ongoing process improvement activities. The
Issues in Emerging Health Technologies Bulletin Process Updated: April 2015 Version 1.0 REVISION HISTORY Periodically, this document will be revised as part of ongoing process improvement activities. The
COMMUNICATION FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT. pursuant to Article 294(6) of the Treaty on the Functioning of the European Union
 EUROPEAN COMMISSION Brussels, 9.3.2017 COM(2017) 129 final 2012/0266 (COD) COMMUNICATION FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT pursuant to Article 294(6) of the Treaty on the Functioning of the
EUROPEAN COMMISSION Brussels, 9.3.2017 COM(2017) 129 final 2012/0266 (COD) COMMUNICATION FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT pursuant to Article 294(6) of the Treaty on the Functioning of the
Charter of the Regional Technical Forum Policy Advisory Committee
 Phil Rockefeller Chair Washington Tom Karier Washington Henry Lorenzen Oregon Bill Bradbury Oregon W. Bill Booth Vice Chair Idaho James Yost Idaho Pat Smith Montana Jennifer Anders Montana Charter of the
Phil Rockefeller Chair Washington Tom Karier Washington Henry Lorenzen Oregon Bill Bradbury Oregon W. Bill Booth Vice Chair Idaho James Yost Idaho Pat Smith Montana Jennifer Anders Montana Charter of the
DNVGL-CP-0338 Edition October 2015
 CLASS PROGRAMME DNVGL-CP-0338 Edition October 2015 The electronic pdf version of this document, available free of charge from http://www.dnvgl.com, is the officially binding version. FOREWORD DNV GL class
CLASS PROGRAMME DNVGL-CP-0338 Edition October 2015 The electronic pdf version of this document, available free of charge from http://www.dnvgl.com, is the officially binding version. FOREWORD DNV GL class
CBD Request to WIPO on the Interrelation of Access to Genetic Resources and Disclosure Requirements
 CBD Request to WIPO on the Interrelation of Access to Genetic Resources and Disclosure Requirements Establishing an adequate framework for a WIPO Response 1 Table of Contents I. Introduction... 1 II. Supporting
CBD Request to WIPO on the Interrelation of Access to Genetic Resources and Disclosure Requirements Establishing an adequate framework for a WIPO Response 1 Table of Contents I. Introduction... 1 II. Supporting
Biomedical Equipment Technician
 Biomedical Equipment Technician Occupational Skill Stards Texas Skill Stards Board Recognized Critical Work Function 1. Install Biomedical Equipment 1.1 Receive, inspect inventory upon delivery 1.2 Deliver
Biomedical Equipment Technician Occupational Skill Stards Texas Skill Stards Board Recognized Critical Work Function 1. Install Biomedical Equipment 1.1 Receive, inspect inventory upon delivery 1.2 Deliver
AMERICAN NATIONAL STANDARD
 ANSI CGATS.5-2003 Supplement - 2005 AMERICAN NATIONAL STANDARD Graphic technology Graphic arts transmission densitometry Terminology, equations, image elements and procedures Supplement SECRETARIAT NPES
ANSI CGATS.5-2003 Supplement - 2005 AMERICAN NATIONAL STANDARD Graphic technology Graphic arts transmission densitometry Terminology, equations, image elements and procedures Supplement SECRETARIAT NPES
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 16142-1 First edition 2016-03-01 Medical devices Recognized essential principles of safety and performance of medical devices Part 1: General essential principles and additional
INTERNATIONAL STANDARD ISO 16142-1 First edition 2016-03-01 Medical devices Recognized essential principles of safety and performance of medical devices Part 1: General essential principles and additional
INTERNATIONAL ATOMIC ENERGY AGENCY 58TH GENERAL CONFERENCE (22 26 September 2014)
 TURKEY INTERNATIONAL ATOMIC ENERGY AGENCY 58TH GENERAL CONFERENCE (22 26 September 2014) Allow me at the outset to congratulate you on your assumption of the Presidency of the 58th Session of the IAEA
TURKEY INTERNATIONAL ATOMIC ENERGY AGENCY 58TH GENERAL CONFERENCE (22 26 September 2014) Allow me at the outset to congratulate you on your assumption of the Presidency of the 58th Session of the IAEA
NIHR / Wellcome Trust King s Clinical Research Facility. Guidance for Investigators
 NIHR / Wellcome Trust King s Clinical Research Facility Guidance for Investigators Introduction The NIHR Clinical Research Facilities provide dedicated facilities to support the delivery of externally-funded
NIHR / Wellcome Trust King s Clinical Research Facility Guidance for Investigators Introduction The NIHR Clinical Research Facilities provide dedicated facilities to support the delivery of externally-funded
Details of the Proposal
 Details of the Proposal Draft Model to Address the GDPR submitted by Coalition for Online Accountability This document addresses how the proposed model submitted by the Coalition for Online Accountability
Details of the Proposal Draft Model to Address the GDPR submitted by Coalition for Online Accountability This document addresses how the proposed model submitted by the Coalition for Online Accountability
Translation University of Tokyo Intellectual Property Policy
 Translation University of Tokyo Intellectual Property Policy February 17, 2004 Revised September 30, 2004 1. Objectives The University of Tokyo has acknowledged the roles entrusted to it by the people
Translation University of Tokyo Intellectual Property Policy February 17, 2004 Revised September 30, 2004 1. Objectives The University of Tokyo has acknowledged the roles entrusted to it by the people
Supporting medical technology development with the analytic hierarchy process Hummel, Janna Marchien
 University of Groningen Supporting medical technology development with the analytic hierarchy process Hummel, Janna Marchien IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
University of Groningen Supporting medical technology development with the analytic hierarchy process Hummel, Janna Marchien IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
A review of the role and costs of clinical commissioning groups
 A picture of the National Audit Office logo Report by the Comptroller and Auditor General NHS England A review of the role and costs of clinical commissioning groups HC 1783 SESSION 2017 2019 18 DECEMBER
A picture of the National Audit Office logo Report by the Comptroller and Auditor General NHS England A review of the role and costs of clinical commissioning groups HC 1783 SESSION 2017 2019 18 DECEMBER
Joint ILAC CIPM Communication regarding the. Accreditation of Calibration and Measurement Services. of National Metrology Institutes.
 Joint ILAC CIPM Communication regarding the Accreditation of Calibration and Measurement Services of National Metrology Institutes 7 March 2012 Authorship This document was prepared by the International
Joint ILAC CIPM Communication regarding the Accreditation of Calibration and Measurement Services of National Metrology Institutes 7 March 2012 Authorship This document was prepared by the International
(Non-legislative acts) DECISIONS
 4.12.2010 Official Journal of the European Union L 319/1 II (Non-legislative acts) DECISIONS COMMISSION DECISION of 9 November 2010 on modules for the procedures for assessment of conformity, suitability
4.12.2010 Official Journal of the European Union L 319/1 II (Non-legislative acts) DECISIONS COMMISSION DECISION of 9 November 2010 on modules for the procedures for assessment of conformity, suitability
CADTH HEALTH TECHNOLOGY MANAGEMENT PROGRAM Horizon Scanning Products and Services Processes
 CADTH HEALTH TECHNOLOGY MANAGEMENT PROGRAM Horizon Scanning Products and Services Processes Service Line: Health Technology Management Program Version: 1.0 Publication Date: September 2017 Report Length:
CADTH HEALTH TECHNOLOGY MANAGEMENT PROGRAM Horizon Scanning Products and Services Processes Service Line: Health Technology Management Program Version: 1.0 Publication Date: September 2017 Report Length:
ISO INTERNATIONAL STANDARD. Robots for industrial environments Safety requirements Part 1: Robot
 INTERNATIONAL STANDARD ISO 10218-1 First edition 2006-06-01 Robots for industrial environments Safety requirements Part 1: Robot Robots pour environnements industriels Exigences de sécurité Partie 1: Robot
INTERNATIONAL STANDARD ISO 10218-1 First edition 2006-06-01 Robots for industrial environments Safety requirements Part 1: Robot Robots pour environnements industriels Exigences de sécurité Partie 1: Robot
Translational scientist competency profile
 C-COMEND Competency profile for Translational Scientists C-COMEND is a two-year European training project supported by the Erasmus plus programme, which started on November 1st 2015. The overall objective
C-COMEND Competency profile for Translational Scientists C-COMEND is a two-year European training project supported by the Erasmus plus programme, which started on November 1st 2015. The overall objective
CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017
 CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017 1. Introduction The goals of the CARRA Publication and Presentation Guidelines are to: a) Promote timely and high-quality presentation
CARRA PUBLICATION AND PRESENTATION GUIDELINES Version April 20, 2017 1. Introduction The goals of the CARRA Publication and Presentation Guidelines are to: a) Promote timely and high-quality presentation
FRAMEWORK Advances in biomedical technology are
 TECHNOLOGY FRAMEWORK Advances in biomedical technology are occurring so rapidly that healthcare professionals can barely keep abreast of the changes. And these advances have cost hospitals dearly. They
TECHNOLOGY FRAMEWORK Advances in biomedical technology are occurring so rapidly that healthcare professionals can barely keep abreast of the changes. And these advances have cost hospitals dearly. They
Advances and Perspectives in Health Information Standards
 Advances and Perspectives in Health Information Standards HL7 Brazil June 14, 2018 W. Ed Hammond. Ph.D., FACMI, FAIMBE, FIMIA, FHL7, FIAHSI Director, Duke Center for Health Informatics Director, Applied
Advances and Perspectives in Health Information Standards HL7 Brazil June 14, 2018 W. Ed Hammond. Ph.D., FACMI, FAIMBE, FIMIA, FHL7, FIAHSI Director, Duke Center for Health Informatics Director, Applied
FY 2008 (October 1, 2007 September 30, 2008) NIMS Compliance Objectives and Metrics for Local Governments
 FY 2008 (October 1, 2007 September 30, 2008) NIMS Compliance Objectives and Metrics for Local Governments Introduction Homeland Security Presidential Directive (HSPD)- 5 Management of Domestic Incidents
FY 2008 (October 1, 2007 September 30, 2008) NIMS Compliance Objectives and Metrics for Local Governments Introduction Homeland Security Presidential Directive (HSPD)- 5 Management of Domestic Incidents
clarification to bring legal certainty to these issues have been voiced in various position papers and statements.
 ESR Statement on the European Commission s proposal for a Regulation on the protection of individuals with regard to the processing of personal data on the free movement of such data (General Data Protection
ESR Statement on the European Commission s proposal for a Regulation on the protection of individuals with regard to the processing of personal data on the free movement of such data (General Data Protection
Training that is standardized and supports the effective operations of NIIMS.
 HISTORY OF THE INCIDENT COMMAND SYSTEM In the early 1970's, Southern California experienced several devastating wildland fires. The overall cost and loss associated with these fires totaled $18 million
HISTORY OF THE INCIDENT COMMAND SYSTEM In the early 1970's, Southern California experienced several devastating wildland fires. The overall cost and loss associated with these fires totaled $18 million
Distribution Restriction Statement Approved for public release; distribution is unlimited.
 CEMP-RA Engineer Regulation 200-1-1 Department of the Army U.S. Army Corps of Engineers Washington, DC 20314-1000 ER 200-1-1 30 May 2000 Environmental Quality POLICY AND GENERAL REQUIREMENTS FOR THE ENVIRONMENTAL
CEMP-RA Engineer Regulation 200-1-1 Department of the Army U.S. Army Corps of Engineers Washington, DC 20314-1000 ER 200-1-1 30 May 2000 Environmental Quality POLICY AND GENERAL REQUIREMENTS FOR THE ENVIRONMENTAL
Stakeholder Involvement. Nuclear Issues. INSAG and IAEA perspective BASIS FOR KNOWN PUBLIC CONCERN. INSAG-20 Stakeholder Involvement in
 BASIS FOR KNOWN PUBLIC CONCERN Stakeholder Involvement in Nuclear issues: INSAG and IAEA perspective In general, at the heart of the public s concern is often an unwillingness to delegate power to centralized
BASIS FOR KNOWN PUBLIC CONCERN Stakeholder Involvement in Nuclear issues: INSAG and IAEA perspective In general, at the heart of the public s concern is often an unwillingness to delegate power to centralized
Technology Needs Assessment
 Technology Needs Assessment CII Research Summary 173-1 Executive Summary The Technology Needs Assessment Research Team was initiated to take a snapshot of current industry technology needs. As a result,
Technology Needs Assessment CII Research Summary 173-1 Executive Summary The Technology Needs Assessment Research Team was initiated to take a snapshot of current industry technology needs. As a result,
NEVADA DEPARTMENT OF TRANSPORTATION Addendum 3 to RFP July 28, 2017
 NEVADA DEPARTMENT OF TRANSPORTATION Addendum 3 to RFP 697-16-016 July 28, 2017 Reference is made to the Request for Proposal (RFP) to Service Providers for Nevada Shared Radio Replacement Project, upon
NEVADA DEPARTMENT OF TRANSPORTATION Addendum 3 to RFP 697-16-016 July 28, 2017 Reference is made to the Request for Proposal (RFP) to Service Providers for Nevada Shared Radio Replacement Project, upon
National Incident Management System
 National Incident Management System Overview Briefing September, 2006 Shelley S. Boone, II DHS-FEMA, Region IV Homeland Security Presidential Directive 5 National Incident Management System (NIMS) A consistent
National Incident Management System Overview Briefing September, 2006 Shelley S. Boone, II DHS-FEMA, Region IV Homeland Security Presidential Directive 5 National Incident Management System (NIMS) A consistent
Upstream Oil and Gas. Spill Prevention, Preparedness, Response, and Recovery. March 2013
 Upstream Oil and Gas Spill Prevention, Preparedness, Response, and Recovery March 2013 Canadian Association of Petroleum Producers (CAPP) Members explore for, develop and produce natural gas, natural gas
Upstream Oil and Gas Spill Prevention, Preparedness, Response, and Recovery March 2013 Canadian Association of Petroleum Producers (CAPP) Members explore for, develop and produce natural gas, natural gas
DRAFT. Cardiac Safety Research Consortium CSRC. Membership Committee Charter. 12September2018. Table of Contents
 DRAFT Cardiac Safety Research Consortium CSRC Membership Committee Charter 12September2018 Table of Contents Purpose of the CSRC Membership Charter and CSRC Membership Committee Composition, Responsibilities
DRAFT Cardiac Safety Research Consortium CSRC Membership Committee Charter 12September2018 Table of Contents Purpose of the CSRC Membership Charter and CSRC Membership Committee Composition, Responsibilities
RADIOLOGY August 2017
 USA EUROPE CHINA BRAZIL The current challenges faced by a Radiologist Too many patients 38% 45% 47% 53% Long working hours 36% 40% 50% 52% Troubleshooting IT and technical issues 14% 23% 28% 33% Limitations
USA EUROPE CHINA BRAZIL The current challenges faced by a Radiologist Too many patients 38% 45% 47% 53% Long working hours 36% 40% 50% 52% Troubleshooting IT and technical issues 14% 23% 28% 33% Limitations
TECHNICAL AND OPERATIONAL NOTE ON CHANGE MANAGEMENT OF GAMBLING TECHNICAL SYSTEMS AND APPROVAL OF THE SUBSTANTIAL CHANGES TO CRITICAL COMPONENTS.
 TECHNICAL AND OPERATIONAL NOTE ON CHANGE MANAGEMENT OF GAMBLING TECHNICAL SYSTEMS AND APPROVAL OF THE SUBSTANTIAL CHANGES TO CRITICAL COMPONENTS. 1. Document objective This note presents a help guide for
TECHNICAL AND OPERATIONAL NOTE ON CHANGE MANAGEMENT OF GAMBLING TECHNICAL SYSTEMS AND APPROVAL OF THE SUBSTANTIAL CHANGES TO CRITICAL COMPONENTS. 1. Document objective This note presents a help guide for
Project Status Update
 Project Status Update Reporting cycle: 1 October 2016 to 30 June 2017 (Year 1) Date: 13 July 2017 Designated Charity: Funded initiative: Snapshot overview: headspace National Youth Mental Health Foundation
Project Status Update Reporting cycle: 1 October 2016 to 30 June 2017 (Year 1) Date: 13 July 2017 Designated Charity: Funded initiative: Snapshot overview: headspace National Youth Mental Health Foundation
Adopting Standards For a Changing Health Environment
 Adopting Standards For a Changing Health Environment November 16, 2018 W. Ed Hammond. Ph.D., FACMI, FAIMBE, FIMIA, FHL7, FIAHSI Director, Duke Center for Health Informatics Director, Applied Informatics
Adopting Standards For a Changing Health Environment November 16, 2018 W. Ed Hammond. Ph.D., FACMI, FAIMBE, FIMIA, FHL7, FIAHSI Director, Duke Center for Health Informatics Director, Applied Informatics
THE ASEAN FRAMEWORK AGREEMENT ON ACCESS TO BIOLOGICAL AND GENETIC RESOURCES
 Draft Text 24 February 2000 THE ASEAN FRAMEWORK AGREEMENT ON ACCESS TO BIOLOGICAL AND GENETIC RESOURCES The Member States of the Association of South East Asian Nations (ASEAN) : CONSCIOUS of the fact
Draft Text 24 February 2000 THE ASEAN FRAMEWORK AGREEMENT ON ACCESS TO BIOLOGICAL AND GENETIC RESOURCES The Member States of the Association of South East Asian Nations (ASEAN) : CONSCIOUS of the fact
This is a preview - click here to buy the full publication
 TECHNICAL REPORT IEC/TR 62794 Edition 1.0 2012-11 colour inside Industrial-process measurement, control and automation Reference model for representation of production facilities (digital factory) INTERNATIONAL
TECHNICAL REPORT IEC/TR 62794 Edition 1.0 2012-11 colour inside Industrial-process measurement, control and automation Reference model for representation of production facilities (digital factory) INTERNATIONAL
COMMISSION RECOMMENDATION. of on access to and preservation of scientific information. {SWD(2012) 221 final} {SWD(2012) 222 final}
 EUROPEAN COMMISSION Brussels, 17.7.2012 C(2012) 4890 final COMMISSION RECOMMENDATION of 17.7.2012 on access to and preservation of scientific information {SWD(2012) 221 final} {SWD(2012) 222 final} EN
EUROPEAN COMMISSION Brussels, 17.7.2012 C(2012) 4890 final COMMISSION RECOMMENDATION of 17.7.2012 on access to and preservation of scientific information {SWD(2012) 221 final} {SWD(2012) 222 final} EN
Automated Digitization of Gram Stains. Centralized Reading. Decentralized Assessment. Improved Quality Management.
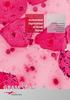 Automated Digitization of Gram Stains Centralized Reading. Decentralized Assessment. Improved Quality Management. A GROWING DEMAND Gram staining is the rapid, easy, and inexpensive method for the assessment
Automated Digitization of Gram Stains Centralized Reading. Decentralized Assessment. Improved Quality Management. A GROWING DEMAND Gram staining is the rapid, easy, and inexpensive method for the assessment
ASSESSMENT OF HOUSING QUALITY IN CONDOMINIUM DEVELOPMENTS IN SRI LANKA: A HOLISTIC APPROACH
 ASSESSMENT OF HOUSING QUALITY IN CONDOMINIUM DEVELOPMENTS IN SRI LANKA: A HOLISTIC APPROACH Dilrukshi Dilani Amarasiri Gunawardana (108495 H) Degree of Master of Science in Project Management Department
ASSESSMENT OF HOUSING QUALITY IN CONDOMINIUM DEVELOPMENTS IN SRI LANKA: A HOLISTIC APPROACH Dilrukshi Dilani Amarasiri Gunawardana (108495 H) Degree of Master of Science in Project Management Department
University of Massachusetts Amherst Libraries. Digital Preservation Policy, Version 1.3
 University of Massachusetts Amherst Libraries Digital Preservation Policy, Version 1.3 Purpose: The University of Massachusetts Amherst Libraries Digital Preservation Policy establishes a framework to
University of Massachusetts Amherst Libraries Digital Preservation Policy, Version 1.3 Purpose: The University of Massachusetts Amherst Libraries Digital Preservation Policy establishes a framework to
Global Alzheimer s Association Interactive Network. Imagine GAAIN
 Global Alzheimer s Association Interactive Network Imagine the possibilities if any scientist anywhere in the world could easily explore vast interlinked repositories of data on thousands of subjects with
Global Alzheimer s Association Interactive Network Imagine the possibilities if any scientist anywhere in the world could easily explore vast interlinked repositories of data on thousands of subjects with
PRINCIPLES AND CRITERIA FOR THE EVALUATION OF SCIENTIFIC ORGANISATIONS IN THE REPUBLIC OF CROATIA
 ashe Agency for Science and Higher Education PRINCIPLES AND CRITERIA FOR THE EVALUATION OF SCIENTIFIC ORGANISATIONS IN THE REPUBLIC OF CROATIA February 2013 Donje Svetice 38/5 10 000 Zagreb, Croatia T
ashe Agency for Science and Higher Education PRINCIPLES AND CRITERIA FOR THE EVALUATION OF SCIENTIFIC ORGANISATIONS IN THE REPUBLIC OF CROATIA February 2013 Donje Svetice 38/5 10 000 Zagreb, Croatia T
NIMS UPDATE 2017 RUPERT DENNIS, FEMA REGION IV, NIMS COORDINATOR. National Preparedness Directorate / National Integration Center.
 NIMS UPDATE 2017 RUPERT DENNIS, FEMA REGION IV, NIMS COORDINATOR National Preparedness Directorate / National Integration Center May 8, 2018 National Incident Management System (NIMS) Overview NIMS provides
NIMS UPDATE 2017 RUPERT DENNIS, FEMA REGION IV, NIMS COORDINATOR National Preparedness Directorate / National Integration Center May 8, 2018 National Incident Management System (NIMS) Overview NIMS provides
Guide to the Requirements for Public Information and Disclosure GD-99.3
 Guide to the Requirements for Public Information and Disclosure GD-99.3 November 2010 Guide to the Requirements for Public Information and Disclosure Guidance Document GD-99.3 Minister of Public Works
Guide to the Requirements for Public Information and Disclosure GD-99.3 November 2010 Guide to the Requirements for Public Information and Disclosure Guidance Document GD-99.3 Minister of Public Works
THE USE OF A SAFETY CASE APPROACH TO SUPPORT DECISION MAKING IN DESIGN
 THE USE OF A SAFETY CASE APPROACH TO SUPPORT DECISION MAKING IN DESIGN W.A.T. Alder and J. Perkins Binnie Black and Veatch, Redhill, UK In many of the high hazard industries the safety case and safety
THE USE OF A SAFETY CASE APPROACH TO SUPPORT DECISION MAKING IN DESIGN W.A.T. Alder and J. Perkins Binnie Black and Veatch, Redhill, UK In many of the high hazard industries the safety case and safety
Library Special Collections Mission, Principles, and Directions. Introduction
 Introduction The old proverb tells us the only constant is change and indeed UCLA Library Special Collections (LSC) exists during a time of great transformation. We are a new unit, created in 2010 to unify
Introduction The old proverb tells us the only constant is change and indeed UCLA Library Special Collections (LSC) exists during a time of great transformation. We are a new unit, created in 2010 to unify
IEEE Broadband Wireless Access Working Group < Proposed PAR to convert P802.16d from Amendment to Revision
 2003-05-15 IEEE C802.16-03/08 Project Title Date Submitted IEEE 802.16 Broadband Wireless Access Working Group Proposed PAR to convert P802.16d from Amendment to Revision 2003-05-15
2003-05-15 IEEE C802.16-03/08 Project Title Date Submitted IEEE 802.16 Broadband Wireless Access Working Group Proposed PAR to convert P802.16d from Amendment to Revision 2003-05-15
SAFEGUARDING ADULTS FRAMEWORK. Prevention and effective responses to neglect, harm and abuse is a basic requirement of modern health care services.
 SAFEGUARDING ADULTS FRAMEWORK Introduction Prevention and effective responses to neglect, harm and abuse is a basic requirement of modern health care services. Safeguarding adults involves a range of additional
SAFEGUARDING ADULTS FRAMEWORK Introduction Prevention and effective responses to neglect, harm and abuse is a basic requirement of modern health care services. Safeguarding adults involves a range of additional
SPACE SITUATIONAL AWARENESS: IT S NOT JUST ABOUT THE ALGORITHMS
 SPACE SITUATIONAL AWARENESS: IT S NOT JUST ABOUT THE ALGORITHMS William P. Schonberg Missouri University of Science & Technology wschon@mst.edu Yanping Guo The Johns Hopkins University, Applied Physics
SPACE SITUATIONAL AWARENESS: IT S NOT JUST ABOUT THE ALGORITHMS William P. Schonberg Missouri University of Science & Technology wschon@mst.edu Yanping Guo The Johns Hopkins University, Applied Physics
This document is a preview generated by EVS
 TECHNICAL REPORT IEC/TR 80002-1 Edition 1.0 2009-09 colour inside Medical device software Part 1: Guidance on the application of ISO 14971 to medical device software IEC/TR 80002-1:2009(E) THIS PUBLICATION
TECHNICAL REPORT IEC/TR 80002-1 Edition 1.0 2009-09 colour inside Medical device software Part 1: Guidance on the application of ISO 14971 to medical device software IEC/TR 80002-1:2009(E) THIS PUBLICATION
Canadian Technology Accreditation Criteria (CTAC) MECHANICAL ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC)
 Preamble Canadian Technology Accreditation Criteria (CTAC) MECHANICAL ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC) These CTAC are applicable to programs having titles involving
Preamble Canadian Technology Accreditation Criteria (CTAC) MECHANICAL ENGINEERING TECHNOLOGY - TECHNICIAN Technology Accreditation Canada (TAC) These CTAC are applicable to programs having titles involving
Standard of Knowledge, Skill and Competence for Practice as an Architectural Technologist
 Standard of Knowledge, Skill and Competence for Practice as an Architectural Technologist RIAI 2010 Contents Foreword 2 Background 3 Development of the Standard.4 Use of the Standard..5 Reading and interpreting
Standard of Knowledge, Skill and Competence for Practice as an Architectural Technologist RIAI 2010 Contents Foreword 2 Background 3 Development of the Standard.4 Use of the Standard..5 Reading and interpreting
CIVIC EPISTEMOLOGIES Civic Epistemologies: Development of a Roadmap for Citizen Researchers in the age of Digital Culture Workshop on the Roadmap
 This project has received funding from the European Union s Seventh Framework Programme for research, technological development and demonstration under grant agreement no 632694 CIVIC EPISTEMOLOGIES Civic
This project has received funding from the European Union s Seventh Framework Programme for research, technological development and demonstration under grant agreement no 632694 CIVIC EPISTEMOLOGIES Civic
Why Projects Fail. NASA s Mars Climate Orbiter Project. Case Study. A High Tech, High Profile Failure
 Why Projects Fail NASA s Mars Climate Orbiter Project Case Study A High Tech, High Profile Failure But the lessons learned are of value to all projects June 2003 Of interest to: Principals, Vice-Chancellors,
Why Projects Fail NASA s Mars Climate Orbiter Project Case Study A High Tech, High Profile Failure But the lessons learned are of value to all projects June 2003 Of interest to: Principals, Vice-Chancellors,
PART XIII Fidelity and Security of Measurement Data
 INSTITUTE OF PETROLEUM PETROLEUM MEASUREMENT MANUAL PART XIII Fidelity and Security of Measurement Data Section 3 Electrical and/or Electronic Data Capture Systems for Flow Metering INSTITUTE OF PETROLEUM
INSTITUTE OF PETROLEUM PETROLEUM MEASUREMENT MANUAL PART XIII Fidelity and Security of Measurement Data Section 3 Electrical and/or Electronic Data Capture Systems for Flow Metering INSTITUTE OF PETROLEUM
1 SERVICE DESCRIPTION
 DNV GL management system ICP Product Certification ICP 4-6-3-5-CR Document number: ICP 4-6-3-5-CR Valid for: All in DNV GL Revision: 2 Date: 2017-05-05 Resp. unit/author: Torgny Segerstedt Reviewed by:
DNV GL management system ICP Product Certification ICP 4-6-3-5-CR Document number: ICP 4-6-3-5-CR Valid for: All in DNV GL Revision: 2 Date: 2017-05-05 Resp. unit/author: Torgny Segerstedt Reviewed by:
Ninth Annual DPHP Meeting. October 9, 2013
 Ninth Annual DPHP Meeting October 9, 2013 1 NHSPI Speaker Introductions James Blumenstock Chief Program Officer Association of State and Territorial Health Officials (ASTHO) Ali Khan, MD, MPH Director,
Ninth Annual DPHP Meeting October 9, 2013 1 NHSPI Speaker Introductions James Blumenstock Chief Program Officer Association of State and Territorial Health Officials (ASTHO) Ali Khan, MD, MPH Director,
Public Art Network Best Practice Goals and Guidelines
 Public Art Network Best Practice Goals and Guidelines The Public Art Network (PAN) Council of Americans for the Arts appreciates the need to identify best practice goals and guidelines for the field. The
Public Art Network Best Practice Goals and Guidelines The Public Art Network (PAN) Council of Americans for the Arts appreciates the need to identify best practice goals and guidelines for the field. The
Konica Minolta has made a commitment as a manufacturer to deliver first-rate products and services that are trusted and needed all over the world.
 7 Special Feature: Corporate Social Responsibility (CSR) Promotion through Business Activities Konica Minolta has made a commitment as a manufacturer to deliver first-rate products and services that are
7 Special Feature: Corporate Social Responsibility (CSR) Promotion through Business Activities Konica Minolta has made a commitment as a manufacturer to deliver first-rate products and services that are
The Electronic Equipment Stewardship Regulations
 ELECTRONIC EQUIPMENT STEWARDSHIP E-10.21 REG 6 1 The Electronic Equipment Stewardship Regulations being Chapter E-10.22 Reg 6 (effective May 1, 2018). NOTE: This consolidation is not official. Amendments
ELECTRONIC EQUIPMENT STEWARDSHIP E-10.21 REG 6 1 The Electronic Equipment Stewardship Regulations being Chapter E-10.22 Reg 6 (effective May 1, 2018). NOTE: This consolidation is not official. Amendments
Public Information and Disclosure RD/GD-99.3
 Public Information and Disclosure RD/GD-99.3 March, 2012 Public Information and Disclosure Regulatory Document RD/GD-99.3 Minister of Public Works and Government Services Canada 2012 Catalogue number CC172-82/2012E-PDF
Public Information and Disclosure RD/GD-99.3 March, 2012 Public Information and Disclosure Regulatory Document RD/GD-99.3 Minister of Public Works and Government Services Canada 2012 Catalogue number CC172-82/2012E-PDF
1. Redistributions of documents, or parts of documents, must retain the SWGIT cover page containing the disclaimer.
 Disclaimer: As a condition to the use of this document and the information contained herein, the SWGIT requests notification by e-mail before or contemporaneously to the introduction of this document,
Disclaimer: As a condition to the use of this document and the information contained herein, the SWGIT requests notification by e-mail before or contemporaneously to the introduction of this document,
