User's Manual. LEICKE Sharon Blood Pressure Monitor with Bluetooth. Product number: LH67402
|
|
|
- Branden Shields
- 5 years ago
- Views:
Transcription
1 User's Manual LEICKE Sharon Blood Pressure Monitor with Bluetooth Product number: LH67402
2 Thank you for purchasing the Blood Pressure Monitor from LEICKE Sharon. We continuously work on the development of our products, our customer support and innovations. Our products have a long service life, are eco-friendly and high performing. Furthermore, every day we do our very best to satisfy you as our customer. That is why you, no matter if private or business customer, are in the focus of our company s efforts. We take your reviews and proposals seriously and evaluate them continuously. That way, we get to know you and your demands on our products and services better and thus allow for positive enhancements. To discover more about LEICKE products or if you have any questions about this product visit our website PACKAGE CONTENTS Before attempting to use this device, please check the packaging and make sure the following items are contained in the package: Number of Name Note Pieces 1 Blood Pressure Monitor (USB interface) Bluetooth USB Cable - 1 User's Manual -
3 Input voltage Input current Measurement mode Measurement range Accuracy TECHNICAL DETAILS USB, 5V 60mA Oscillographic testing mode Rated cuff pressure: 0mmHg-300mmHg(0kPa-40kPa) Measurement pressure: 40mmHg-230mmHg ( 5.3kPa-30.7kPa ) pulse value:(40-199)beat/minute Pressure: 5 C 40 C within ± 3mmHg (0.4kPa) pulse value: ±5% Normal working condition Temperature: 5 C to 40 C Relative humidity: 85%RH Atmospheric pressure: 86kPa to 106kPa Storage & transportation condition Degree of protection Protection against ingress of water Software version Device classification RF Frequency Range Transmitting Distance Weight Dimensions Temperature: -20 C to 60 C Relative humidity: 10%RH to 93%RH Atmospheric pressure: 50kPa to 106kPa Type BF applied part IP22 V01 Battery Powered Mode: Internally Powered ME Equipment AC Adaptor charged Mode: Class II ME Equipment 2402 MHz to 2480 MHz 10 m 108 g Size: 79.8 mm x 72.5 mm x 13.2 mm
4 PRODUCT INFORMATION The device features blood pressure measurement, pulse rate measurement and the result storage. Readings taken by this device are equivalent to those obtained by a trained observer using the cuff and stethoscope auscultation method. This manual contains important safety and care information, and provides step by step instructions for using the product. Please do read this user manual carefully and thoroughly before use. FEATURES: Systolic Blood Pressure Diastolic Blood Pressure Pulse Rate Memory: Up to 60 pieces of records MEASUREMENT PRINCIPLE This product uses the Oscillometric Measuring Method to detect blood pressure. Before every measurement, the unit establishes a zero point equivalent to the atmospheric pressure. Then it starts inflating the cuff. Meanwhile, the unit detects pressure oscillation generated by beat-to-beat pulsatile, which is used to determine the systolic pressure and diastolic pressure as well as pulse rate. The device also compares the longest and the shortest intervals of detected pulse wave with the average value, and then calculates the standard deviation. The monitor will light up a warning symbol when the calculated standard deviation is larger than or equal to 15.
5 SAFETY INFORMATION The signs below might be in the user manual, labelling or other components. They are the requirement of standard and using. Symbol for THE OPERATION GUIDE MUST BE READ Symbol for COMPLIES WITH MDD 93/42/EEC REQUIREMENTS Symbol for MANUFACTURER Symbol for TYPE BF APPLIED PARTS Symbol for ENVIRONMENT PROTECTION - Electrical waste products should not be disposed of with household waste. Please recycle where facilities exist. Check with your local authority or retailer for recycling advice Symbol for SERIAL NUMBER Symbol for DIRECT CURRENT Symbol for Including RF transmitter Caution: These notes must be observed to prevent any damage to the device. Symbol for Authorised Representative in the European Community Symbol for MANUFACTURE DATE INDICATIONS FOR USE The LEICKE Sharon Blood Pressure Monitor is a digital monitor intended for measuring blood pressure and heartbeat rate with wrist circumferences ranging from 13.5 cm to 21.5 cm ( about 5¼ inches to 8½ inches ). It is intended for adult indoor use only.
6 CAUTION: * Intended for adult indoor use only. Pregnant women, neonatal patients, pre-eclamptic patients and patients with severe obesity must not use the device. If in need, please consult a professional doctor. * Intended for non-invasive measuring and monitoring of arterial blood pressure. Not intended for use on extremities other than wrist or for functions other than obtaining a blood pressure measurement. * Please use device under user manual specified environment, otherwise accuracy may be influenced. * Do not confuse self-monitoring with self-diagnosis. This unit allows you to monitor your blood pressure. Do not start or end medical treatment without asking a physician for treatment advice. * If you are taking medication, consult your physician to determine the most appropriate time for your measurement. Never change a prescribed medication without your physician s consent. * If the cuff s pressure exceeds 40 kpa (300 mmhg), the unit will automatically deflate. If the cuff doesn t deflate when its pressure exceeds 40 kpa (300 mmhg), detach cuff from wrist and press the START/STOP button to stop inflation. * Do not use the monitor under a strong electromagnetic field (e.g. medical RF equipment) that radiates interference signals or electrical fast transient/ burst signals. * The maximum temperature of the device is 42.5 C the environmental temperature being 40 C. * The device is not AP/APG equipment. It is not suitable for use in the presence of a flammable anaesthetic mixture with air (or oxygen, nitrous oxide). * Please keep the unit out of reach of infants, children or pets, since inhalation or swallowing of small parts is dangerous or even fatal. * Please use ACCESSORIES and detachable parts specified / authorised by MANUFACTURER. Otherwise, it may cause damage to the unit or endanger the user / patient. * The patient is an intended operator. The patient can measure, transmit data and charge battery under normal circumstances and maintain the device and its accessories according to the user manual. * The blood pressure monitor, its adaptor, and the cuff are suitable for use within the patient's environment. If you are allergic to dacron or plastic, please don t use this device. * The device is not intended for PATIENT transport outside a healthcare facility. * This device cannot be used with HF surgical equipment at the same time. * There is a PTC current limiter in the monitor, whose specification is 8V and 0.5A. When the voltage and current exceed the limiting value, the monitor will stop working. * The adaptor is specified as a part of ME equipment. * The device is not suitable for public use. * The adapter insulates the device from the main supply. Do not position the plug in a position where it is difficult to disconnect from the supply mains. * Sensitive people, including pregnant women pre-eclamptic patients, patients who implanted medical electronic instruments and have atrial fibrillation (AF), premature ventricular beats and peripheral arterial disease (PAD)., should avoid using the unit whenever possible. * This unit is not suitable for continuous monitoring during medical emergencies or operations. * Manufacturer will make available on request circuit diagrams, component parts list etc.
7 LCD DISPLAY SIGNAL SYMBOL DESCRIPTION EXPLANATION Systolic Blood Pressure High blood pressure SYS DIA mmhg Unit Diastolic Blood Pressure Pulse Low Battery IHB Detector Bluetooth Error Memory Time Heartbeat High blood pressure Beat/minute Low battery - please charge Measurement unit of blood pressure (1mmHg=0.133kPa) Irregular Heartbeat Detector Successful Bluetooth Connection Error Recalling the history records Hour: Minute (Month/Day/Year) Heartbeat detection during the measurement
8 Monitor Components SYSTOLIC DIASTOLIC PULSE RATE MEM/UP BUTTON TIME START/STOP BUTTON SET/DOWN BUTTON Component list of pressure measuring system: CUFF (Type BF applied part) 1. PCBA 2. Air Pipe 3. Pump 4. Valve 5. Cuff USB Interface
9 Charging the Device 1. The battery of the monitor is a built-in rechargeable lithium-ion battery, the battery current is 420 mah. 2. Please use the USB cable to charge the battery, just like the following pictures: Method 1 Method 2 Charging the power under following circumstances: displays on the LCD The LCD display dims When switching on the monitor, the LCD doesn t light up. CAUTION: 1. The battery of the monitor is built-in rechargeable lithium-ion battery, please do not disassemble yourself or by unauthorized maintenance personnel. 2. When used normally, it can be charged about 300 times, if the battery cannot be charged normally or the blood pressure monitor cannot be used normally, please contact authorized maintenance personnel. If you measure three times per day, and the battery was fully charged, it can be used for about 20 days. 3. Store and use the blood pressure monitor in a cool, dry and ventilated environment. Avoid fire and heat sources, or the battery may explode. 4. Only use the authorized USB cable (5V 1A) to charge power. 5. During the charging process, the blood pressure monitor displays: When charging is finished, please pull the plug in time.
10 CAUTION: 6. When charging, do not touch charging connector and patient simultaneously. 7. Do not attempt to replace your blood pressure monitor s battery. It is built-in and not changeable. 8. Only charge the battery in accordance with the user instructions supplied with the blood pressure monitor. 9. Avoid charging your blood pressure monitor in extremely high or low temperatures. 10. Do not use your blood pressure monitor while you are charging it. 11. Do not attempt to disassemble the blood pressure monitor or force open the built-in battery. 12. Do not clean the blood pressure monitor when it is being charged. Always unplug the charger first before cleaning the blood pressure monitor. 13. Do not dispose of your blood pressure monitor in a fire. The battery could explode causing injury or death. 14. Batteries (battery pack or batteries installed) shall not be exposed to excessive heat such as sunshine, fire or the like. Activate Your Blood Pressure Monitor Getting the Blood Pressure Monitor, the first thing to do after charging is to activate it. Please press and hold the SET button to activate it and enter setting mode.
11 Setting Date and Time Please proceed to time setting before your initial use so as to ensure each piece of record is labelled with a time stamp. (Year Range: ; Time Format: 12 Hours) 1. When the monitor is OFF, press and hold SET button for 3 seconds to enter Time Setting Mode. 2. As pictured in the right, the blinking number represents the [HOUR]. Press MEM button to change the number. Each press will increase the number by one in a cycling manner. 3. Press SET button again to confirm [HOUR]. Then the number representing [MINUTE] blinks.
12 4. Repeat steps 2 and 3 to confirm [MINUTE]. Setting Date and Time 5. Repeat steps 2 and 3 to confirm [MONTH], [DAY] and [YEAR]. 6. After confirming [YEAR], the LCD will display done and then shut off.
13 Positioning the Cuff 1. Remove all accessories (watch, bracelet, etc.) from your left wrist. If your physician has diagnosed you with poor circulation in your left wrist, use your right wrist. 2. Roll or push up your sleeve to expose the skin. 3. Apply the cuff to your left wrist with your palm facing up. 4. Position the edge of the cuff about 1-2 cm below the wrist. 5. Fasten the wrist cuff around your wrist, leaving no extra room between the cuff and your skin. If the cuff is too loose, the measurement will not be accurate. 6. Patients with Hypertension: The middle of the cuff should be at the level of the right atrium of the heart; Before starting measurement, please sit comfortably with legs uncrossed, feet flat on the floor, back and arm supported. Rest for 5 minutes before measuring. Wait at least 3 minutes between measurements. This allows your blood circulation to recover. For a useful comparison, try to measure under similar conditions. For example, take daily measurements at approximately the same time, on the same wrist, or as directed by a physician.
14 PAIRING THE BLOOD PRESSURE MONITOR WITH YOUR MOBILE DEVICE Download and install the MedM Health APP to your mobile device which supports Bluetooth 4.0 technology from the APP Store or Google Play. 1. Turn on Bluetooth and the app. Make sure both are ON when pairing is proceeding. 2. When the monitor is OFF, press and hold the START/STOP button to start pairing. These symbols will be shown on the LCD alternatively, indicating pairing is proceeding. If SUCCEED, this symbol will be shown on the LCD. If FAIL, than this symbol (Bluetooth ERROR) will be shown on the LCD. 3. The monitor will shut off after pairing process is complete.
15 Start Measurement 1. After correctly positioning the cuff, press START/STOP button to turn on the monitor, and it will complete the measurement process. Adjust to zero. Inflating and measuring. Display and save the measuring result.
16 2. This device will proceed to data transmission after measurement. The Bluetooth symbol blinking on the LCD indicates data transmission. 3. If the data is successfully transmitted, the LCD will display done. If data transmission fails, the LCD will display BLUETOOTH ERROR instead. 4. Press START/STOP button to turn off the monitor. Otherwise if there is no operation, it will switch off within 1 minute.
17 Recall the Records 1. Press MEM button to access the memory when the monitor is off. The monitor will display the calculated value of the latest readings first. 2. Press MEM/UP button or SET/DOWN button to rotate the history records. MEM/UP to go forward; SET/DOWN to go backward. CAUTION: The most recent record (1) is shown first. Each new measurement is assigned to the first (1) record. All other records are pushed back one digit (e.g., 2 becomes 3, and so on), and the last record (60) is dropped from the list.
18 Delete the Records When you did not obtain the accurate measurement, you can clear all the measuring results by following the steps below. 1. Under Memory Recalling Mode, press and hold both the MEM button and the SET button for 3 seconds. 2. The LCD will display del done, indicating that memory clearing is complete. And then it will shut down. CAUTION: Under Memory Recalling Mode, if you wish to give up clearing, press START/STOP to turn off the monitor. 3. When there is no memory in the monitor, if you press the MEM button to look up history, the LCD will display as pictured to the right.
19 Tips for Measurement Measurements may be inaccurate if taken in the following circumstances. Within 1 hour after dinner or drinking Immediate measurement after tea, coffee, smoking Within 20 minutes after taking a bath When talking or moving your fingers In a very cold environment When you need to pee urgently
20 Maintenance To obtain the best performance, please follow the instructions below. Put in a dry place and avoid sunshine. Avoid immersing it in water. Clean it with a dry cloth in case. Avoid shaking and collisions. Avoid dusty environment and unstable temperature surrounding. Use a slightly damp cloth to remove dirt. Avoid washing the cuff. CAUTION: 1. Please make sure the unit functions safely and it is in proper working conditions before use. Don t service or maintain while the device is in use. 2. If you have any problems with this device, such as setting up, maintaining or using, please contact the customer service. Don t open or repair the device by yourself. 3. Please report to LEICKE if any unexpected operation or events occur. 4. Cleaning: Dust environment may affect the performance of the unit. Please use a soft cloth to remove dirt from the device and cuff before and after use. 5. Calibration: The manufacturer does not require such preventive inspections or calibration by other persons and will make available on request circuit diagrams, component part list, etc. 6. Disposal: Degraded sensors may result in inaccurate measurement while loosened electrodes may cause the monitor s failure to power on. Please dispose of ACCESSORIES, detachable parts, and ME EQUIPMENT according to local guidelines.
21 About Blood Pressure What are systolic pressure and diastolic pressure? When ventricles contract and pump blood out of the heart, the blood pressure reaches its maximum value in the cycle, which is called systolic pressure. When the ventricles relax, the blood pressure reaches its minimum value in the cycle, which is called diastolic pressure. What is the standard blood pressure classification? The blood pressure classification published by World Health Organization (WHO) and International Society of Hypertension (ISH) in 1999 is as follows: CAUTION: Only a physician can tell your normal BP range. Please contact a physician if your measuring result falls out of the range. Please note that only a physician can tell whether your blood pressure value has reached a dangerous point.
22 About Blood Pressure Irregular Heartbeat Detector: An irregular heartbeat is detected when the heartbeat rhythm varies while the unit is measuring the systolic and diastolic blood pressure. During each measurement, this equipment records the heartbeat intervals and works out the standard deviation. If the calculated value is larger than or equal to 15, the irregular heartbeat symbol appears when the measurement results are displayed. CAUTION: The appearance of the IHB icon indicates that a pulse irregularity consistent with an irregular heartbeat was detected during measurement. Usually this is NOT a cause for concern. However, if the symbol appears often, we recommend you seek medical advice. Please note that the device does not replace a cardiac examination, but serves to detect pulse irregularities at an early stage. Why does my blood pressure fluctuate throughout the day? Individual blood pressure varies multiple times every day. It is also affected by the way you tie your cuff and your measurement position, so please take the measurement under the same conditions. If the person takes medicine, the pressure will vary more. Wait at least 3 minutes for another measurement.
23 About Blood Pressure Why do I get a different blood pressure at home compared to the hospital? The blood pressure is different even throughout the day due to weather, emotion, exercise etc. Also, there is the white coat effect, which means blood pressure usually increases in clinical settings. What do you need to pay attention to when you measure your blood pressure at home: If the cuff is tied properly. If the cuff is too tight or too loose. If the cuff is tied on the wrist. If you feel anxious. Taking 2-3 deep breaths before beginning will be better for measuring. Advice: Relax yourself for 4-5 minutes until you calm down. Is the result the same if measuring on the right wrist? It is ok for both wrists, but there will be some different results for different people. We suggest you measure the same wrist every time.
24 Problem Description No Power Symptom Display is dim or will not light up. FAQ Possible Cause/ Possible Solution Power is exhausted. Please charge. Low Batteries Error Message Shows on the display Shows Battery is low. Please charge. Data communication has failed Make sure that phone s Bluetooth is switched on or within the distance range 1 shows Inflation is slow or the cuff is not secure Refasten the cuff and then measure again. 2 shows 3 shows 10 or 11 shows 20 shows The cuff is very tight. Refasten the cuff and then measure again. Refasten the cuff and then measure again. The pressure of the cuff is too high. Relax for a moment and then measure again. The monitor detected motion, talking or the pulse is too poor while measuring. The device does not detect the pulse signal. Loosen the clothing on the
25 Problem Description Error Message Error message FAQ Possible Cause/ Symptom Possible Solution Relax for a moment and 21 shows then measure again. The treatment of the measurement failed. EExx,shows on Retake the measurement. the display. If the problem persists, A calibration error contact the retailer or our occurred customer service department for further assistance. Refer to the warranty for contact information and return instructions. Complied European Standards List Risk management Labelling User manual General Requirements for Safety Electromagnetic compatibility Performance requirements Clinical investigation Usability Software life-cycle processes ISO/EN 14971:2012 Medical devices Application of risk management to medical devices EN 980 Symbols for use in the labelling of medical devices EN 1041: 2008 Medical equipment manufacturers to provide information EN : 2006 Medical electrical equipment - Part 1: General requirements for basic safety and essential performance IEC/EN : 2010 Medical electrical equipment -- Part 1-11: General requirements for basic safety and essential performance - Collateral standard: Requirements for medical electrical equipment and medical electrical systems used in the home healthcare environment IEC/EN :2007 Medical electrical equipment -Part 1-2: General requirements for basic safety and essential performance - Collateral standard: Electromagnetic compatibility- Requirements and tests EN :1995+A2:2009 Non-invasive blood pressure Part 1: General requirements EN :1997+A2:2009 Non-invasive blood pressure Part 3: Supplementary requirements for electromechanical blood pressure measuring system EN : 2004 Automatic Blood Pressure Monitor overall system Interventional accuracy of the testing process IEC/EN : 2010 Medical electrical equipment -- Part 1-6: General requirements for basic safety and essential performance - Collateral Standard: Usability IEC/EN 62366: 2007 Medical devices - Application of usability engineering to medical devices IEC/EN 62304:2006+AC: 2008 Medical device software - Software life cycle processes
26 EMC Guidance Table 1 Guidance and MANUFACTURER s declaration ELECTROMAGNETIC EMISSIONS- for all ME EQUIPMENT and ME SYSTEMS Guidance and manufacturer s declaration electromagnetic emissions The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. Emissions test Compliance Electromagnetic environment - guidance The device must emit electromagnetic RF emissions energy in order to perform its intended Group 2 CISPR 11 function. Nearby electronic equipment may be affected. RF emissions CISPR 11 Class B Harmonic emissions IEC Voltage fluctuations/ flicker emissions IEC Not applicable Not applicable
27 EMC Guidance Table 2 Guidance and MANUFACTURER s declaration ELECTROMAGNETIC IMMUNITY for all ME EQUIPMENT and ME SYSTEMS Guidance and manufacturer s declaration electromagnetic immunity The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment Electromagnetic IMMUNITY test IEC test level Compliance level environment - Electrostatic discharge (ESD) IEC Electrical fast transient/burst IEC Surge IEC Voltage dips, short interruptions and voltage variations on power supply input lines IEC Power frequency (50/60Hz) magnetic field IEC ±6 kv contact ±8 kv air ±2 kv for power supply lines ±1 kv for input/output lines ±1 kv line(s) to line(s) ±2 kv line(s) to earth <5% UT (>95% dip in UT) for 0.5 cycle 40% UT (60% dip in UT ) for 5 cycles 70% UT (30% dip in UT ) for 25 cycles <5% UT (>95% dip in UT ) for 5 s 3A/m ±6 kv contact ±8 kv air ±2 kv for power supply lines ±1 kv line(s) to line(s) <5% UT (>95% dip in UT) for 0.5 cycle 40% UT (60% dip in UT ) for 5 cycles 70% UT (30% dip in UT ) for 25 cycles <5% UT (>95% dip in UT ) for 5 s 3A/m guidance Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30%. Mains power quality should be that of a typical commercial or hospital environment. Mains power quality should be that of a typical commercial or hospital environment. Mains power quality should be that of a typical commercial or hospital environment. If the user of the device requires continued operation during power mains interruptions, it is recommended that the device be powered from an uninterruptible power supply or a battery. Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. NOTE: UT is the a.c. mains voltage prior to application of the test level.
28 EMC Guidance Table 4 Guidance and MANUFACTURER s declaration ELECTROMAGNETIC IMMUNITY for ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING Guidance and manufacturer s declaration electromagnetic immunity The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. IMMUNITY test Conducted RF IEC Radiated RF IEC IEC test level 3 Vrms 150 khz to 80 MHz 3 V/m 80 MHz to 2.5 GHz Compliance level 3 Vrms 3 V/m Electromagnetic environment - guidance Portable and mobile RF communications equipment should be used no closer to any part of the device, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter. Recommended separation distance d = P d = P 80 MHz to 800 MHz d = P 800 MHz to 2,5 GHz where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in metres (m). Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, a should be less than the compliance level in each frequency range b. Interference may occur in the vicinity of equipment marked with the following symbol: NOTE 1: At 80 MHz and 800 MHz, the higher frequency range applies. NOTE 2: These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. a Field strengths from fixed transmitters, such as base stations for radio (cellular / cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the device is used exceeds the applicable RF compliance level above, the device should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the device. b Over the frequency range 150 khz to 80 MHz, field strengths should be less than 3V/m.
29 EMC Guidance Table 6 Recommended separation distances between portable and mobile RF communications equipment and the ME EQUIPMENT or ME SYSTEM - for ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING Recommended separation distances between portable and mobile RF communications equipment and the device. The device is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the device can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the device as recommended below, according to the maximum output power of the communications equipment. Rated maximum Separation distance according to frequency of transmitter (m) output power of transmitter (W) 150 khz to 80 MHz 80 MHz to 800 MHz 800 MHz to 2.5 GHz d = P d = P d = P For transmitters rated at a maximum output power not listed above, the recommended separation distance d in metres (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer. NOTE 1: At 80MHz and 800MHz, the separation distance for the higher frequency range applies. NOTE 2: These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people.
30 FCC Interference Statement This equipment has been tested and found to comply with the limits for a Class B digital device, pursuant to Part 15 of the FCC Rules. These limits are designed to provide reasonable protection against harmful interference in a residential installation. This equipment generates, uses and can radiate radio frequency energy and, if not installed and used in accordance with the instructions, may cause harmful interference to radio communications. However, there is no guarantee that interference will not occur in a particular installation. If this equipment does cause harmful interference to radio or television reception, which can be determined by turning the equipment off and on, the user is encouraged to try to correct the interference by one of the following measures: - Reorient or relocate the receiving antenna. - Increase the separation between the equipment and receiver. - Connect the equipment into an outlet on a circuit different from that to which the receiver is connected. - Consult the dealer or an experienced radio/tv technician for help. This device complies with Part 15 of the FCC Rules. Operation is subject to the following conditions: This device may not cause harmful interference, and (2) this device must accept any interference received, including interference that may cause undesired operation. FCC-Caution: FCC Caution: Any changes or modifications not expressly approved by the party responsible for compliance could void the user's authority to operate this equipment. IMPORTANT NOTE: Radiation Exposure Statement: This equipment complies with FCC radiation exposure limits set forth for an uncontrolled environment. End users should follow the specific instructions in the user's manual to comply with the FCC rules. This transmitter must not be co-located or operating in conjunction with any other antenna or transmitter. DISPOSAL This device contains materials that must not be disposed of as household waste. Please check local laws concerning the applicable disposal regulations. Protect the environment by participating in recycling programs.
31 CONTACT If you have any questions, please feel free to contact us. PRODUCTION & WHOLESALE LEICKE GmbH Dohnanyistr Leipzig Germany Web: Mail:
INTRODUCTION. 4 SAFETY INSTRUCTIONS. 5 ABOUT THIS DEVICE. 6 FIRST OPERATION. 7 THE DISPLAY. 8 ATTACHING THE WRIST SLEEVE. 9 CORRECT MEASUREMENT.
 Table of contents INTRODUCTION.............................. 4 SAFETY INSTRUCTIONS........................ 5 ABOUT THIS DEVICE.......................... 6 FIRST OPERATION............................ 7
Table of contents INTRODUCTION.............................. 4 SAFETY INSTRUCTIONS........................ 5 ABOUT THIS DEVICE.......................... 6 FIRST OPERATION............................ 7
Powered Traction Unit OPERATION MANUAL
 Powered Traction Unit OPERATION MANUAL CONTENTS Symbols Safety precautions Symbol for CAUTION Symbol for CONSULT INSTRUCTIONS FOR USE Symbol for SERIAL NUMBER Symbol for CATALOGUE NUMBER Symbol for AUTHORISED
Powered Traction Unit OPERATION MANUAL CONTENTS Symbols Safety precautions Symbol for CAUTION Symbol for CONSULT INSTRUCTIONS FOR USE Symbol for SERIAL NUMBER Symbol for CATALOGUE NUMBER Symbol for AUTHORISED
General Safety/EMC and Electrical Information for i-limb ultra and i-limb digits
 1. General Safety 1.1 The i-limb ultra and i-limb digits devices are electrical devices, which under certain circumstances could present an electrical shock hazard to the user. Please read the accompanying
1. General Safety 1.1 The i-limb ultra and i-limb digits devices are electrical devices, which under certain circumstances could present an electrical shock hazard to the user. Please read the accompanying
Guidance and Declaration - Electromagnetic Compatibility (EMC) for the Delfi PTS ii Portable Tourniquet System
 Guidance and Declaration - Electromagnetic Compatibility (EMC) for the Delfi TS ii ortable Tourniquet System Guidance and manufacturer s declaration electromagnetic emissions The TS ii ortable Tourniquet
Guidance and Declaration - Electromagnetic Compatibility (EMC) for the Delfi TS ii ortable Tourniquet System Guidance and manufacturer s declaration electromagnetic emissions The TS ii ortable Tourniquet
BIODEX MULTI- JOINT SYSTEM
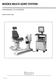 BIODEX MULTI- JOINT SYSTEM CONFORMANCE TO STANDARDS 850-000, 840-000, 852-000 FN: 18-139 5/18 Contact information Manufactured by: Biodex Medical Systems, Inc. 20 Ramsey Road, Shirley, New York, 11967-4704
BIODEX MULTI- JOINT SYSTEM CONFORMANCE TO STANDARDS 850-000, 840-000, 852-000 FN: 18-139 5/18 Contact information Manufactured by: Biodex Medical Systems, Inc. 20 Ramsey Road, Shirley, New York, 11967-4704
D C 01/2019 3
 D-0117968-C 01/2019 3 4 D-0117968-C 01/2019 Screw Driver Screw Driver Unplug both the Red & Blue connectors. (see above) Place a small flat head screw driver on the small orange tabs and push down while
D-0117968-C 01/2019 3 4 D-0117968-C 01/2019 Screw Driver Screw Driver Unplug both the Red & Blue connectors. (see above) Place a small flat head screw driver on the small orange tabs and push down while
Electromagnetic compatibility Guidance and manufacturer s declaration DIN EN :2007 (IEC :2007)
 Compressor set Equipment Under Test (EUT) Type 028 Type 047 Type 052 Type 085 Electromagnetic compatibility Guidance and manufacturer s declaration DIN EN 60601-1-2:2007 (IEC 60601-1-2:2007) 2017 PARI
Compressor set Equipment Under Test (EUT) Type 028 Type 047 Type 052 Type 085 Electromagnetic compatibility Guidance and manufacturer s declaration DIN EN 60601-1-2:2007 (IEC 60601-1-2:2007) 2017 PARI
English
 English Specifications Type Power Source Vibration Frequency Maximum Output Power Consumption Water Pressure Lighting NE134 AC120V 50/60Hz AC230V 50/60Hz 28~32kHz 8W Max. 42VA 0.1~0.5MPa (1~5kgf/cm
English Specifications Type Power Source Vibration Frequency Maximum Output Power Consumption Water Pressure Lighting NE134 AC120V 50/60Hz AC230V 50/60Hz 28~32kHz 8W Max. 42VA 0.1~0.5MPa (1~5kgf/cm
WRIST BLOOD PRESSURE MONITOR
 WRIST BLOOD PRESSURE MONITOR Instruction Manual MODEL: ABP801 www.accumed.com TABLE OF CONTENTS INTRODUCTION... 1 NOTES ON SAFETY... 1 ABOUT BLOOD PRESSURE... 3 PRECAUTIONS BEFORE US... 4 FEATURES OF THE
WRIST BLOOD PRESSURE MONITOR Instruction Manual MODEL: ABP801 www.accumed.com TABLE OF CONTENTS INTRODUCTION... 1 NOTES ON SAFETY... 1 ABOUT BLOOD PRESSURE... 3 PRECAUTIONS BEFORE US... 4 FEATURES OF THE
TH008F Multi-function Infrared Forehead Thermometer
 TH008F Multi-function Infrared Forehead Thermometer Specifications Functions Temperature measurement range: Forehead mode: 34~42.2 C (93.2~108 F), Surface mode: -22~80 C (-7.6~176 F) Operating temperature
TH008F Multi-function Infrared Forehead Thermometer Specifications Functions Temperature measurement range: Forehead mode: 34~42.2 C (93.2~108 F), Surface mode: -22~80 C (-7.6~176 F) Operating temperature
HeRO duet
 HeRO duet CUSTOMER SERVICE TABLE OF CONTENTS TABLE OF CONTENTS OVERVIEW OVERVIEW OVERVIEW OVERVIEW USING HeRO duet USING HeRO duet USING HeRO duet Current HeRO Score HeRO USING HeRO duet USING HeRO duet
HeRO duet CUSTOMER SERVICE TABLE OF CONTENTS TABLE OF CONTENTS OVERVIEW OVERVIEW OVERVIEW OVERVIEW USING HeRO duet USING HeRO duet USING HeRO duet Current HeRO Score HeRO USING HeRO duet USING HeRO duet
User Instruction Computer Assisted Local Analgesia. 337 Marion, Le Gardeur QC, Canada, J5Z 4W8
 User Instruction Computer Assisted Local Analgesia 1-800-667-9622 337 Marion, Le Gardeur QC, Canada, J5Z 4W8 USER INSTRUCTION Congratulations on your new CALAJECT! Please read these instructions thoroughly
User Instruction Computer Assisted Local Analgesia 1-800-667-9622 337 Marion, Le Gardeur QC, Canada, J5Z 4W8 USER INSTRUCTION Congratulations on your new CALAJECT! Please read these instructions thoroughly
Rolyan Splint Pan OPERATION MANUAL. Item # Small Item # Large
 Rolyan Splint Pan OPERATION MANUAL Item #081544816 - Small Item #081544808 Large PLEASE READ THIS ENTIRE MANUAL BEFORE OPERATING YOUR NEW SPLINT PAN. Failure to follow these instructions could result in
Rolyan Splint Pan OPERATION MANUAL Item #081544816 - Small Item #081544808 Large PLEASE READ THIS ENTIRE MANUAL BEFORE OPERATING YOUR NEW SPLINT PAN. Failure to follow these instructions could result in
Nursing Beds with Dewert drive system
 Nursing Beds with Dewert drive system GB Casa Med Classic 4 / Classic (FS) Casa Med Ultra / Ultra (FS) Casa Med Classic Low Casa Med Classic (FS) 4 / Classic / Casa FS Med / Casa Classic Med Low Ultra
Nursing Beds with Dewert drive system GB Casa Med Classic 4 / Classic (FS) Casa Med Ultra / Ultra (FS) Casa Med Classic Low Casa Med Classic (FS) 4 / Classic / Casa FS Med / Casa Classic Med Low Ultra
OtoRead - Technical Specifications Page 0. Technical Specifications. OtoRead D A 2017/06
 OtoRead - Technical Specifications Page 0 Technical Specifications OtoRead D-0116698-A 2017/06 OtoRead - Technical Specifications Page 1 OtoRead TM Configuration Overview The OtoReadTM is available in
OtoRead - Technical Specifications Page 0 Technical Specifications OtoRead D-0116698-A 2017/06 OtoRead - Technical Specifications Page 1 OtoRead TM Configuration Overview The OtoReadTM is available in
Biological Safety. Electromagnetic Compatibility (EMC) Observe the following precautions related to biological safety.
 Biological Safety Observe the following precautions related to biological safety. WARNING: Non-medical (commercial) grade peripheral monitors have not been verified or validated by SonoSite as being suitable
Biological Safety Observe the following precautions related to biological safety. WARNING: Non-medical (commercial) grade peripheral monitors have not been verified or validated by SonoSite as being suitable
PHYSIOFLOW Q-LINK TM
 PHYSIOFLOW Q-LINK TM Service Manual Thursday, 20 October 2016 First placing on the market : 18 January 2012 User Manual PhysioFlow Q-Link 1/17 Table of contents 1. General Information... 3 About this manual...
PHYSIOFLOW Q-LINK TM Service Manual Thursday, 20 October 2016 First placing on the market : 18 January 2012 User Manual PhysioFlow Q-Link 1/17 Table of contents 1. General Information... 3 About this manual...
The following symbol indicates that the device is MR-unsafe:
 The following symbol indicates that the device is MR-unsafe: MR Unsafe Do not use this equipment In the MRI scan room Patient must follow the doctor s instructions and should not perform a self-assessment
The following symbol indicates that the device is MR-unsafe: MR Unsafe Do not use this equipment In the MRI scan room Patient must follow the doctor s instructions and should not perform a self-assessment
AUTOMATIC BLOOD PRESSURE MONITOR (Arm Cuff Type) INSTRUCTION MANUAL
 AUTOMATIC BLOOD PRESSURE MONITOR (Arm Cuff Type) INSTRUCTION MANUAL Model: SPBP-04 English Instruction Guide Thank you for choosing Advocate as your monitor of choice. The first, most important part of
AUTOMATIC BLOOD PRESSURE MONITOR (Arm Cuff Type) INSTRUCTION MANUAL Model: SPBP-04 English Instruction Guide Thank you for choosing Advocate as your monitor of choice. The first, most important part of
Technical Specifications Micromedical VisualEyes 505 by Interacoustics
 VisualEyes 505 - Technical Specifications Page 0 Technical Specifications Micromedical VisualEyes 505 by Interacoustics D-0115523-B 2018/02 VisualEyes 505 - Technical Specifications Page 1 Included and
VisualEyes 505 - Technical Specifications Page 0 Technical Specifications Micromedical VisualEyes 505 by Interacoustics D-0115523-B 2018/02 VisualEyes 505 - Technical Specifications Page 1 Included and
USER MANUAL MHS-2500I. Please take time to read these instructions before starting to use the scale. Version /17
 USER MANUAL MHS-2500I Please take time to read these instructions before starting to use the scale Version 1.0 05/17 Contents Introduction 3 Product Specification 3 Safety Instructions 4 Explanation of
USER MANUAL MHS-2500I Please take time to read these instructions before starting to use the scale Version 1.0 05/17 Contents Introduction 3 Product Specification 3 Safety Instructions 4 Explanation of
Operation Guide Model: 405-HT V3.0
 MODEL 405-HT Operation Guide Model: 405-HT V3.0 Fully Automatic Wrist Cuff Blood Pressure Monitor (ELECTRONIC SPHYGMOMANOMETER) OPERATION GUIDE INDEX IMPORTANT INFORMATION... 2 CONTENTS AND DISPLAY INDICATORS...
MODEL 405-HT Operation Guide Model: 405-HT V3.0 Fully Automatic Wrist Cuff Blood Pressure Monitor (ELECTRONIC SPHYGMOMANOMETER) OPERATION GUIDE INDEX IMPORTANT INFORMATION... 2 CONTENTS AND DISPLAY INDICATORS...
GTIN:
 GTIN: 00894912002920 TABLE OF CONTENTS Introduction...1 Features...1 Indications for Use...1 Contraindications...1 Warnings & Precautions...2 Controls...3 Operation...4 Recharging the Battery...5 Replacing
GTIN: 00894912002920 TABLE OF CONTENTS Introduction...1 Features...1 Indications for Use...1 Contraindications...1 Warnings & Precautions...2 Controls...3 Operation...4 Recharging the Battery...5 Replacing
(c) Medisave UK. Notice. Edition 1. i-pad Operator s Manual
 Edition 1 Notice i-pad Operator s Manual CU Medical Systems, Inc. reserves the right to make changes on the device specifications contained in this manual at any time without prior notice or obligation
Edition 1 Notice i-pad Operator s Manual CU Medical Systems, Inc. reserves the right to make changes on the device specifications contained in this manual at any time without prior notice or obligation
Central Blood Pressure Meter Model cbp301. Operating Manual
 Central Blood Pressure Meter Model cbp301 Operating Manual Document cbp301-009 Issue 4 September 2011 Contents Introduction... 3 Package Contents... 5 Warnings and Cautions... 6 Contraindications... 7
Central Blood Pressure Meter Model cbp301 Operating Manual Document cbp301-009 Issue 4 September 2011 Contents Introduction... 3 Package Contents... 5 Warnings and Cautions... 6 Contraindications... 7
Operating Manual Infrared thermometer
 Operating Manual Infrared thermometer Model:IT-121 Professional Fast Accurate 1 CONTENTS 1 I n t r o d u c t i o n................ 3 1.1Product intended use 3 2 Basic principle 3 3 Pr o d u c t f e a t
Operating Manual Infrared thermometer Model:IT-121 Professional Fast Accurate 1 CONTENTS 1 I n t r o d u c t i o n................ 3 1.1Product intended use 3 2 Basic principle 3 3 Pr o d u c t f e a t
DENTAL X-RAY OPERATOR'S INSTRUCTIONS. (for USA) Wall Mount Type...WK
 M 505 DENTAL X-RAY OPERATOR'S INSTRUCTIONS (for USA) Wall Mount Type...WK WARNING This X-ray equipment may be dangerous to patient and operator unless safe exposure factors, operating instructions and
M 505 DENTAL X-RAY OPERATOR'S INSTRUCTIONS (for USA) Wall Mount Type...WK WARNING This X-ray equipment may be dangerous to patient and operator unless safe exposure factors, operating instructions and
USER MANUAL M-200. Please take time to read these instructions before starting to use the scale. Version /06
 USER MANUAL M-200 Please take time to read these instructions before starting to use the scale Version 1.0 07/06 Contents Introduction 3 Product Specification 3 Safety Instructions 4 Explanation of Graphic
USER MANUAL M-200 Please take time to read these instructions before starting to use the scale Version 1.0 07/06 Contents Introduction 3 Product Specification 3 Safety Instructions 4 Explanation of Graphic
VN415/VO425 Specifications
 VN415/VO425 Specifications Item No. 8105231-1 01/2014 Contents VN415 Specifications... 1 1.1 Technical Standards:...1 1.2 System Requirements:...1 1.3 Included and Optional Parts...2 VO425 Specifications...
VN415/VO425 Specifications Item No. 8105231-1 01/2014 Contents VN415 Specifications... 1 1.1 Technical Standards:...1 1.2 System Requirements:...1 1.3 Included and Optional Parts...2 VO425 Specifications...
Technical Data. Electrocardiograph ECG-1250K TD.ECG1250_L. This technical data may be revised or replaced by Nihon Kohden at any time without notice.
 Technical Data Electrocardiograph ECG-1250K This technical data may be revised or replaced by Nihon Kohden at any time without notice. TD.ECG1250_L Specifications ECG input Input impedance: 20 MΩ Electrode
Technical Data Electrocardiograph ECG-1250K This technical data may be revised or replaced by Nihon Kohden at any time without notice. TD.ECG1250_L Specifications ECG input Input impedance: 20 MΩ Electrode
Not for print. Microphone Test Device with SONNET MTD Adapter. User Manual. Cochlear Implants NOT FOR PRINT
 Cochlear Implants Microphone Test Device with SONNET MTD Adapter User Manual AW32690_1.0 (English) Table of contents 1. Table of contents 2. INTRODUCTION 3 Product description 3 3. INTENDED USE INDICATIONS
Cochlear Implants Microphone Test Device with SONNET MTD Adapter User Manual AW32690_1.0 (English) Table of contents 1. Table of contents 2. INTRODUCTION 3 Product description 3 3. INTENDED USE INDICATIONS
EVOLV TM Upper Arm Blood Pressure Monitor
 INSTRUCTION MANUAL EVOLV TM Upper Arm Blood Pressure Monitor Model BP7000 ENGLISH TABLE OF CONTENTS Before using the monitor INTRODUCTION...4 INTENDED USE...5 IMPORTANT SAFETY INFORMATION...6 Operating
INSTRUCTION MANUAL EVOLV TM Upper Arm Blood Pressure Monitor Model BP7000 ENGLISH TABLE OF CONTENTS Before using the monitor INTRODUCTION...4 INTENDED USE...5 IMPORTANT SAFETY INFORMATION...6 Operating
MedRx Avant Polar HIT AH-I-MPHITS-5 Effective 11/07/11
 INSTALLATION MANUAL 2 Contents Getting To Know Your AVANT POLAR HIT TM... 4 Setting up the System... 6 Software Installation... 7 Driver Installation Windows 7... 10 Driver Installation Windows XP... 13
INSTALLATION MANUAL 2 Contents Getting To Know Your AVANT POLAR HIT TM... 4 Setting up the System... 6 Software Installation... 7 Driver Installation Windows 7... 10 Driver Installation Windows XP... 13
INSTRUCTIONS FOR YOUR NEW PLASMAFLOW. Vascular Therapy System (Compressible Limb Sleeve Device)
 INSTRUCTIONS FOR YOUR NEW PLASMAFLOW Vascular Therapy System (Compressible Limb Sleeve Device) Customer Service Toll Free: 888-508-0712 Email: CustomerService@manamed.net Web: www.manamed.net 1511 W. Alton
INSTRUCTIONS FOR YOUR NEW PLASMAFLOW Vascular Therapy System (Compressible Limb Sleeve Device) Customer Service Toll Free: 888-508-0712 Email: CustomerService@manamed.net Web: www.manamed.net 1511 W. Alton
Welch Allyn Home Scale (T- RPM-SCALE100)
 Welch Allyn Home Scale (T- RPM-SCALE100) Directions for use 901077 Weight scale, Software Version 1.0 2017 Welch Allyn. All rights are reserved. To support the intended use of the product described in
Welch Allyn Home Scale (T- RPM-SCALE100) Directions for use 901077 Weight scale, Software Version 1.0 2017 Welch Allyn. All rights are reserved. To support the intended use of the product described in
DIGITAL BLUETOOTH THEROMETER PBT
 DIGITAL BLUETOOTH THEROMETER PBT Index 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. Introduction Important Information Before Use Product Identification Description of LCD Display Battery Installation
DIGITAL BLUETOOTH THEROMETER PBT Index 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. Introduction Important Information Before Use Product Identification Description of LCD Display Battery Installation
Connevans.info. DeafEquipment.co.uk. This product may be purchased from Connevans Limited secure online store at
 Connevans.info Solutions to improve the quality of life Offering you choice Helping you choose This product may be purchased from Connevans Limited secure online store at www.deafequipment.co.uk DeafEquipment.co.uk
Connevans.info Solutions to improve the quality of life Offering you choice Helping you choose This product may be purchased from Connevans Limited secure online store at www.deafequipment.co.uk DeafEquipment.co.uk
SAVI SCOUT Surgical Guidance System. Console Operation Manual
 SAVI SCOUT Surgical Guidance System Console Operation Manual 2 Copyrights and Trademarks 2016 Cianna Medical, Inc. All rights reserved. Patents pending. Cianna Medical and SAVI are registered trademarks
SAVI SCOUT Surgical Guidance System Console Operation Manual 2 Copyrights and Trademarks 2016 Cianna Medical, Inc. All rights reserved. Patents pending. Cianna Medical and SAVI are registered trademarks
Symbol for 'the device complies with European Medical Device Directive 93/42/EEC requirements' refers to the notified body.
 DL8765 EN-UK Specifications are subject to change without notice 2017 Koninklijke Philips N.V. Find the latest user manual on www.philips.com/support All rights reserved. Philips Consumer Lifestyle BV
DL8765 EN-UK Specifications are subject to change without notice 2017 Koninklijke Philips N.V. Find the latest user manual on www.philips.com/support All rights reserved. Philips Consumer Lifestyle BV
Congratulations on your purchase and welcome to Philips! To fully benefit from the support that Philips offers, register your product at
 DL8765 Specifications are subject to change without notice. 2015 Koninklijke Philips N.V. All rights reserved Philips Consumer Lifestyle BV Tussendiepen 4, 9206AD Drachten, Netherlands Fax +31 (0)512594316
DL8765 Specifications are subject to change without notice. 2015 Koninklijke Philips N.V. All rights reserved Philips Consumer Lifestyle BV Tussendiepen 4, 9206AD Drachten, Netherlands Fax +31 (0)512594316
#
 INSTALLATION MANUAL 2 Contents Getting To Know Your AVANT POLAR HIT TM... 4 Setting up the System... 6 Software Installation... 7 Driver Installation Windows 7... 10 Driver Installation Windows XP... 13
INSTALLATION MANUAL 2 Contents Getting To Know Your AVANT POLAR HIT TM... 4 Setting up the System... 6 Software Installation... 7 Driver Installation Windows 7... 10 Driver Installation Windows XP... 13
Symbol for 'the device complies with European Medical Device Directive 93/42/EEC requirements' refers to the notified body.
 DL8760 EN-UK Specifications are subject to change without notice 2017 Koninklijke Philips N.V. Find the latest user manual on www.philips.com/support All rights reserved. Philips Consumer Lifestyle BV
DL8760 EN-UK Specifications are subject to change without notice 2017 Koninklijke Philips N.V. Find the latest user manual on www.philips.com/support All rights reserved. Philips Consumer Lifestyle BV
M-400 M-410 M-420 M-430
 USER MANUAL M-400 M-410 M-420 M-430 Please take time to read these instructions before starting to use the scale Version 1.1 10/17 Contents Introduction 3 Product Specification 3 Safety Instructions 4
USER MANUAL M-400 M-410 M-420 M-430 Please take time to read these instructions before starting to use the scale Version 1.1 10/17 Contents Introduction 3 Product Specification 3 Safety Instructions 4
Ceiling Model Ceiling Model for LEDview Plus and Heliodent Plus. Installation Requirements. New as of: English
 New as of: 03.2017 Ceiling Model Ceiling Model for LEDview Plus and Heliodent Plus Installation Requirements English Installation Requirements Ceiling Version = Dentsply Sirona Installation Requirements
New as of: 03.2017 Ceiling Model Ceiling Model for LEDview Plus and Heliodent Plus Installation Requirements English Installation Requirements Ceiling Version = Dentsply Sirona Installation Requirements
Warning: READ ALL INSTRUCTIONS BEFORE USING
 Find the latest user manual on www.philips.com/support Specifications are subject to change without notice. 2017 Koninklijke Philips N.V. All rights reserved Manufactured for: Philips Personal Health a
Find the latest user manual on www.philips.com/support Specifications are subject to change without notice. 2017 Koninklijke Philips N.V. All rights reserved Manufactured for: Philips Personal Health a
EN-US. Warning: READ ALL INSTRUCTIONS BEFORE USING DL8760 ENGLISH
 DL8760 EN-US 1 The numbers in the figures below refer to figure numbers between brackets in the text. 1 2 3 13 12 11 14 4 5 6 7 8 10 9 15 2 2 1 3 5 6 8 11 9 12 4 7 10 13 2-3 cm 4 1 5 "- 1 5 " ENGLISH General
DL8760 EN-US 1 The numbers in the figures below refer to figure numbers between brackets in the text. 1 2 3 13 12 11 14 4 5 6 7 8 10 9 15 2 2 1 3 5 6 8 11 9 12 4 7 10 13 2-3 cm 4 1 5 "- 1 5 " ENGLISH General
User Manual. Glass Body Fat Analyzer LS203-B. version:1.0
 version:1.0 User Manual Glass Body Fat Analyzer LS203-B Contains FCC ID: OU9AW8001-LS GUANGDONG TRANSTEK MEDICAL ELECTRONICS CO., LTD Zone A, 5/F., Investment Building, No. 12, Huizhan East Rd., Torch
version:1.0 User Manual Glass Body Fat Analyzer LS203-B Contains FCC ID: OU9AW8001-LS GUANGDONG TRANSTEK MEDICAL ELECTRONICS CO., LTD Zone A, 5/F., Investment Building, No. 12, Huizhan East Rd., Torch
User Manual TMB-1598-BA. Blood Pressure Monitor. Version:1.0. Wrist Type
 Version:1.0 User Manual Blood Pressure Monitor TMB-1598-BA Wrist Type Guangdong Transtek Medical Electronics Co., Ltd. Zone B, No.105,Dongli Road, Torch Development District, Zhongshan,528437,Guangdong,China
Version:1.0 User Manual Blood Pressure Monitor TMB-1598-BA Wrist Type Guangdong Transtek Medical Electronics Co., Ltd. Zone B, No.105,Dongli Road, Torch Development District, Zhongshan,528437,Guangdong,China
TV Transmitter. User Guide Master
 TV Transmitter User Guide Master Content Before you start 3 Included in delivery 4 Components 4 Getting started 5 Connecting to power supply 5 Connecting to audio devices 6 Pairing the transmitter 7 Daily
TV Transmitter User Guide Master Content Before you start 3 Included in delivery 4 Components 4 Getting started 5 Connecting to power supply 5 Connecting to audio devices 6 Pairing the transmitter 7 Daily
User Manual UAM-880. Blood Pressure Monitor. Arm Type. version:1.0. Thank you for purchasing a ZEWA Blood Pressure Monitor.
 version:1.0 User Manual Blood Pressure Monitor UAM-880 Arm Type Manufactured for Zewa, Inc. 12960 Commerce Lakes Drive#29 Fort Myers, FL 33913 USA www.zewa.com Toll Free Customer Service: 1-888-993-3592
version:1.0 User Manual Blood Pressure Monitor UAM-880 Arm Type Manufactured for Zewa, Inc. 12960 Commerce Lakes Drive#29 Fort Myers, FL 33913 USA www.zewa.com Toll Free Customer Service: 1-888-993-3592
: 0089 GTIN
 GTIN: 00894912002050 PRECAUTIONS PROBE Non-invasive probes are for transcutaneous use only Probe transducer tips are thin and delicate. Be careful not to drop or hit the probe tip. After use, protect the
GTIN: 00894912002050 PRECAUTIONS PROBE Non-invasive probes are for transcutaneous use only Probe transducer tips are thin and delicate. Be careful not to drop or hit the probe tip. After use, protect the
Fully Automatic Blood Pressure Monitor for the Upper Arm INSTRUCTION MANUAL
 Fully Automatic Blood Pressure Monitor for the Upper Arm INSTRUCTION MANUAL GT-868UF Contents Designated use... 26 Precautionary measures... 27 Warranty... 29 Information you should know before operating
Fully Automatic Blood Pressure Monitor for the Upper Arm INSTRUCTION MANUAL GT-868UF Contents Designated use... 26 Precautionary measures... 27 Warranty... 29 Information you should know before operating
BLOOD PRESSURE MONITOR
 E BLOOD PRESSURE MONITOR Instructions and Guarantee BPW-9100 NORMAL BLOOD PRESSURE FLUCTUATION All physical activity, excitement, stress, eating, drinking, smoking, body posture and many other activities
E BLOOD PRESSURE MONITOR Instructions and Guarantee BPW-9100 NORMAL BLOOD PRESSURE FLUCTUATION All physical activity, excitement, stress, eating, drinking, smoking, body posture and many other activities
USER MANUAL. Sens it SENS IT 2.4
 USER MANUAL www.sensit.io Sens it SENS IT 2.4 SUMMARY SAFETY INSTRUCTIONS 4 I. CONTENT OF THE PACK 4 II. PRESENTATION 5 III. HOW TO START 8 IV. TECHNICAL SPECIFICATIONS 9 V. WARNING STATEMENTS 10 VI. CREDITS
USER MANUAL www.sensit.io Sens it SENS IT 2.4 SUMMARY SAFETY INSTRUCTIONS 4 I. CONTENT OF THE PACK 4 II. PRESENTATION 5 III. HOW TO START 8 IV. TECHNICAL SPECIFICATIONS 9 V. WARNING STATEMENTS 10 VI. CREDITS
L NKTEMP. Non-contact. Infrared Thermometer. User Manual LMP001
 L NKTEMP Non-contact Infrared Thermometer User Manual LMP001 TABLE OF CONTENTS Product Description Intended Use Product Features Safety Precautions Using the Thermometer Taking a Measurement Temperature
L NKTEMP Non-contact Infrared Thermometer User Manual LMP001 TABLE OF CONTENTS Product Description Intended Use Product Features Safety Precautions Using the Thermometer Taking a Measurement Temperature
ihealth Wireless Blood Pressure Monitor (BP5) OWNER S MANUAL Table of Contents
 ihealth Wireless Blood Pressure Monitor (BP5) OWNER S MANUAL Table of Contents INTRODUCTION... 2 PACKAGE CONTENTS...2 INTENDED USE... 2 BLOOD PRESSURE CLASSIFICATION FOR ADULTS...2 CONTRAINDICATION...3
ihealth Wireless Blood Pressure Monitor (BP5) OWNER S MANUAL Table of Contents INTRODUCTION... 2 PACKAGE CONTENTS...2 INTENDED USE... 2 BLOOD PRESSURE CLASSIFICATION FOR ADULTS...2 CONTRAINDICATION...3
The following languages can be found on our website: French. German. Spanish. Italian. Swedish. Portuguese. Russian. Dutch.
 Manufactured By: 5580 S. Nogales Hwy. Tucson, Az 85706 USA Telephone: 800-975-7987 Fax: 520-294-6061 www.westmedinc.com PN 74586, Rev. 10 MT Promedt Consulting GmbH Altenhofstr. 80 66386 St. Ingbert, Germany
Manufactured By: 5580 S. Nogales Hwy. Tucson, Az 85706 USA Telephone: 800-975-7987 Fax: 520-294-6061 www.westmedinc.com PN 74586, Rev. 10 MT Promedt Consulting GmbH Altenhofstr. 80 66386 St. Ingbert, Germany
Relief. At Last. User Manual For an effective treatment, read the entire User Manual before using Kyrobak.
 Relief. At Last. User Manual For an effective treatment, read the entire User Manual before using Kyrobak. Contents Know Your Kyrobak 2 Safety Instructions 3 Before You Start 4 How to Use Kyrobak 5 Operating
Relief. At Last. User Manual For an effective treatment, read the entire User Manual before using Kyrobak. Contents Know Your Kyrobak 2 Safety Instructions 3 Before You Start 4 How to Use Kyrobak 5 Operating
Transcutaneous Electrical Nerve Stimulator
 Transcutaneous Electrical Nerve Stimulator Simple, effective pain relief *For the temporary relief of pain as part of current medication programme. Distributor Medi-Direct International Ltd. Unit 24, Wilford
Transcutaneous Electrical Nerve Stimulator Simple, effective pain relief *For the temporary relief of pain as part of current medication programme. Distributor Medi-Direct International Ltd. Unit 24, Wilford
Wireless Blood Pressure Monitor
 Wireless Blood Pressure Monitor Misuratore di pressione wireless Monitor de presión arterial inalámbrico Drahtloses blutdruckmessgerät Draadloze bloeddrukmeter MANUEL DE L'UTILISATEUR MANUAL DEL PROPIETARIO
Wireless Blood Pressure Monitor Misuratore di pressione wireless Monitor de presión arterial inalámbrico Drahtloses blutdruckmessgerät Draadloze bloeddrukmeter MANUEL DE L'UTILISATEUR MANUAL DEL PROPIETARIO
User instructions for the ABI-system 100
 User instructions for the ABI-system 100 0124 User instructions for the ABI-system 100 1 ABI-system 100 2 CA04 cuffs (for upper arms) 2 CL04 cuffs (for legs) 1 ABI software CD 1 USB cable 1 boso mains
User instructions for the ABI-system 100 0124 User instructions for the ABI-system 100 1 ABI-system 100 2 CA04 cuffs (for upper arms) 2 CL04 cuffs (for legs) 1 ABI software CD 1 USB cable 1 boso mains
CRUX II/BTGPS USER GUIDE. Model:D1598
 CRUX II/BTGPS USER GUIDE Model:D1598 0 Federal Communication Commission Interference Statement This equipment has been tested and found to comply with the limits for a Class B digital device, pursuant
CRUX II/BTGPS USER GUIDE Model:D1598 0 Federal Communication Commission Interference Statement This equipment has been tested and found to comply with the limits for a Class B digital device, pursuant
Product Name: COMPER SMART DOPPLER FETAL MONITOR. Product Model: DFMX
 DFMX User Manual Document Num: History of Change: User Manual JS02114DFMX V1.0 Issue Date: Version: Editor: Reviewer: Approver: Date:xx-xx-xx Date:xx-xx-xx Date:xx-xx-xx Product Name: COMPER SMART DOPPLER
DFMX User Manual Document Num: History of Change: User Manual JS02114DFMX V1.0 Issue Date: Version: Editor: Reviewer: Approver: Date:xx-xx-xx Date:xx-xx-xx Date:xx-xx-xx Product Name: COMPER SMART DOPPLER
Product Manual. Getting Started with Roadie 2.
 MOL NUMBER RD200 Product Manual Getting Started with Roadie 2. This manual is a quick start guide for Roadie 2. Please read the following instructions and conditions before using Roadie 2. For a more comprehensive
MOL NUMBER RD200 Product Manual Getting Started with Roadie 2. This manual is a quick start guide for Roadie 2. Please read the following instructions and conditions before using Roadie 2. For a more comprehensive
USER MANUAL MODEL: BM-162
 USER MANUAL MODEL: BM-162 Parents Unit: A. Name Power ON/OFF Key Music Key PTT Key Volume - Key Microphone Power & Low battery indicator LCD display Volume + Key Night Light and torch Key Speaker -Belt
USER MANUAL MODEL: BM-162 Parents Unit: A. Name Power ON/OFF Key Music Key PTT Key Volume - Key Microphone Power & Low battery indicator LCD display Volume + Key Night Light and torch Key Speaker -Belt
SJM MRI Activator. Handheld Device. User's Manual. Model EX4000
 SJM MRI Activator Handheld Device User's Manual Model EX4000 Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical or one of its subsidiaries. ST. JUDE MEDICAL
SJM MRI Activator Handheld Device User's Manual Model EX4000 Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical or one of its subsidiaries. ST. JUDE MEDICAL
Exergen TAT 5000S RS232 TTL Supplemental Instructions for Use
 www.exergen.com/s Exergen TAT 5000S RS232 TTL Supplemental Instructions for Use For additional specifications, see GE Healthcare CARESCAPE V100 Vital Signs Monitor Operator's Manual, Section 12. Symbol
www.exergen.com/s Exergen TAT 5000S RS232 TTL Supplemental Instructions for Use For additional specifications, see GE Healthcare CARESCAPE V100 Vital Signs Monitor Operator's Manual, Section 12. Symbol
2015 Koninklijke Philips N.V. All rights reserved Philips Consumer Lifestyle BV Tussendiepen 4, 9206AD Drachten, Netherlands
 SCH740 2015 Koninklijke Philips N.V. All rights reserved Philips Consumer Lifestyle BV Tussendiepen 4, 9206AD Drachten, Netherlands 4213.354.3816.3 (11/2015) >75% recycled paper >75% papier recyclé 2 3
SCH740 2015 Koninklijke Philips N.V. All rights reserved Philips Consumer Lifestyle BV Tussendiepen 4, 9206AD Drachten, Netherlands 4213.354.3816.3 (11/2015) >75% recycled paper >75% papier recyclé 2 3
ENGLISH. Important safety information Read this important information carefully before you use the device and save it for future reference.
 SCH740 EN www.philips.com/avent Philips Consumer Lifestyle BV Tussendiepen 4, 9206 AD Drachten, Netherlands Trademarks owned by the Philips Group. 2017 Koninklijke Philips N.V. All rights reserved Find
SCH740 EN www.philips.com/avent Philips Consumer Lifestyle BV Tussendiepen 4, 9206 AD Drachten, Netherlands Trademarks owned by the Philips Group. 2017 Koninklijke Philips N.V. All rights reserved Find
Husqvarna Fleet Services Machine Sensor. Machine Sensor Please read these instructions carefully before using the device. Original instructions
 Husqvarna Fleet Services Machine Sensor Machine Sensor Please read these instructions carefully before using the device. Original instructions Safety and Regulatory Information IMPORTANT: A complete User
Husqvarna Fleet Services Machine Sensor Machine Sensor Please read these instructions carefully before using the device. Original instructions Safety and Regulatory Information IMPORTANT: A complete User
Wireless SingStar Microphone Instruction Manual
 Wireless SingStar Microphone Instruction Manual 2008 Sony Computer Entertainment Europe. SingStar is a trademark or a registered trademark of Sony Computer Entertainment Europe. 2, PLAYSTATION and PlayStation
Wireless SingStar Microphone Instruction Manual 2008 Sony Computer Entertainment Europe. SingStar is a trademark or a registered trademark of Sony Computer Entertainment Europe. 2, PLAYSTATION and PlayStation
Enphase. Mobile Connect Installation Guide
 Enphase Mobile Connect Installation Guide Mobile Connect Installation Guide Contact Information Enphase Energy Inc. 1420 N. McDowell Blvd. Petaluma, CA 94954 http://www.enphase.com support@enphaseenergy.com
Enphase Mobile Connect Installation Guide Mobile Connect Installation Guide Contact Information Enphase Energy Inc. 1420 N. McDowell Blvd. Petaluma, CA 94954 http://www.enphase.com support@enphaseenergy.com
OPERATOR'S INSTRUCTIONS DENTAL X-RAY MODEL 096
 DENTAL X-RAY MODEL 096 OPERATOR'S INSTRUCTIONS 0197 WARNING: This X-ray equipment may be dangerous to patients and operators unless safe exposure factors and operating instructions are observed. R Table
DENTAL X-RAY MODEL 096 OPERATOR'S INSTRUCTIONS 0197 WARNING: This X-ray equipment may be dangerous to patients and operators unless safe exposure factors and operating instructions are observed. R Table
H2 Check Operating Manual
 H2 Check Operating Manual Federal (USA) law restricts this device to sale by or on the order of a physician or licensed practitioner. Micro Direct, Inc. 803 Webster Street Lewiston, ME 04240 1-800-588-3381
H2 Check Operating Manual Federal (USA) law restricts this device to sale by or on the order of a physician or licensed practitioner. Micro Direct, Inc. 803 Webster Street Lewiston, ME 04240 1-800-588-3381
Compressor Nebulizer. Model No: 9R-021 series. Please read the instruction manual before use.
 Compressor Nebulizer Model No: 9R-021 series Please read the instruction manual before use. IMPORTANT SAFEGUARDS...... 1 1. INTRODUCTION... 6 2. PRODUCT DESCRIPTION... 7 3. OPERATION... 8 4. CLEANING...
Compressor Nebulizer Model No: 9R-021 series Please read the instruction manual before use. IMPORTANT SAFEGUARDS...... 1 1. INTRODUCTION... 6 2. PRODUCT DESCRIPTION... 7 3. OPERATION... 8 4. CLEANING...
OPERATOR S MANUAL AN APPLIED DIGITAL COMPANY
 OPERATOR S MANUAL AN APPLIED DIGITAL COMPANY Contents: VeriChip H2 Reader Assembly Part Number 600-000515-000 (includes all of the following): USER INSTRUCTIONS Description Part Number Description The
OPERATOR S MANUAL AN APPLIED DIGITAL COMPANY Contents: VeriChip H2 Reader Assembly Part Number 600-000515-000 (includes all of the following): USER INSTRUCTIONS Description Part Number Description The
CCR24T CCR24R. User s Guide WIRELESS TRANSMITTER SYSTEM WARRANTY SERVICE CARD WARRANTY CARD
 WARRANTY SERVICE CARD WARRANTY CARD PRODUCT NAME Wireless Transceiver System PERIOD MODEL NAME CCR24GEN YEAR PURCHASE DATE.. 200_ From the date of WARRANTY PERIOD.. 200_ purchase. CUSTOMER S ADDRESS :
WARRANTY SERVICE CARD WARRANTY CARD PRODUCT NAME Wireless Transceiver System PERIOD MODEL NAME CCR24GEN YEAR PURCHASE DATE.. 200_ From the date of WARRANTY PERIOD.. 200_ purchase. CUSTOMER S ADDRESS :
INSTALLATION MANUAL AVANT HIT+ Hearing Instrument Test Chamber.
 INSTALLATION MANUAL AVANT Hearing Instrument Test Chamber HIT+ www.medrx-int.com Contents Getting To Know Your AVANT HIT+. 3 Computer Requirements 4 Setup System. 5 Software Installation.. 6 Sound Card
INSTALLATION MANUAL AVANT Hearing Instrument Test Chamber HIT+ www.medrx-int.com Contents Getting To Know Your AVANT HIT+. 3 Computer Requirements 4 Setup System. 5 Software Installation.. 6 Sound Card
Complete guide to icube II. Wireless programming
 GB Complete guide to icube II Wireless programming Table of contents Quick reference page...3 Indicator lights...4 Overview...5 First time setup...6 Connecting icube II to your computer...7 Battery status...8
GB Complete guide to icube II Wireless programming Table of contents Quick reference page...3 Indicator lights...4 Overview...5 First time setup...6 Connecting icube II to your computer...7 Battery status...8
START HERE HEARPHONES CONVERSATION-ENHANCING HEADPHONES. Please read and keep all safety and use instructions.
 START HERE HEARPHONES CONVERSATION-ENHANCING HEADPHONES Please read and keep all safety and use instructions. Bose Hear Download the Bose Hear app. Press the Power/Bluetooth button. If wearing the headphones,
START HERE HEARPHONES CONVERSATION-ENHANCING HEADPHONES Please read and keep all safety and use instructions. Bose Hear Download the Bose Hear app. Press the Power/Bluetooth button. If wearing the headphones,
What is Moto? bring happiness to life. The lights Pressuresensitive
 User Manual 1 bring happiness to life What is Moto? Moto tiles are a tool for physical play. They are included under the product category Playware, which combines robotics with play to create products
User Manual 1 bring happiness to life What is Moto? Moto tiles are a tool for physical play. They are included under the product category Playware, which combines robotics with play to create products
HD STETH TM Quick Start User Guide
 Thank you and for choosing the futuristic HD Steth TM manufactured by HD Medical Inc. USA Indications for Use (IFU) HD Steth is an electronic stethoscope meant to assist a qualified clinician to capture,
Thank you and for choosing the futuristic HD Steth TM manufactured by HD Medical Inc. USA Indications for Use (IFU) HD Steth is an electronic stethoscope meant to assist a qualified clinician to capture,
Multi-Control Panel for built-in system. Multi Pad for NLX / imd OPERATION MANUAL OM-E0538E 001
 Multi-Control Panel for built-in system Multi Pad for NLX / imd OPERATION MANUAL OM-E0538E 001 Thank you for purchasing Multi Pad. Read this Operation Manual carefully before use for operation instructions
Multi-Control Panel for built-in system Multi Pad for NLX / imd OPERATION MANUAL OM-E0538E 001 Thank you for purchasing Multi Pad. Read this Operation Manual carefully before use for operation instructions
By Paul Aylett at 12:08 pm, Apr 13, 2016
 TD2 Series Content Introduction Parts Features General Warnings and Safety Using your TENS Pain Reliever Troubleshooting Positions for use Specification Compatibility EMC Maintenance and Cautions Explanation
TD2 Series Content Introduction Parts Features General Warnings and Safety Using your TENS Pain Reliever Troubleshooting Positions for use Specification Compatibility EMC Maintenance and Cautions Explanation
User Manual. Before Using Your WiTouch Pro Device
 User Manual Before Using Your WiTouch Pro Device Sync the Remote Control and the WiTouch Pro Device. Using the provided screwdriver, remove the back cover from the WiTouch Pro device. 2. Remove the clear
User Manual Before Using Your WiTouch Pro Device Sync the Remote Control and the WiTouch Pro Device. Using the provided screwdriver, remove the back cover from the WiTouch Pro device. 2. Remove the clear
CardioMessenger II-S Transmitter for BIOTRONIK Home Monitoring. Technical Manual
 CardioMessenger II-S Transmitter for BIOTRONIK Home Monitoring 362454_D_GA_CM_II-S_US.indd 1 Technical Manual 17.04.08 16:28:16 by BIOTRONIK GmbH & Co. KG All rights reserved. Specifications subject to
CardioMessenger II-S Transmitter for BIOTRONIK Home Monitoring 362454_D_GA_CM_II-S_US.indd 1 Technical Manual 17.04.08 16:28:16 by BIOTRONIK GmbH & Co. KG All rights reserved. Specifications subject to
Product Manual N.2 WEARABLE ECG. PRODUCT MANUAL
 Product Manual WEARABLE ECG www.vitaljacket.com www.biodevices.pt Biodevices S.A. Av. D. Afonso Henriques, 1462 1º Traseiras 4450-013 Matosinhos Portugal N.2 O NOVO NORTE PROGRAMA OPERACIONAL REGIONAL
Product Manual WEARABLE ECG www.vitaljacket.com www.biodevices.pt Biodevices S.A. Av. D. Afonso Henriques, 1462 1º Traseiras 4450-013 Matosinhos Portugal N.2 O NOVO NORTE PROGRAMA OPERACIONAL REGIONAL
Remote Control Outlets Operating Instructions
 Remote Control Outlets Operating Instructions - FOR INDOOR OR OUTDOOR USE - IMPORTANT SAFEGUARDS Signal Word Definitions NOTE: These are general definitions only; all may not pertain to the actual product
Remote Control Outlets Operating Instructions - FOR INDOOR OR OUTDOOR USE - IMPORTANT SAFEGUARDS Signal Word Definitions NOTE: These are general definitions only; all may not pertain to the actual product
ADAE portable X ray machine Operation manual
 ADAE portable X ray machine Operation manual CONTENTS ABOUT THE SYMBOL----------------------------------------------------------3 INTRODUCTION-----------------------------------------------------------------4
ADAE portable X ray machine Operation manual CONTENTS ABOUT THE SYMBOL----------------------------------------------------------3 INTRODUCTION-----------------------------------------------------------------4
Quick Start Guide. Antenna Alignment Tool AIMWLLR0-35. QSG rev 7 AIMWLLR0-35 [NRB-0200] QSG.indd 1
![Quick Start Guide. Antenna Alignment Tool AIMWLLR0-35. QSG rev 7 AIMWLLR0-35 [NRB-0200] QSG.indd 1 Quick Start Guide. Antenna Alignment Tool AIMWLLR0-35. QSG rev 7 AIMWLLR0-35 [NRB-0200] QSG.indd 1](/thumbs/86/94268876.jpg) Quick Start Guide Antenna Alignment Tool AIMWLLR0-35 QSG-00097 rev 7 AIMWLLR0-35 [NRB-0200] QSG.indd 1 Welcome This quick start guide is designed to familiarize you with the features and use of the NetComm
Quick Start Guide Antenna Alignment Tool AIMWLLR0-35 QSG-00097 rev 7 AIMWLLR0-35 [NRB-0200] QSG.indd 1 Welcome This quick start guide is designed to familiarize you with the features and use of the NetComm
Draft. User s Manual. Transmitter Model EX1150
 User s Manual Merlin @home Transmitter Model EX1150 CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. 2008 St. Jude Medical Cardiac Rhythm Management Division.
User s Manual Merlin @home Transmitter Model EX1150 CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. 2008 St. Jude Medical Cardiac Rhythm Management Division.
BodyclockStimplusManual.qx8_Layout 1 17/08/ :53 Page 1. Body Clock Stimplus. Instructions for use
 BodyclockStimplusManual.qx8_Layout 1 17/08/2012 11:53 Page 1 Body Clock Stimplus TM Instructions for use BodyclockStimplusManual.qx8_Layout 1 17/08/2012 11:53 Page 2 Acupuncture is an ancient Chinese therapy
BodyclockStimplusManual.qx8_Layout 1 17/08/2012 11:53 Page 1 Body Clock Stimplus TM Instructions for use BodyclockStimplusManual.qx8_Layout 1 17/08/2012 11:53 Page 2 Acupuncture is an ancient Chinese therapy
MDW-45 Converter RS RS-422/485
 www.westermo.com MDW-45 Converter RS-232 - RS-422/485 2 6617-2203 General information Legal information The contents of this document are provided as is. Except as required by applicable law, no warranties
www.westermo.com MDW-45 Converter RS-232 - RS-422/485 2 6617-2203 General information Legal information The contents of this document are provided as is. Except as required by applicable law, no warranties
DRAFT ONLY FOR CLINICAL TESTING Nucleus 7 Remote Control User Guide CR310
 DRAFT ONLY FOR CLINICAL TESTING Nucleus 7 Remote Control User Guide CR310 The Cochlear Nucleus 7 Remote Control (model number: CR310) is a hand-held device for controlling the commonly used functions of
DRAFT ONLY FOR CLINICAL TESTING Nucleus 7 Remote Control User Guide CR310 The Cochlear Nucleus 7 Remote Control (model number: CR310) is a hand-held device for controlling the commonly used functions of
OPERATOR'S INSTRUCTIONS DX-073 DENTAL X-RAY
 DENTAL X-RAY DX-073 Wall Mount Type...WK Ceiling Mount Type...CK Floor Mount Type...FK1/2 Mobil Mount Type...FM Room Mount Type...RKII OPERATOR'S INSTRUCTIONS 0197 WARNING : This X-ray equipment may be
DENTAL X-RAY DX-073 Wall Mount Type...WK Ceiling Mount Type...CK Floor Mount Type...FK1/2 Mobil Mount Type...FM Room Mount Type...RKII OPERATOR'S INSTRUCTIONS 0197 WARNING : This X-ray equipment may be
USER'S GUIDE. EcoQuest International 310 T. Elmer Cox Drive Greeneville, TN (423) Setup. Operation. Specifications.
 USER'S GUIDE Setup Operation Specifications Warranty (43) 638-746 CAUTION: Read manual carefully for proper procedures and operation. CONTENTS SPECIFICATIONS Specifications...............................................................3
USER'S GUIDE Setup Operation Specifications Warranty (43) 638-746 CAUTION: Read manual carefully for proper procedures and operation. CONTENTS SPECIFICATIONS Specifications...............................................................3
SAFETY INFORMATION IMPORTANT FCC LICENSING INFORMATION
 This device complies with part 15 of the FCC Rules. Operation is subject to the following two conditions: (1) This device does not cause harmful interference, and (2) This device must accept any interference
This device complies with part 15 of the FCC Rules. Operation is subject to the following two conditions: (1) This device does not cause harmful interference, and (2) This device must accept any interference
Philips body analysis scale with Bluetooth Smart DL8781, DL8780 EN-US
 Philips body analysis scale with Bluetooth Smart DL8781, DL8780 EN-US 1 3 4 5 1 2 6 7 8 9 10 11 12 English 6 6 English Contents Introduction 7 General 7 Explanation of symbols 8 General description (Fig.
Philips body analysis scale with Bluetooth Smart DL8781, DL8780 EN-US 1 3 4 5 1 2 6 7 8 9 10 11 12 English 6 6 English Contents Introduction 7 General 7 Explanation of symbols 8 General description (Fig.
USER MANUAL Digital Wireless Gateway U9120-W4 (P/N: 44002G-01)
 USER MANUAL Digital Wireless Gateway U9120-W4 (P/N: 44002G-01) 19549P-82 (11-16) 2016 DAVID CLARK COMPANY INCORPORATED Cautions and Warnings READ AND SAVE THESE INSTRUCTIONS. Follow the instructions in
USER MANUAL Digital Wireless Gateway U9120-W4 (P/N: 44002G-01) 19549P-82 (11-16) 2016 DAVID CLARK COMPANY INCORPORATED Cautions and Warnings READ AND SAVE THESE INSTRUCTIONS. Follow the instructions in
Osmolarity System USER MANUAL FOR QUANTITATIVE MEASUREMENT OF OSMOLARITY OF OCULAR TISSUES. osdcare.com
 Osmolarity System FOR QUANTITATIVE MEASUREMENT OF OSMOLARITY OF OCULAR TISSUES USER MANUAL osdcare.com I-PEN User Guide I-PEN User Guide TABLE OF CONTENTS I-PEN I-PEN is a trademark of I-MED Pharma Inc.
Osmolarity System FOR QUANTITATIVE MEASUREMENT OF OSMOLARITY OF OCULAR TISSUES USER MANUAL osdcare.com I-PEN User Guide I-PEN User Guide TABLE OF CONTENTS I-PEN I-PEN is a trademark of I-MED Pharma Inc.
