Application Note. The New 2D Superresolution Mode for ZEISS Airyscan 120 nm Lateral Resolution without Acquiring a Z-stack
|
|
|
- Ethan Harrington
- 6 years ago
- Views:
Transcription
1 The New 2D Superresolution Mode for ZEISS Airyscan 120 nm Lateral Resolution without Acquiring a Z-stack
2 The New 2D Superresolution Mode for ZEISS Airyscan 120 nm Lateral Resolution without Acquiring a Z-stack Author: Joseph Huff, Annette Bergter, Jan Birkenbeil, Ingo Kleppe, Ralf Engelmann Carl Zeiss Microscopy GmbH, Germany Date: November 2017 Utilizing a pinhole-plane imaging concept, Airyscan allows for simultaneous improvement in resolution and signal-to-noise by capitalizing on an innovative 32-channel GaAsP photomultiplier tube (PMT) array detector. Each detection channel functions as a very small pinhole to increase resolution while the overall detector design delivers better signal-to-noise than traditional GaAsP-based confocal systems. In the past, a stack of at least five z-slices had to be deconvolved to get usable images with an optical section thinner than 1 Airy unit. Now, the new 2D Superresolution mode for Airyscan delivers images with the thinnest optical section (0.2 Airy units) from a single image while maintaining the light collection efficiency of a much larger 1.25-Airy-unit pinhole. Introduction The creation of optical section images in biomedical research started in 1955, when Marvin Minsky laid the foundation for confocal microscopy 1. In an effort to improve the contrast of images for the study of the central nervous system, Minsky placed an aperture in a conjugate plane relative to the microscope objective s focus. He discovered that he could create an optical section within a three-dimensional biological sample by blocking any light that did not originate from the focal plane. Hence, Minsky was able to generate high-contrast images to study nervous system circuitry (prepared via the Golgi method) free from out-of-focus scatter from imaging planes deep within scattering samples. This confocal concept was the foundation of modern laser scanning confocal microscopy. Creation of the optical section Conventional confocal laser scanning microscopes leverage a combination of a physical aperture (the so-called pinhole) and a unitary detector (PMT, hybrid detector, avalanche photodiode, etc.) to create an optically sectioned image 2. D. Figure 1 Dependence of optical section thickness (A), resolution (B) and signal transmission (C) on pinhole diameter. The Airyscan principal illustrated to show the projection of 1.25 AU onto a 32 element GaAsP PMT array (D). 2
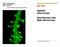
3 Intensity 70,000 60,000 50,000 40,000 30,000 20,000 10, Distance (micron) Figure 2 Airyscan technology can resolve the labeled ends of nanorulers with the defined distance of 120 nm, using a 2D image (1 optical section). The measurement line within the image represents a distance of 110 nm and both ends can be clearly discriminated. GATTA-SIM 120B, distance = 120 nm, Alexa 488. GATTAquant DNA Nanotechnologies, Naturally, by varying the aperture size, one can also change the thickness of the optical section (Fig. 1a). In addition to the thickness of the optical section, the resulting image resolution and image signal-to-noise are directly related to the aperture size (Fig. 1b, c). As the pinhole is closed, less signal can reach the unitary detector, which significantly reduces the image signal-to-noise. Traditional confocal microscopes demand the sacrifice of optical sectioning and resolution for the sake of a usable signal-to-noise ratio and acceptable sample bleaching and phototoxicity. Consequently, a pinhole size of 1 Airy unit (AU) has become the default for confocal microscopes. In contrast to traditional confocal detectors, Airyscan combines the resolution benefits of imaging with a small pinhole with the collection efficiency of a large pinhole. This design delivers a simultaneous increase in resolution and signal-tonoise by projecting 1.25 AU onto a 32-channel GaAsP PMT array detector. Each detector element behaves as a 0.2-AU pinhole while the collection efficiency of a 1.25-AU pinhole is maintained 3, 4 (Fig. 1d). Moreover, linear deconvolution is used to extend the resolution beyond what a 0.2-AU pinhole provides, resulting in a resolution increase in all three spatial dimensions. The new 2D Superresolution mode for Airyscan improves the optical sectioning by a factor of 2.4 for a single optical section by utilizing a new exclusive processing algorithm in place of the linear deconvolution step 5, 6 (Fig. 1a and 3). Along with improved optical sectioning, Airyscan now provides lateral resolution to 120 nm for 2D and 3D data sets (z-stacks) and 350 nm axial resolution for z-stacks (Fig. 2). The new 2D Superresolution mode Since its launch, Airyscan s purpose has been to ensure that all precious fluorescence emission photons are preserved to provide the highest sensitivity of any confocal. This focus on system sensitivity has resulted in unprecedented image quality, resolution and sectioning for 3D stacks. Airyscan 2D images have always offered an improvement in lateral resolution while maintaining an optical slice thickness of 1.25 AU Figure 3 Drosophila melanogaster neuromuscular junctions stained for Bruchpilot (BRP). BRP is arranged in a characteristic ring-like structure, which cannot be resolved by confocal images (A). Airyscan 2D processing is able to resolve most of the structure (B), while the new Airyscan 2D Superresolution mode (C) reveals more detail. Some neuromuscular junctions that are slightly visible out of focus in (B) are no longer present in (C, arrow), due to the thinner optical section. Sample courtesy of J. Pielage, Friedrich Miescher Institute for Biomedical Research, Basel, Switzerland 3
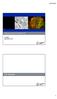
4 Figure 4 These series of images, showing single sections of the anterior central nervous system (labelled by Alexa 561) of a larval Platynereis dumerilii, depict the increasing sectioning power of confocal imaging techniques. In fluorescent widefield (A), most of the structural information is lost due to the out-of-focus light contributing to the image. To eliminate this blur, confocal microscope remove the out-of-focus emission (B) by producing an optical sectioning using a pinhole. Capturing the same image section with an Airyscan detector improved resolution of finer structures even further (C) and finally allowed to capture the perfect optical section with Airyscan 2D Superresolution (D) utilizing the best resolution of a sole optical slice. (Fig. 3 and 4). The new 2D Superresolution mode for Airyscan takes advantage of the fact that the confocal point spread function entangles information from the x, y and z planes in the pinhole plane. Airyscan measures the emission fluorescence distribution already in a single image acquisition, thereby yielding information about how the signal is entangled. The new exclusive algorithm of the 2D Superresolution mode makes it possible to distinguish and separate the light originating in the focal plane from light originating outside of the focal plane. This allows researchers to obtain perfect optical sections, without the need to acquire a z-stack (Fig. 3 5). To get the same optical section on a traditional confocal, a researcher would have to close the pinhole to 0.2 AU and sacrifice > 95 % of all emission signal (in both the plane of interest and out-of-focus planes). This would limit the information that could be obtained from the plane of interest. Summary The Airyscan detector from ZEISS delivers more information from the pinhole plane than traditional confocal microscopes. With this information, researchers get simultaneous improvements in resolution and signal-to-noise and an increase in acquisition speeds. The new 2D Superresolution mode improves the achievable resolution of a confocal microscope to 120 nm laterally and delivers a 2.4 thinner optical section. In image stacks, an axial resolution of 350 nm is achieved. 4
5 Figure 5 TOMM 20 of the outer membrane of the mitochondria is labelled by Alexa 568 in this series of images, depicting single optical slices. While the lumen of the mitochondria cannot be resolved by pure confocal imaging (A), it can be visualized by Airyscan imaging and 2D processing (B). To improve resolution even further using Airyscan, the acquisition of several z-slices was necessary (C, one optical section depicted). This full resolution potential can now be utilized with one single z-section (D), limiting light exposure and time effort to the absolute minimum. The lower row of images show a corresponding region (blue frame) at higher magnification. Note the difference of Airyscan 2D (B) compared to Airyscan 3D processing (C) and 2D Superresolution (D) processing (arrows), to clearly visualize the mitochondrial lumen in (C) and (D). Sample: courtesy of Michael W. Davidson, The Florida State University, Tallahassee, USA References: [1] Minsky, M. Scanning 10, (1988). [2] Conchello, J.-A. & Lichtman, J.W. Nat. Methods 2, (2005). [3] Huff, J. Nat. Methods 12, i ii (2015). [4] Huff, J. Nat. Methods 12, i ii (2016). [5] Wilhelm, S. Confocal Laser Scanning Microscopy (ZEISS, 2010). [6] Pawley, J. Handbook of Biological Confocal Microscopy 3 rd ed. (Springer, 2006). 5
6 Not for therapeutic, treatment or medical diagnostic evidence. Not all products are available in every country. Contact your local ZEISS representative for more information. EN_41_013_161 CZ Design, scope of delivery and technical progress subject to change without notice. Carl Zeiss Microscopy GmbH Carl Zeiss Microscopy GmbH Jena, Germany
Technology Note ZEISS LSM 880 with Airyscan
 Technology Note ZEISS LSM 880 with Airyscan Introducing the Fast Acquisition Mode ZEISS LSM 880 with Airyscan Introducing the Fast Acquisition Mode Author: Dr. Annette Bergter Carl Zeiss Microscopy GmbH,
Technology Note ZEISS LSM 880 with Airyscan Introducing the Fast Acquisition Mode ZEISS LSM 880 with Airyscan Introducing the Fast Acquisition Mode Author: Dr. Annette Bergter Carl Zeiss Microscopy GmbH,
Point Spread Function. Confocal Laser Scanning Microscopy. Confocal Aperture. Optical aberrations. Alternative Scanning Microscopy
 Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
Bi177 Lecture 5 Adding the Third Dimension Wide-field Imaging Point Spread Function Deconvolution Confocal Laser Scanning Microscopy Confocal Aperture Optical aberrations Alternative Scanning Microscopy
Technology Note. The Airyscan Detector from ZEISS Confocal Imaging with Improved Signal-to-Noise Ratio and Superresolution
 Technology Note Confocal The Airyscan Detector from ZEISS Confocal Imaging with Improved Signal-to-Noise Ratio and Superresolution Airyscan The Airyscan Detector from ZEISS Confocal Imaging with Improved
Technology Note Confocal The Airyscan Detector from ZEISS Confocal Imaging with Improved Signal-to-Noise Ratio and Superresolution Airyscan The Airyscan Detector from ZEISS Confocal Imaging with Improved
Travel to New Dimensions- LSM 880. The Resolution of a Microscope is limited. The Resolution of a Microscope is limited. Image. Image. Object.
 Travel to New Dimensions- LSM 880 LSM 880: The Power of Sensitivity Our Latest Member of the LSM 880 with GaAsP Detectors Sensitivity, and Ease of Use Innovative High-End Laser Scanning Microscopes from
Travel to New Dimensions- LSM 880 LSM 880: The Power of Sensitivity Our Latest Member of the LSM 880 with GaAsP Detectors Sensitivity, and Ease of Use Innovative High-End Laser Scanning Microscopes from
Multifluorescence The Crosstalk Problem and Its Solution
 Multifluorescence The Crosstalk Problem and Its Solution If a specimen is labeled with more than one fluorochrome, each image channel should only show the emission signal of one of them. If, in a specimen
Multifluorescence The Crosstalk Problem and Its Solution If a specimen is labeled with more than one fluorochrome, each image channel should only show the emission signal of one of them. If, in a specimen
BASICS OF CONFOCAL IMAGING (PART I)
 BASICS OF CONFOCAL IMAGING (PART I) INTERNAL COURSE 2012 LIGHT MICROSCOPY Lateral resolution Transmission Fluorescence d min 1.22 NA obj NA cond 0 0 rairy 0.61 NAobj Ernst Abbe Lord Rayleigh Depth of field
BASICS OF CONFOCAL IMAGING (PART I) INTERNAL COURSE 2012 LIGHT MICROSCOPY Lateral resolution Transmission Fluorescence d min 1.22 NA obj NA cond 0 0 rairy 0.61 NAobj Ernst Abbe Lord Rayleigh Depth of field
Confocal and 2-photon Imaging. October 15, 2010
 Confocal and 2-photon Imaging October 15, 2010 Review Optical Elements Adapted from Sluder & Nordberg 2007 Review Optical Elements Collector Lens Adapted from Sluder & Nordberg 2007 Review Optical Elements
Confocal and 2-photon Imaging October 15, 2010 Review Optical Elements Adapted from Sluder & Nordberg 2007 Review Optical Elements Collector Lens Adapted from Sluder & Nordberg 2007 Review Optical Elements
Bi/BE 227 Winter Assignment #3. Adding the third dimension: 3D Confocal Imaging
 Bi/BE 227 Winter 2016 Assignment #3 Adding the third dimension: 3D Confocal Imaging Schedule: Jan 20: Assignment Jan 20-Feb 8: Work on assignment Feb 10: Student PowerPoint presentations. Goals for this
Bi/BE 227 Winter 2016 Assignment #3 Adding the third dimension: 3D Confocal Imaging Schedule: Jan 20: Assignment Jan 20-Feb 8: Work on assignment Feb 10: Student PowerPoint presentations. Goals for this
The Zeiss AiryScan System, Confocal Four.
 The Zeiss AiryScan System, Confocal Four. Overview. The Zeiss AiryScan module is a segmented, radially stacked GaASP detector and collector system designed to subsample the airy disk of a point emission
The Zeiss AiryScan System, Confocal Four. Overview. The Zeiss AiryScan module is a segmented, radially stacked GaASP detector and collector system designed to subsample the airy disk of a point emission
Confocal Microscopy. (Increasing contrast and resolu6on using op6cal sec6oning) Lecture 7. November 2017
 Confocal Microscopy (Increasing contrast and resolu6on using op6cal sec6oning) Lecture 7 November 2017 3 Flavours of Microscope Confocal Laser Scanning Problem: Out of Focus Light Spinning disc 2-Photon
Confocal Microscopy (Increasing contrast and resolu6on using op6cal sec6oning) Lecture 7 November 2017 3 Flavours of Microscope Confocal Laser Scanning Problem: Out of Focus Light Spinning disc 2-Photon
Bio 407. Applied microscopy. Introduction into light microscopy. José María Mateos. Center for Microscopy and Image Analysis
 Center for Microscopy and Image Analysis Bio 407 Applied Introduction into light José María Mateos Fundamentals of light Compound microscope Microscope composed of an objective and an additional lens (eyepiece,
Center for Microscopy and Image Analysis Bio 407 Applied Introduction into light José María Mateos Fundamentals of light Compound microscope Microscope composed of an objective and an additional lens (eyepiece,
Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope
 Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Verify that main power switches on the
Training Guide for Carl Zeiss LSM 5 LIVE Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Verify that main power switches on the
Basics of confocal imaging (part I)
 Basics of confocal imaging (part I) Swiss Institute of Technology (EPFL) Faculty of Life Sciences Head of BIOIMAGING AND OPTICS BIOP arne.seitz@epfl.ch Lateral resolution BioImaging &Optics Platform Light
Basics of confocal imaging (part I) Swiss Institute of Technology (EPFL) Faculty of Life Sciences Head of BIOIMAGING AND OPTICS BIOP arne.seitz@epfl.ch Lateral resolution BioImaging &Optics Platform Light
Training Guide for Leica SP8 Confocal/Multiphoton Microscope
 Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
Training Guide for Leica SP8 Confocal/Multiphoton Microscope LAS AF v3.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON power switch for epifluorescence
Zeiss 780 Training Notes
 Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
Zeiss 780 Training Notes Turn on Main Switch, System PC and Components Switches 780 Start up sequence Do you need the argon laser (458, 488, 514 nm lines)? Yes Turn on the laser s main power switch and
Training Guide for Carl Zeiss LSM 510 META Confocal Microscope
 Training Guide for Carl Zeiss LSM 510 META Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON Components and System/PC switches
Training Guide for Carl Zeiss LSM 510 META Confocal Microscope AIM 4.2 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2017) Power ON Routine 1 2 Turn ON Components and System/PC switches
Practical work no. 3: Confocal Live Cell Microscopy
 Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Practical work no. 3: Confocal Live Cell Microscopy Course Instructor: Mikko Liljeström (MIU) 1 Background Confocal microscopy: The main idea behind confocality is that it suppresses the signal outside
Training Guide for Carl Zeiss LSM 880 with AiryScan FAST
 Training Guide for Carl Zeiss LSM 880 with AiryScan FAST ZEN 2.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2018) Power ON Routine 1 2 Turn ON Main Switch from the remote control
Training Guide for Carl Zeiss LSM 880 with AiryScan FAST ZEN 2.3 Optical Imaging & Vital Microscopy Core Baylor College of Medicine (2018) Power ON Routine 1 2 Turn ON Main Switch from the remote control
Non-Descanned FLIM Detection in Multiphoton Microscopes
 Non-Descanned FLIM Detection in Multiphoton Microscopes Abstract. Multiphoton microscopes use a femtosecond NIR laser to excite fluorescence in the sample. Excitation is performed via a multi-photon absorption
Non-Descanned FLIM Detection in Multiphoton Microscopes Abstract. Multiphoton microscopes use a femtosecond NIR laser to excite fluorescence in the sample. Excitation is performed via a multi-photon absorption
Shreyash Tandon M.S. III Year
 Shreyash Tandon M.S. III Year 20091015 Confocal microscopy is a powerful tool for generating high-resolution images and 3-D reconstructions of a specimen by using point illumination and a spatial pinhole
Shreyash Tandon M.S. III Year 20091015 Confocal microscopy is a powerful tool for generating high-resolution images and 3-D reconstructions of a specimen by using point illumination and a spatial pinhole
Zeiss 880 Training Notes Zen 2.3
 Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
Zeiss 880 Training Notes Zen 2.3 1 Turn on the HXP 120V Lamp 2 Turn on Main Power Switch Turn on the Systems PC Switch Turn on the Components Switch. 3 4 5 Turn on the PC and log into your account. Start
Introduction to light microscopy
 Center for Microscopy and Image Anaylsis Introduction to light microscopy Basic concepts of imaging with light Urs Ziegler ziegler@zmb.uzh.ch Light interacting with matter Absorbtion Refraction Diffraction
Center for Microscopy and Image Anaylsis Introduction to light microscopy Basic concepts of imaging with light Urs Ziegler ziegler@zmb.uzh.ch Light interacting with matter Absorbtion Refraction Diffraction
Light Microscopy. Upon completion of this lecture, the student should be able to:
 Light Light microscopy is based on the interaction of light and tissue components and can be used to study tissue features. Upon completion of this lecture, the student should be able to: 1- Explain the
Light Light microscopy is based on the interaction of light and tissue components and can be used to study tissue features. Upon completion of this lecture, the student should be able to: 1- Explain the
3D light microscopy techniques
 3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 1 1 2! NA = 0.5! NA 2D imaging
Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev. Microscopy course, Michmoret Dec 2005
 Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev Why use confocal microscopy? Principles of the laser scanning confocal microscope. Image resolution. Manipulating the
Why and How? Daniel Gitler Dept. of Physiology Ben-Gurion University of the Negev Why use confocal microscopy? Principles of the laser scanning confocal microscope. Image resolution. Manipulating the
Development of a High-speed Super-resolution Confocal Scanner
 Development of a High-speed Super-resolution Confocal Scanner Takuya Azuma *1 Takayuki Kei *1 Super-resolution microscopy techniques that overcome the spatial resolution limit of conventional light microscopy
Development of a High-speed Super-resolution Confocal Scanner Takuya Azuma *1 Takayuki Kei *1 Super-resolution microscopy techniques that overcome the spatial resolution limit of conventional light microscopy
INTRODUCTION TO MICROSCOPY. Urs Ziegler THE PROBLEM
 INTRODUCTION TO MICROSCOPY Urs Ziegler ziegler@zmb.uzh.ch THE PROBLEM 1 ORGANISMS ARE LARGE LIGHT AND ELECTRONS: ELECTROMAGNETIC WAVES v = Wavelength ( ) Speed (v) Frequency ( ) Amplitude (A) Propagation
INTRODUCTION TO MICROSCOPY Urs Ziegler ziegler@zmb.uzh.ch THE PROBLEM 1 ORGANISMS ARE LARGE LIGHT AND ELECTRONS: ELECTROMAGNETIC WAVES v = Wavelength ( ) Speed (v) Frequency ( ) Amplitude (A) Propagation
Inside the LSM 880 NLO + Airyscan
 Inside the LSM 880 NLO + Airyscan Overview of the Newest High-End Point Scanning Solution from Carl Zeiss Microscopy Matt Curtis 3D Imaging Specialist John Dirnberger Account Manager Washington University
Inside the LSM 880 NLO + Airyscan Overview of the Newest High-End Point Scanning Solution from Carl Zeiss Microscopy Matt Curtis 3D Imaging Specialist John Dirnberger Account Manager Washington University
1 Co Localization and Working flow with the lsm700
 1 Co Localization and Working flow with the lsm700 Samples -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ BrDU with alexa 488. -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ no BrDU (but with
1 Co Localization and Working flow with the lsm700 Samples -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ BrDU with alexa 488. -1 slide = mousse intestine, Dapi / Ki 67 with Cy3/ no BrDU (but with
Operation Guide for the Leica SP2 Confocal Microscope Bio-Imaging Facility Hunter College October 2009
 Operation Guide for the Leica SP2 Confocal Microscope Bio-Imaging Facility Hunter College October 2009 Introduction of Fluoresence Confocal Microscopy The first confocal microscope was invented by Princeton
Operation Guide for the Leica SP2 Confocal Microscope Bio-Imaging Facility Hunter College October 2009 Introduction of Fluoresence Confocal Microscopy The first confocal microscope was invented by Princeton
Introduction to light microscopy
 Center for Microscopy and Image Anaylsis Introduction to light Basic concepts of imaging with light Urs Ziegler ziegler@zmb.uzh.ch Microscopy with light 1 Light interacting with matter Absorbtion Refraction
Center for Microscopy and Image Anaylsis Introduction to light Basic concepts of imaging with light Urs Ziegler ziegler@zmb.uzh.ch Microscopy with light 1 Light interacting with matter Absorbtion Refraction
Akinori Mitani and Geoff Weiner BGGN 266 Spring 2013 Non-linear optics final report. Introduction and Background
 Akinori Mitani and Geoff Weiner BGGN 266 Spring 2013 Non-linear optics final report Introduction and Background Two-photon microscopy is a type of fluorescence microscopy using two-photon excitation. It
Akinori Mitani and Geoff Weiner BGGN 266 Spring 2013 Non-linear optics final report Introduction and Background Two-photon microscopy is a type of fluorescence microscopy using two-photon excitation. It
Reflecting optical system to increase signal intensity. in confocal microscopy
 Reflecting optical system to increase signal intensity in confocal microscopy DongKyun Kang *, JungWoo Seo, DaeGab Gweon Nano Opto Mechatronics Laboratory, Dept. of Mechanical Engineering, Korea Advanced
Reflecting optical system to increase signal intensity in confocal microscopy DongKyun Kang *, JungWoo Seo, DaeGab Gweon Nano Opto Mechatronics Laboratory, Dept. of Mechanical Engineering, Korea Advanced
Megapixel FLIM with bh TCSPC Modules
 Megapixel FLIM with bh TCSPC Modules The New SPCM 64-bit Software Abstract: Becker & Hickl have recently introduced version 9.60 of their SPCM TCSPC data acquisition software. SPCM version 9.60 not only
Megapixel FLIM with bh TCSPC Modules The New SPCM 64-bit Software Abstract: Becker & Hickl have recently introduced version 9.60 of their SPCM TCSPC data acquisition software. SPCM version 9.60 not only
Nikon. King s College London. Imaging Centre. N-SIM guide NIKON IMAGING KING S COLLEGE LONDON
 N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
N-SIM guide NIKON IMAGING CENTRE @ KING S COLLEGE LONDON Starting-up / Shut-down The NSIM hardware is calibrated after system warm-up occurs. It is recommended that you turn-on the system for at least
Rapid Adaptive Optical Recovery of Optimal Resolution over Large Volumes
 SUPPLEMENTARY MATERIAL Rapid Adaptive Optical Recovery of Optimal Resolution over Large Volumes Kai Wang, Dan Milkie, Ankur Saxena, Peter Engerer, Thomas Misgeld, Marianne E. Bronner, Jeff Mumm, and Eric
SUPPLEMENTARY MATERIAL Rapid Adaptive Optical Recovery of Optimal Resolution over Large Volumes Kai Wang, Dan Milkie, Ankur Saxena, Peter Engerer, Thomas Misgeld, Marianne E. Bronner, Jeff Mumm, and Eric
Confocal imaging on the Leica TCS SP8. 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software:
 Confocal imaging on the Leica TCS SP8 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software: 4) Do not touch the microscope while the software is initializing. Choose your options: Turn
Confocal imaging on the Leica TCS SP8 1) Turn the system on. 2) Use TCS user account. 3) Start LAS X software: 4) Do not touch the microscope while the software is initializing. Choose your options: Turn
Leica TCS SP8 Quick Start Guide
 Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, Fluorescent Light for the microscope stand. 2. Turn on the Scanner Power (1) on the front
Leica TCS SP8 Quick Start Guide Leica TCS SP8 System Overview Start-Up Procedure 1. Turn on the CTR Control Box, Fluorescent Light for the microscope stand. 2. Turn on the Scanner Power (1) on the front
Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each
 Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each lens with a 1 Airy unit pinhole setting. Many water lenses
Supplemental Figure 1: Histogram of 63x Objective Lens z axis Calculated Resolutions. Results from the MetroloJ z axis fits for 5 beads from each lens with a 1 Airy unit pinhole setting. Many water lenses
Zeiss LSM 510 Confocor III Training Notes. Center for Cell Analysis & Modeling
 Zeiss LSM 510 Confocor III Training Notes Center for Cell Analysis & Modeling Confocor 3 Start Up Go to System Module Turn on Main Switch, System/ PC, and Components Switches Do you need the arc lamp?
Zeiss LSM 510 Confocor III Training Notes Center for Cell Analysis & Modeling Confocor 3 Start Up Go to System Module Turn on Main Switch, System/ PC, and Components Switches Do you need the arc lamp?
Advanced Optical Microscopy lecture. 03. December 2012 Kai Wicker
 Advanced Optical Microscopy lecture 03. December 2012 Kai Wicker Today: Optical transfer functions (OTF) and point spread functions (PSF) in incoherent imaging. 1. Quick revision: the incoherent wide-field
Advanced Optical Microscopy lecture 03. December 2012 Kai Wicker Today: Optical transfer functions (OTF) and point spread functions (PSF) in incoherent imaging. 1. Quick revision: the incoherent wide-field
Instant super-resolution imaging in live cells and embryos via analog image processing
 Nature Methods Instant super-resolution imaging in live cells and embryos via analog image processing Andrew G. York, Panagiotis Chandris, Damian Dalle Nogare, Jeffrey Head, Peter Wawrzusin, Robert S.
Nature Methods Instant super-resolution imaging in live cells and embryos via analog image processing Andrew G. York, Panagiotis Chandris, Damian Dalle Nogare, Jeffrey Head, Peter Wawrzusin, Robert S.
The DCS-120 Confocal Scanning FLIM System
 he DCS-120 Confocal Scanning FLIM System he bh DCS-120 confocal scanning FLIM system converts a conventional microscope into a high-performance fluorescence lifetime imaging system. he system is based
he DCS-120 Confocal Scanning FLIM System he bh DCS-120 confocal scanning FLIM system converts a conventional microscope into a high-performance fluorescence lifetime imaging system. he system is based
3D light microscopy techniques
 3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 2D imaging 3D imaging Resolution
3D light microscopy techniques The image of a point is a 3D feature In-focus image Out-of-focus image The image of a point is not a point Point Spread Function (PSF) 1D imaging 2D imaging 3D imaging Resolution
Confocal Microscopy. Kristin Jensen
 Confocal Microscopy Kristin Jensen 17.11.05 References Cell Biological Applications of Confocal Microscopy, Brian Matsumoto, chapter 1 Studying protein dynamics in living cells,, Jennifer Lippincott-Schwartz
Confocal Microscopy Kristin Jensen 17.11.05 References Cell Biological Applications of Confocal Microscopy, Brian Matsumoto, chapter 1 Studying protein dynamics in living cells,, Jennifer Lippincott-Schwartz
LSM 800 Confocal Microscope Standard Operation Protocol
 LSM 800 Confocal Microscope Standard Operation Protocol Turning on the system 1. Switch on the Main switch (labeled 1 and 2 ) mounted on the wall. 2. Turn the Laser Key (labeled 3 ) 90 clockwise for power
LSM 800 Confocal Microscope Standard Operation Protocol Turning on the system 1. Switch on the Main switch (labeled 1 and 2 ) mounted on the wall. 2. Turn the Laser Key (labeled 3 ) 90 clockwise for power
Confocal Laser Scanning Microscopy
 Name of the Core Facility: Confocal Laser Scanning Microscopy CORE Forschungszentrum Immunologie Mainz Welcome to the CSLM Core Facility: The CLSM Core Facility enables working groups to incorporate high
Name of the Core Facility: Confocal Laser Scanning Microscopy CORE Forschungszentrum Immunologie Mainz Welcome to the CSLM Core Facility: The CLSM Core Facility enables working groups to incorporate high
LSM 710 Confocal Microscope Standard Operation Protocol
 LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
LSM 710 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Switch on Main power switch 2. Switch on System / PC power button 3. Switch on Components power button 4.
ADVANCED METHODS FOR CONFOCAL MICROSCOPY II. Jean-Yves Chatton Sept. 2006
 ADVANCED METHODS FOR CONFOCAL MICROSCOPY II Jean-Yves Chatton Sept. 2006 Workshop outline Confocal microscopy of living cells and tissues X-Z scanning Time series Bleach: FRAP, photoactivation Emission
ADVANCED METHODS FOR CONFOCAL MICROSCOPY II Jean-Yves Chatton Sept. 2006 Workshop outline Confocal microscopy of living cells and tissues X-Z scanning Time series Bleach: FRAP, photoactivation Emission
5/4/2015 INTRODUCTION TO LIGHT MICROSCOPY. Urs Ziegler MICROSCOPY WITH LIGHT. Image formation in a nutshell. Overview of techniques
 INTRODUCTION TO LIGHT MICROSCOPY Urs Ziegler ziegler@zmb.uzh.ch MICROSCOPY WITH LIGHT INTRODUCTION TO LIGHT MICROSCOPY Image formation in a nutshell Overview of techniques Widefield microscopy Resolution
INTRODUCTION TO LIGHT MICROSCOPY Urs Ziegler ziegler@zmb.uzh.ch MICROSCOPY WITH LIGHT INTRODUCTION TO LIGHT MICROSCOPY Image formation in a nutshell Overview of techniques Widefield microscopy Resolution
Microscopy. Matti Hotokka Department of Physical Chemistry Åbo Akademi University
 Microscopy Matti Hotokka Department of Physical Chemistry Åbo Akademi University What s coming Anatomy of a microscope Modes of illumination Practicalities Special applications Basic microscope Ocular
Microscopy Matti Hotokka Department of Physical Chemistry Åbo Akademi University What s coming Anatomy of a microscope Modes of illumination Practicalities Special applications Basic microscope Ocular
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement
 Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
Boulevard du Temple Daguerrotype (Paris,1838) a busy street? Nyquist sampling for movement CONFOCAL MICROSCOPY BioVis Uppsala, 2017 Jeremy Adler Matyas Molnar Dirk Pacholsky Widefield & Confocal Microscopy
More fancy SPIM, Even fancier SPIM
 More fancy SPIM, Even fancier SPIM Last class Light sheet microscopy Fancy SPIM (ispim, dspim, etc ) This class Multi camera SPIM SIM SPIM Bessels d x,y = λ em 2 NA d z = 2 NA λ ex + n(1 cosθ λ em 1 IsoView
More fancy SPIM, Even fancier SPIM Last class Light sheet microscopy Fancy SPIM (ispim, dspim, etc ) This class Multi camera SPIM SIM SPIM Bessels d x,y = λ em 2 NA d z = 2 NA λ ex + n(1 cosθ λ em 1 IsoView
Confocal Microscopy Confocal Microscopy Acousto-optic products
 Confocal Microscopy Confocal Microscopy Acousto-optic products AA OPTO-ELECTRONIC QUANTA TECH Introduction Confocal microscopy is an imaging technique used to increase micrograph contrast and/or to reconstruct
Confocal Microscopy Confocal Microscopy Acousto-optic products AA OPTO-ELECTRONIC QUANTA TECH Introduction Confocal microscopy is an imaging technique used to increase micrograph contrast and/or to reconstruct
Zeiss LSM880 Operating Instructions. UTMB Optical Microscopy Core Jan. 16, 2018
 Zeiss LSM880 Operating Instructions UTMB Optical Microscopy Core Jan. 16, 2018 1 1. Power up the microscope Sing the LOGBOOK Steps below will provide power to the computer and all of the microscope components.
Zeiss LSM880 Operating Instructions UTMB Optical Microscopy Core Jan. 16, 2018 1 1. Power up the microscope Sing the LOGBOOK Steps below will provide power to the computer and all of the microscope components.
NIH Public Access Author Manuscript Opt Lett. Author manuscript; available in PMC 2010 August 9.
 NIH Public Access Author Manuscript Published in final edited form as: Opt Lett. 2010 January 1; 35(1): 67 69. Autoconfocal transmission microscopy based on two-photon induced photocurrent of Si photodiodes
NIH Public Access Author Manuscript Published in final edited form as: Opt Lett. 2010 January 1; 35(1): 67 69. Autoconfocal transmission microscopy based on two-photon induced photocurrent of Si photodiodes
Confocal Imaging Through Scattering Media with a Volume Holographic Filter
 Confocal Imaging Through Scattering Media with a Volume Holographic Filter Michal Balberg +, George Barbastathis*, Sergio Fantini % and David J. Brady University of Illinois at Urbana-Champaign, Urbana,
Confocal Imaging Through Scattering Media with a Volume Holographic Filter Michal Balberg +, George Barbastathis*, Sergio Fantini % and David J. Brady University of Illinois at Urbana-Champaign, Urbana,
Introduction to light microscopy
 Center for Microscopy and Image Anaylsis Introduction to light microscopy (an overview) Microscopy with light Components of a light microscope 1. Light source 2. Objective 3. Sample or specimen holder
Center for Microscopy and Image Anaylsis Introduction to light microscopy (an overview) Microscopy with light Components of a light microscope 1. Light source 2. Objective 3. Sample or specimen holder
You won t be able to measure the incident power precisely. The readout of the power would be lower than the real incident power.
 1. a) Given the transfer function of a detector (below), label and describe these terms: i. dynamic range ii. linear dynamic range iii. sensitivity iv. responsivity b) Imagine you are using an optical
1. a) Given the transfer function of a detector (below), label and describe these terms: i. dynamic range ii. linear dynamic range iii. sensitivity iv. responsivity b) Imagine you are using an optical
Nature Methods: doi: /nmeth Supplementary Figure 1. Schematic of 2P-ISIM AO optical setup.
 Supplementary Figure 1 Schematic of 2P-ISIM AO optical setup. Excitation from a femtosecond laser is passed through intensity control and shuttering optics (1/2 λ wave plate, polarizing beam splitting
Supplementary Figure 1 Schematic of 2P-ISIM AO optical setup. Excitation from a femtosecond laser is passed through intensity control and shuttering optics (1/2 λ wave plate, polarizing beam splitting
CCAM s Selection of. Zeiss Microscope Objectives
 CCAM s Selection of Zeiss Microscope Objectives 1. Magnification Image scale 2. Resolution The minimum separation distance between two points that are clearly resolved. The resolution of an objective is
CCAM s Selection of Zeiss Microscope Objectives 1. Magnification Image scale 2. Resolution The minimum separation distance between two points that are clearly resolved. The resolution of an objective is
Fast, high-contrast imaging of animal development with scanned light sheet based structured-illumination microscopy
 nature methods Fast, high-contrast imaging of animal development with scanned light sheet based structured-illumination microscopy Philipp J Keller, Annette D Schmidt, Anthony Santella, Khaled Khairy,
nature methods Fast, high-contrast imaging of animal development with scanned light sheet based structured-illumination microscopy Philipp J Keller, Annette D Schmidt, Anthony Santella, Khaled Khairy,
DIC Imaging using Laser Scanning Microscopes (LSMs) on Axio Imager Stands
 DIC Imaging using Laser Scanning Microscopes (LSMs) on Axio Imager Stands Differential Interference Contrast (DIC) imaging is a technique used to increase contrast in brightfield images. In confocal systems,
DIC Imaging using Laser Scanning Microscopes (LSMs) on Axio Imager Stands Differential Interference Contrast (DIC) imaging is a technique used to increase contrast in brightfield images. In confocal systems,
PET Detectors. William W. Moses Lawrence Berkeley National Laboratory March 26, 2002
 PET Detectors William W. Moses Lawrence Berkeley National Laboratory March 26, 2002 Step 1: Inject Patient with Radioactive Drug Drug is labeled with positron (β + ) emitting radionuclide. Drug localizes
PET Detectors William W. Moses Lawrence Berkeley National Laboratory March 26, 2002 Step 1: Inject Patient with Radioactive Drug Drug is labeled with positron (β + ) emitting radionuclide. Drug localizes
Spectral Imaging with the Opterra Multipoint Scanning Confocal
 Spectral Imaging with the Opterra Multipoint Scanning Confocal Outline Opterra design overview Scan Modes Light Path Spectral Imaging with Opterra Drosophila larva heart. Opterra Design Overview Supravideo
Spectral Imaging with the Opterra Multipoint Scanning Confocal Outline Opterra design overview Scan Modes Light Path Spectral Imaging with Opterra Drosophila larva heart. Opterra Design Overview Supravideo
Development of Laser Confocal Microscopy for Internal Defect Measurement
 Development of Laser Confocal Microscopy for Internal Defect Measurement Chia-Liang Yeh*, Fu-Cheng Yang, Wei-Hsiung Tsai, and Keng-Li Lin Center for Measurement Standards, Industrial Technology Research
Development of Laser Confocal Microscopy for Internal Defect Measurement Chia-Liang Yeh*, Fu-Cheng Yang, Wei-Hsiung Tsai, and Keng-Li Lin Center for Measurement Standards, Industrial Technology Research
Introduction to light microscopy
 Center for Microscopy and Image Anaylsis Introduction to light Imaging with light / Overview of techniques Urs Ziegler ziegler@zmb.uzh.ch Light interacting with matter Absorbtion Refraction Diffraction
Center for Microscopy and Image Anaylsis Introduction to light Imaging with light / Overview of techniques Urs Ziegler ziegler@zmb.uzh.ch Light interacting with matter Absorbtion Refraction Diffraction
IC 2 S High Performance Objectives
 M i c r o s c o p y f r o m C a r l Z e i s s IC 2 S igh Performance Objectives for Biomedical Applications with Laser Based Imaging Systems LSM,, ConfoCor, TIRF and ELYRA Carl Zeiss offers a large range
M i c r o s c o p y f r o m C a r l Z e i s s IC 2 S igh Performance Objectives for Biomedical Applications with Laser Based Imaging Systems LSM,, ConfoCor, TIRF and ELYRA Carl Zeiss offers a large range
Confocal, Airyscan and Structured Illumination Superresolution microscopy Mayandi Sivaguru
 Confocal, Airyscan and Structured Illumination Superresolution microscopy Mayandi Sivaguru Theory, Light Path, Resolution Comparison Performance comparison Why and When Choose Airyscan OR SR-SIM Superresolution
Confocal, Airyscan and Structured Illumination Superresolution microscopy Mayandi Sivaguru Theory, Light Path, Resolution Comparison Performance comparison Why and When Choose Airyscan OR SR-SIM Superresolution
Confocal Microscopy and Related Techniques
 Confocal Microscopy and Related Techniques Chau-Hwang Lee Associate Research Fellow Research Center for Applied Sciences, Academia Sinica 128 Sec. 2, Academia Rd., Nankang, Taipei 11529, Taiwan E-mail:
Confocal Microscopy and Related Techniques Chau-Hwang Lee Associate Research Fellow Research Center for Applied Sciences, Academia Sinica 128 Sec. 2, Academia Rd., Nankang, Taipei 11529, Taiwan E-mail:
ANSWER KEY Lab 2 (IGB): Bright Field and Fluorescence Optical Microscopy and Sectioning
 Phys598BP Spring 2016 University of Illinois at Urbana-Champaign ANSWER KEY Lab 2 (IGB): Bright Field and Fluorescence Optical Microscopy and Sectioning Location: IGB Core Microscopy Facility Microscope:
Phys598BP Spring 2016 University of Illinois at Urbana-Champaign ANSWER KEY Lab 2 (IGB): Bright Field and Fluorescence Optical Microscopy and Sectioning Location: IGB Core Microscopy Facility Microscope:
Microscopy from Carl Zeiss
 Microscopy from Carl Zeiss Contents Page Contents... 1 Introduction... 1 Starting the System... 2 Introduction to ZEN Efficient Navigation... 5 Setting up the microscope... 10 Configuring the beam path
Microscopy from Carl Zeiss Contents Page Contents... 1 Introduction... 1 Starting the System... 2 Introduction to ZEN Efficient Navigation... 5 Setting up the microscope... 10 Configuring the beam path
Multicolor 4D Fluorescence Microscopy using Ultrathin Bessel Light sheets
 SUPPLEMENTARY MATERIAL Multicolor 4D Fluorescence Microscopy using Ultrathin Bessel Light sheets Teng Zhao, Sze Cheung Lau, Ying Wang, Yumian Su, Hao Wang, Aifang Cheng, Karl Herrup, Nancy Y. Ip, Shengwang
SUPPLEMENTARY MATERIAL Multicolor 4D Fluorescence Microscopy using Ultrathin Bessel Light sheets Teng Zhao, Sze Cheung Lau, Ying Wang, Yumian Su, Hao Wang, Aifang Cheng, Karl Herrup, Nancy Y. Ip, Shengwang
Operating Instructions for Zeiss LSM 510
 Operating Instructions for Zeiss LSM 510 Location: GNL 6.312q (BSL3) Questions? Contact: Maxim Ivannikov, maivanni@utmb.edu 1 Attend A Complementary Training Before Using The Microscope All future users
Operating Instructions for Zeiss LSM 510 Location: GNL 6.312q (BSL3) Questions? Contact: Maxim Ivannikov, maivanni@utmb.edu 1 Attend A Complementary Training Before Using The Microscope All future users
Leica SP8 TCS Users Manual
 Leica SP8 TCS Users Manual Follow the procedure for start up and log on as posted in the lab. Please log on with your account only and do not share your password with anyone. We track and confirm usage
Leica SP8 TCS Users Manual Follow the procedure for start up and log on as posted in the lab. Please log on with your account only and do not share your password with anyone. We track and confirm usage
Multidimensional Imaging with the Opterra Multipoint Scanning Confocal System
 Multidimensional Imaging with the Opterra Multipoint Scanning Confocal System Outline Opterra design overview Light Path Scan Modes Performance Application data Drosophila larva heart. Courtesy of Nikon
Multidimensional Imaging with the Opterra Multipoint Scanning Confocal System Outline Opterra design overview Light Path Scan Modes Performance Application data Drosophila larva heart. Courtesy of Nikon
CCAM Microscope Objectives
 CCAM Microscope Objectives Things to consider when selecting an objective Magnification Numerical Aperture (NA) resolving power and light intensity of the objective Working Distance distance between the
CCAM Microscope Objectives Things to consider when selecting an objective Magnification Numerical Aperture (NA) resolving power and light intensity of the objective Working Distance distance between the
TRAINING MANUAL. Multiphoton Microscopy LSM 510 META-NLO
 TRAINING MANUAL Multiphoton Microscopy LSM 510 META-NLO September 2010 Multiphoton Microscopy Training Manual Multiphoton microscopy is only available on the LSM 510 META-NLO system. This system is equipped
TRAINING MANUAL Multiphoton Microscopy LSM 510 META-NLO September 2010 Multiphoton Microscopy Training Manual Multiphoton microscopy is only available on the LSM 510 META-NLO system. This system is equipped
Supplemental Method Information Zeiss LSM710
 Supplemental Method Information Zeiss LSM710 1 Under the Light Path window set up the confocal for imaging a green dye (Alexa488-EGFP). For example, set up the light path as shown here using the 488 nm
Supplemental Method Information Zeiss LSM710 1 Under the Light Path window set up the confocal for imaging a green dye (Alexa488-EGFP). For example, set up the light path as shown here using the 488 nm
Microscopic Structures
 Microscopic Structures Image Analysis Metal, 3D Image (Red-Green) The microscopic methods range from dark field / bright field microscopy through polarisation- and inverse microscopy to techniques like
Microscopic Structures Image Analysis Metal, 3D Image (Red-Green) The microscopic methods range from dark field / bright field microscopy through polarisation- and inverse microscopy to techniques like
Fundamentals of Light Microscopy II: Fluorescence, Deconvolution, Confocal, Multiphoton, Spectral microscopy. Integrated Microscopy Course
 Fundamentals of Light Microscopy II: Fluorescence, Deconvolution, Confocal, Multiphoton, Spectral microscopy Integrated Microscopy Course Review Lecture 1: Microscopy Basics Light train Kohler illumination*
Fundamentals of Light Microscopy II: Fluorescence, Deconvolution, Confocal, Multiphoton, Spectral microscopy Integrated Microscopy Course Review Lecture 1: Microscopy Basics Light train Kohler illumination*
Nikon Instruments Europe
 Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
Nikon Instruments Europe Recommendations for N-SIM sample preparation and image reconstruction Dear customer, We hope you find the following guidelines useful in order to get the best performance out of
Education in Microscopy and Digital Imaging
 Contact Us Carl Zeiss Education in Microscopy and Digital Imaging ZEISS Home Products Solutions Support Online Shop ZEISS International ZEISS Campus Home Interactive Tutorials Basic Microscopy Spectral
Contact Us Carl Zeiss Education in Microscopy and Digital Imaging ZEISS Home Products Solutions Support Online Shop ZEISS International ZEISS Campus Home Interactive Tutorials Basic Microscopy Spectral
Leica SPEII confocal microscope. Short Manual
 Leica SPEII confocal microscope Short Manual Switching ON sequence: 1. Turn on the Workstation under the bench (top, far right). 2. Turn on the Supply Unit - Laser box (big green switch first and then
Leica SPEII confocal microscope Short Manual Switching ON sequence: 1. Turn on the Workstation under the bench (top, far right). 2. Turn on the Supply Unit - Laser box (big green switch first and then
Resolution. Diffraction from apertures limits resolution. Rayleigh criterion θ Rayleigh = 1.22 λ/d 1 peak at 2 nd minimum. θ f D
 Microscopy Outline 1. Resolution and Simple Optical Microscope 2. Contrast enhancement: Dark field, Fluorescence (Chelsea & Peter), Phase Contrast, DIC 3. Newer Methods: Scanning Tunneling microscopy (STM),
Microscopy Outline 1. Resolution and Simple Optical Microscope 2. Contrast enhancement: Dark field, Fluorescence (Chelsea & Peter), Phase Contrast, DIC 3. Newer Methods: Scanning Tunneling microscopy (STM),
MULTIPHOTON MICROSCOPY. Matyas Molnar Dirk Pacholsky
 MULTIPHOTON MICROSCOPY Matyas Molnar Dirk Pacholsky Information Information given here about 2 Photon microscopy were mainly taken from these sources: Background information on 2-Photon microscopy: http://micro.magnet.fsu.edu/primer/techniques/fluorescence/multiphoton/
MULTIPHOTON MICROSCOPY Matyas Molnar Dirk Pacholsky Information Information given here about 2 Photon microscopy were mainly taken from these sources: Background information on 2-Photon microscopy: http://micro.magnet.fsu.edu/primer/techniques/fluorescence/multiphoton/
VivaTome. Discover the Dynamics of Life. The Entry-level System that Captures Dynamic Processes with Outstanding Image Quality.
 Microscopy from Carl Zeiss VivaTome Discover the Dynamics of Life The Entry-level System that Captures Dynamic Processes with Outstanding Image Quality. Innovative Technology Captures Dynamic Processes
Microscopy from Carl Zeiss VivaTome Discover the Dynamics of Life The Entry-level System that Captures Dynamic Processes with Outstanding Image Quality. Innovative Technology Captures Dynamic Processes
Final Exam, 150 points PMB 185: Techniques in Light Microscopy
 Final Exam, 150 points Name PMB 185: Techniques in Light Microscopy Point value is in parentheses at the end of each question. Note: GFP = green fluorescent protein ; CFP = cyan fluorescent protein ; YFP
Final Exam, 150 points Name PMB 185: Techniques in Light Microscopy Point value is in parentheses at the end of each question. Note: GFP = green fluorescent protein ; CFP = cyan fluorescent protein ; YFP
LSM 780 Confocal Microscope Standard Operation Protocol
 LSM 780 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Sign on log sheet according to Actual start time 2. Check Compressed Air supply for the air table 3. Switch
LSM 780 Confocal Microscope Standard Operation Protocol Basic Operation Turning on the system 1. Sign on log sheet according to Actual start time 2. Check Compressed Air supply for the air table 3. Switch
3 Choose the Channels button and set the Channel Settings. Set the Pinhole to 1 Airy unit.
 1 Set up the confocal light path for imaging a green dye (e.g. Alexa488-EGFP). For example, under the Configuration Control window the light path could be set up as shown here using the 488 nm LASER (found
1 Set up the confocal light path for imaging a green dye (e.g. Alexa488-EGFP). For example, under the Configuration Control window the light path could be set up as shown here using the 488 nm LASER (found
1 Set up the confocal light path for imaging a green dye (Alexa488-EGFP). For example, the
 1 Set up the confocal light path for imaging a green dye (Alexa488-EGFP). For example, the light path as shown here using the 488 nm LASER (Laser Unit 1) reflecting off of the 405/488 nm Dichroic mirror
1 Set up the confocal light path for imaging a green dye (Alexa488-EGFP). For example, the light path as shown here using the 488 nm LASER (Laser Unit 1) reflecting off of the 405/488 nm Dichroic mirror
BIOIMAGING AND OPTICS PLATFORM EPFL SV PTBIOP LASER SCANNING CONFOCAL MICROSCOPY PRACTICAL CONSIDERATIONS
 LASER SCANNING CONFOCAL MICROSCOPY PRACTICAL CONSIDERATIONS IMPORTANT PARAMETERS Pixel dwell time Zoom and pixel number PIXEL DWELL TIME How much time signal is collected at every pixel Very small values,
LASER SCANNING CONFOCAL MICROSCOPY PRACTICAL CONSIDERATIONS IMPORTANT PARAMETERS Pixel dwell time Zoom and pixel number PIXEL DWELL TIME How much time signal is collected at every pixel Very small values,
ZEISS LSM510META confocal manual
 ZEISS LSM510META confocal manual Switching on the system 1) Switch on the Remote Control button located on the table to the right of the microscope. This is the main switch for the whole system including
ZEISS LSM510META confocal manual Switching on the system 1) Switch on the Remote Control button located on the table to the right of the microscope. This is the main switch for the whole system including
TCSPC at Wavelengths from 900 nm to 1700 nm
 TCSPC at Wavelengths from 900 nm to 1700 nm We describe picosecond time-resolved optical signal recording in the spectral range from 900 nm to 1700 nm. The system consists of an id Quantique id220 InGaAs
TCSPC at Wavelengths from 900 nm to 1700 nm We describe picosecond time-resolved optical signal recording in the spectral range from 900 nm to 1700 nm. The system consists of an id Quantique id220 InGaAs
長庚大學共軛焦顯微鏡課程 長庚大學共軛焦顯微鏡課程. Spot light 長庚大學
 長庚大學共軛焦顯微鏡課程 Spot light 長庚大學共軛焦顯微鏡課程 20071030 長庚大學 Basic principle of Laser Scanning Confocal Microscopy The application of LSM 510 META detector Multiphoton microscopy basic principle and introduction
長庚大學共軛焦顯微鏡課程 Spot light 長庚大學共軛焦顯微鏡課程 20071030 長庚大學 Basic principle of Laser Scanning Confocal Microscopy The application of LSM 510 META detector Multiphoton microscopy basic principle and introduction
Axio Zoom.V16 The Fluorescence Zoom Microscope for Large Fields
 Product Information Interactive PDF internet-link video/animation Release 1.0 It s About Brilliance. Because Only the Best Is Good Enough In Brief The Advantages The Applications In 1994, the molecular
Product Information Interactive PDF internet-link video/animation Release 1.0 It s About Brilliance. Because Only the Best Is Good Enough In Brief The Advantages The Applications In 1994, the molecular
DIC Imaging using Laser Scanning Microscopes (LSM) on Inverted Stands
 DIC Imaging using Laser Scanning Microscopes (LSM) on Inverted Stands Differential Interference Contrast (DIC) imaging is a technique used to increase contrast in brightfield images. In confocal systems,
DIC Imaging using Laser Scanning Microscopes (LSM) on Inverted Stands Differential Interference Contrast (DIC) imaging is a technique used to increase contrast in brightfield images. In confocal systems,
Sensitive measurement of partial coherence using a pinhole array
 1.3 Sensitive measurement of partial coherence using a pinhole array Paul Petruck 1, Rainer Riesenberg 1, Richard Kowarschik 2 1 Institute of Photonic Technology, Albert-Einstein-Strasse 9, 07747 Jena,
1.3 Sensitive measurement of partial coherence using a pinhole array Paul Petruck 1, Rainer Riesenberg 1, Richard Kowarschik 2 1 Institute of Photonic Technology, Albert-Einstein-Strasse 9, 07747 Jena,
Tissue Preparation ORGANISM IMAGE TISSUE PREPARATION. 1) Fixation: halts cell metabolism, preserves cell/tissue structure
 Lab starts this week! ANNOUNCEMENTS - Tuesday or Wednesday 1:25 ISB 264 - Read Lab 1: Microscopy and Imaging (see Web Page) - Getting started on Lab Group project - Organ for investigation - Lab project
Lab starts this week! ANNOUNCEMENTS - Tuesday or Wednesday 1:25 ISB 264 - Read Lab 1: Microscopy and Imaging (see Web Page) - Getting started on Lab Group project - Organ for investigation - Lab project
FLUORESCENCE MICROSCOPY. Matyas Molnar and Dirk Pacholsky
 FLUORESCENCE MICROSCOPY Matyas Molnar and Dirk Pacholsky 1 The human eye perceives app. 400-700 nm; best at around 500 nm (green) Has a general resolution down to150-300 μm (human hair: 40-250 μm) We need
FLUORESCENCE MICROSCOPY Matyas Molnar and Dirk Pacholsky 1 The human eye perceives app. 400-700 nm; best at around 500 nm (green) Has a general resolution down to150-300 μm (human hair: 40-250 μm) We need
