Palladium-Catalyzed Benzo[d]isoxazole Synthesis by C-H Activation/[4+1]Annulation
|
|
|
- Tyrone Hardy
- 5 years ago
- Views:
Transcription
1 Palladium-Catalyzed Benzo[d]isoxazole Synthesis by C-H Activation/[4+1]Annulation Pingping Duan, a Yunfang Yang, a Xinhao Zhang, a Rong Ben, b Yiyong Yan, a Lu Dai, a Mei Hong, a Dongqi Wang,* a Yun-Dong Wu* a and Jing Zhao* a,b Supporting Information Table of contents 1. General Informations S2 2. Synthesis and Characterization of Starting Materials S2-S5 3. Condition Screening S5-S7 4. Experimental Procedure and Characterization of Products S7-S17 5. X-ray Crystallographic Data for 3-(p-tolyl)benzisoxazole S17-S26 6. Mechanistic Studies S27-S31 7. DFT Calculations S31-S52 8. The Synthesis and Characterization of Key Intermediate 6 S52-S53 9. References S H MR and 13 C MR Spectra of Those Compounds S54-S139
2 1. General Informations All reactions were carried out under an atmosphere of nitrogen unless otherwise noted. Reaction temperatures were reported as those of the oil bath. The dry solvents used were purified by distillation and were transferred under nitrogen. Commercially available reagents were purchased from Adamas-beta, Sigma-Aldrich, Alfa Aesar, TCI, Accela, J&K and Aladdin and used as received unless otherwise stated. Palladium (II) trifluoroacetate was purchased from Sinocompound Technology Co., Ltd. Reactions were monitored with analytical thin-layer chromatography (TLC) on silica. 1 H MR and 13 C MR data were recorded on Bruker nuclear resonance (400 MHz and 500MHz) spectrometers, respectively. Chemical shifts (δ) are given in ppm relative to TMS. The residual solvent signals were used as references and the chemical shifts converted to the TMS scale (CDCl 3 : δ H =7.26 ppm, δ c =77.16 ppm; DMS-d 6 : δ H = 2.50 ppm, δ C = ppm; MeD-d 4 : δ H =3.31 ppm, δ c =49.00 ppm; Acetone-d 6 : δ H = 2.05 ppm, δ C = ppm, ppm). HRMS (ESI and APCI) analysis were performed by the Analytical Instrumentation Center at Peking University, Shenzhen Graduate School and (HRMS) data were reported with ion mass/charge (m/z) ratios as values in atomic mass units. All melting points were uncorrected. 2. Synthesis and Characterization of Starting Materials General procedure for preparation of substrates 1a-1j R B(H)2 + H CuCl Pyridine 4A sieves 1,2-Dichloroethane rt R H 2 H 2 MeH/CHCl 3, rt R H2 Acetic anhydride Et 2, 0 C to rt R H Following a literature report, 1 in a 50 ml round-bottom flask, -hydroxyphthalimide (1.0 eq), cooper (I) chloride (1.0 eq), freshly activated 4 Å molecular sieves (250mg/mmol), and phenylboronic acid (2.0 eq) were combined in 1,2-dichloroethane (0.2 M). The pyridine (1.1 eq) was then added to the suspension. The reaction mixture was open to the atmosphere and stirred at room temperature over 24-48h. Upon completion, silica gel was added to the flask and the solvent was removed under vacuum. The desired -aryloxyphthalimides were obtained by flash column chromatography on silica gel. Hydrazine monohydrate (3.0 eq) was added to the solution of -aryloxyphthalimide (1.0 eq) in 10% MeH in CHCl 3 (0.1 M). The reaction was allowed to stir at room temperature overnight. Upon completion, the reaction mixture was filtered off and washed with CH 2 Cl 2. The filtrate was concentrated under reduced pressure, and purified by flash silica gel column chromatography to S-2 1a-1j
3 give the corresponding -aryloxyamine. In a 20 ml round-bottom flask, -aryloxyamine (1.0 eq) was dissolved in ether (0.2 M).The flask was cooled in an ice bath, to which acetic anhydride (2.0 eq) was slowly added. The ice bath was allowed to warm to room temperature and the mixture was stirred for 3 h at room temperature. The reaction mixture was concentrated under reduced pressure and purified by flash silica gel column chromatography to give the corresponding -aryloxyacetamide. Characterization of Substrates 1 HAc 1a, white solid, yield: 60% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.32 (t, J = 7.9 Hz, 2H), 7.01 (t, J = 7.6 Hz, 3H), 1.92 (s, 3H); 13 C MR (101 MHz, DMS-d 6 ): δ , , , , , IR (cm -1 ): 3107, 2907, 1645, 1539, 1506, 743, 689. HRMS (ESI) calculated for C 8 H 9 2 a (+): ; Found: Melting Point: o C. HAc Me 1b, white solid, yield: 56% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.15 (t, J = 7.7 Hz, 2H), 6.99 (d, J = 8.0 Hz, 1H), 6.92 (t, J = 7.3 Hz, 1H), 2.21 (s, 3H), 1.92 (s, 3H); 13 C MR (101MHz, DMS-d 6 ): δ , , , , , , , 19.44, IR (cm -1 ): 3177,2984,2808,1653, 1506, 1456, 1119, 991, 750. HRMS (ESI) calculated for C 9 H 11 2 a (+): ; Found: Melting Point: HAc Me 1c, white solid, yield: 50% 1 H MR (400 MHz, Acetone-d 6 ): δ (s, 1H), 7.16 (t, J = 7.0 Hz, 1H), 6.85 (d, J = 14.0 Hz, 3H), 2.29 (s, 3H), 1.97 (s, 3H); 13 C MR (101 MHz, Acetone-d 6 ): δ , , , , , , , 20.54, HRMS (ESI) calculated for C 9 H 11 2 a (+): ; Found: S-3
4 HAc Me 1d, white solid, yield: 45% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.10 (d, J = 8.0 Hz, 2H), 6.90 (d, J = 7.9 Hz, 2H), 2.24 (s, 3H), 1.90 (s, 3H); 13 C MR (101 MHz, DMS-d 6 ): δ , , , , , 20.52, 19.84; HRMS (ESI) calculated for C 9 H 11 2 a (+): ; Found: Melting Point: HAc Me 1e, brick-red solid, yield: 50% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.20 (t, J = 8.1 Hz, 1H), (m, 3H), 3.73 (s, 3H), 1.90 (s, 3H); 13 C MR (101 MHz, DMS-d 6 ): δ , , , , , , 99.59, 55.68, 19.83; IR (cm -1 ): 3177, 2957, 1684, 1607, 1489, 1134, 685; HRMS (ESI) calculated for C 9 H 11 3 a(+): ; Found: Melting Point: HAc F 1f, white solid, yield: 35% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.34 (dd, J = 15.4, 8.0 Hz, 1H), 6.85 (dd, J = 15.3, 9.9 Hz, 3H), 1.92 (s, 3H); 13 C MR (101MHz, DMS-d 6 ): δ , (J 1F = 244.5), (J 3F = 10.6), (J 3F = 10.2), , (J 2F = 21.4), (J 2F = 26.7), HRMS (ESI) calculated for C 8 H 8 2 Fa(+): ; Found: Melting Point: HAc Cl 1g, white solid, yield: 40% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.33 (t, J = 8.3 Hz, 1H), 7.08 (m, 2H), 6.99 (d, J = 8.1 Hz, 1H), 1.92 (s, 3H); 13 C MR (101 MHz, DMS-d 6 ): δ , , , , , , , HRMS (ESI) calculated for C 8 H 8 2 acl(+): ; Found: Melting Point: S-4
5 HAc Br 1h, white solid, yield: 30% 1H MR (400 MHz, DMS-d6): δ (s, 1H), 7.27 (t, J = 8.2 Hz, 1H), 7.21 (d, J = 5.4 Hz, 2H), 7.03 (d, J = 8.1 Hz, 1H), 1.92 (s, 3H); 13 C MR (101 MHz, DMS-d6):δ167.91, , , , , , , IR (cm -1 ): 3102, 2911, 1653, 1506, 991, 779. HRMS (ESI) calculated for C 8 H 8 2 abr(+): ; Found: Melting Point: o C. HAc Cl 1i, white solid, yield: 45% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.35 (d, J = 8.8 Hz, 2H), 7.03 (d, J = 8.6 Hz, 2H), 1.91 (s, 3H); 13 C MR (101 MHz, DMS-d 6 ): δ , , , , , IR (cm -1 ): 3109, 2911, 1663, 1506, 989, 824. HRMS (ESI) calculated for C 8 H 8 2 acl(+): ; Found: Melting Point: HAc Br 1j, white solid, yield: 35% 1 H MR (400 MHz, DMS-d 6 ): δ (s, 1H), 7.47 (d, J = 8.7 Hz, 2H), 6.98 (d, J = 8.5 Hz, 2H), 1.91 (s, 3H); 13 C MR (101 MHz, DMS-d 6 ): δ , , , , , HRMS (ESI) calculated for C 8 H 8 2 abr(+): ; Found: Melting Point: Condition Sreening Initial screening quickly identified Pd(TFA) 2 was a good catalyst and this reaction required oxidants. Table S1. Condition Screening a,b S-5
6 HAc + H 10 mol% Pd(TFA)2 20 mol% Ligand xidant 0.5 eq. Additive Solvent, T, 20h 2 Entry Solvent Ligand xidant Additiv e Temperat ure Yield 1 toluene pyridine 4 eq TBHP - 80.R. 2 toluene bpy 4 eq TBHP - 80.R. 3 toluene Cyclohexyl 4 eq TBHP % JohnPhos 4 toluene JohnPhos 4 eq TBHP % 5 toluene DavePhos 4 eq TBHP % 6 toluene - 4 eq - 80.R. Ag 2 C 3 7 toluene - 4 eq AgAc - 80.R. 8 toluene - 4 eq Ag 2-80.R. 9 toluene - 4 eq K 2 S R. 10 toluene - 4 eq - 80.R. Cu(Ac) 2 11 DCE - 4 eq TBHP % 12 THF - 4 eq TBHP % 13 1,4-dioxane - 4 eq TBHP % 14 MeH - 4 eq TBHP % 15 CH 3 C - 4 eq TBHP % 16 DMS - 4 eq TBHP % 17 xylene 4 eq TBHP 80 37% 18 t-amh - 4 eq TBHP % 19 t-buh - 4 eq TBHP % 20 PhCl - 4 eq TBHP - 80.R. 21 t-amh Cyclohexyl 4 eq TBHP 80 48% JohnPhos 22 t-amh - 4 eq TBHP PivH 80 26% 23 t-amh - 4 eq TBHP Cs 2 C 80.R. 24 t-amh - 4 eq TBHP CsAc 80.R. 25 t-amh - 4 eq TBHP K 2 C 3 80.R. 26 t-amh - 4 eq TBHP - r.t 42% 27 t-amh - 4 eq TBHP % 28 t-amh - 4 eq TBHP % 29 t-amh - 4 eq TBHP % S-6 3
7 30 t-amh - 4 eq TBHP % 31 c t-amh - 4 eq TBHP % 32 t-amh - 4 eq TBHP % 33 t-amh - 4 eq TBHP % 34 t-amh - 4 eq TBHP % 35 d t-amh - 4 eq TBHP % 36 e t-amh - 4 eq TBHP % 37 f t-amh - 4 eq TBHP % 38 g t-amh - 4 eq TBHP % 39 t-amh R. 40 t-amh - 1 eq TBHP % 41 t-amh eq TBHP % 42 t-amh - 2 eq TBHP % 43 t-amh eq TBHP % 44 t-amh - 3 eq TBHP % 45 t-amh eq TBHP % 46 t-amh eq DTBP - 60.R. 47 t-amh Ph 3 P 2.5 eq TBHP % 48 t-amh Ph 3 P= 2.5 eq TBHP % a GC yield using mesitylene as an internal standard. b All reactions were kept in dark place. c solated yield. d reaction under air. e reaction was not kept in dark place. f Pd(Ac) 2 instead of Pd(TFA) 2.. g 5%Pd(TFA) 2. ote: Di-tert-butyl peroxide (DTBP). P P P Me 2 Cyclohexyl JohnPhos JohnPhos DavePhos L1 L2 4. Experimental Procedure and Characterization of Products R 1 HAc + R 2 H 2 equiv 10 mol% Pd(TFA)2 2.5 equiv TBHP t-amh, 60 C 20h, 2 R1 R 2 -Phenoxyacetamides substrates (1) (0.5 mmol) and Pd(TFA) 2 (10 mol%) were weighed into a 25ml pressure tube, to which was added tert-amyl alcohol (2 ml) and aldehydes (2) (1 mmol) and TBHP (1.25 mmol) in a glove box. The reaction vessel was stirred at 60 for 20 h. Then the reaction mixture was cooled and diluted with ethyl acetate, and added with saturated ahs 3. The aqueous phase was extracted with ethyl acetate. Then the organic phase was washed with saturated acl and dried S-7
8 with anhydrous a 2 S 4. The solvent was evaporated and the residue was purified by column chromatography on silica gel (petroleum ether/etac =100:1) to give the desired product. Characterization Data CH 3 3aa, white solid, yield: 78% 1 H MR (400 MHz, CDCl3): δ 7.93 (d, J = 8.0 Hz, 1H), 7.88 (d, J = 8.0 Hz, 2H), 7.64 (d, J = 8.4 Hz, 1H), 7.59 (t, J = 7.6 Hz, 1H), 7.37 (dd, J=7.2, 4.4, 3H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl3): δ , , , , , , , , , , , IR (cm-1): 3080, 2945, 1611, 1491, 1429, 1236, 824, 741. HRMS (APCI) calculated for C14H12(+): ; Found: Melting Point: CH 3 3ab, white solid, yield: 70% 1 H MR (400 MHz, CDCl 3 ): δ 7.94 (d, J = 8.0 Hz, 1H), 7.80 (s, 1H), 7.77 (d, J = 7.7 Hz, 1H), 7.65 (d, J = 8.4 Hz, 1H), (m, 1H), 7.46 (t, J = 7.6 Hz, 1H), (m, 2H), 2.48 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , , , HRMS (APCI) calculated for C 14 H 12 (+): ; Found: CH 3 3ac, white solid, yield: 56% 1 H MR (400 MHz, CDCl 3 ): δ (m, 1H), (m, 2H), 7.54 (d, J=7.5, 1H), (m, 2H), (m, 2H), 2.44 (s, 3H); 13 C MR (101 S-8
9 MHz, CDCl 3 ): δ , , , , , , , , , , , , , IR (cm -1 ): 2924, 2860, 1508, 1456, 750, 669. HRMS (APCI) calculated for C 14 H 12 (+): ; Found: ad, pale yellow oil, yield: 82% 1 H MR (500 MHz, CDCl 3 ): δ (m, 2H), 7.94 (d, J = 8.0 Hz, 1H), 7.66 (d, J = 8.4 Hz, 1H), (m, 1H), (m, 3H), (m, 1H); 13 C MR (126 MHz, CDCl 3 ): δ , , , , , , , , , , IR (cm -1 ): 3063, 2926, 2855, 1612, 1597, 1491, 1373, 897, 876, 750, 696. HRMS (APCI) calculated for C 13 H 10 (+): ; Found: CH 3 3ae, white solid, yield: 90% 1 H MR (400 MHz, CDCl 3 ): δ7.93 (d, J=8.8, 3H), 7.65 (d, J = 8.4 Hz, 1H), (m, 1H), (m, 1H), (m, 2H), 3.91 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , ,114.61,110.16, HRMS (APCI) calculated for C 14 H 12 2 (+): ; Found: CH 3 3af, yellow solid, yield: 66% 1 H MR (400 MHz, CDCl 3 ):δ7.95 (d, J=8.0, 1H), 7.66 (d, J=8.4, 1H), (m, 1H), 7.56 (d, J=7.5, 1H), 7.49 (dd, J=14.8, 6.6, 2H), 7.39 (t, J=7.4, 1H), (m, 1H), 3.91 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , , , S-9
10 IR (cm -1 ): 3262, 2922, 2849, 1585, 1506, 1489, 1456, 1196, 1036, 758,696. HRMS (APCI) calculated for C 14 H 12 2 (+): ; Found: CH 3 3ag, pale yellow oil, yield: 56% 1 H MR (400 MHz, CDCl 3 ): δ 7.70 (d, J=8.0, 1H), 7.67 (dd, J=7.5, 1.7, 1H), 7.63 (d, J=8.4, 1H), (m, 2H), (m, 1H), 7.12 (m, 2H), 3.87 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , , , HRMS (APCI) calculated for C 14 H 12 2 (+): ; Found: Me Me 3ah, white solid, yield: 53% 1 H MR (400 MHz, CDCl 3 ): δ 7.95 (d, J = 8.0 Hz, 1H), 7.66 (d, J = 8.4 Hz, 1H), (m, 1H), (m, 1H), 7.12 (d, J = 2.3 Hz, 2H), 6.64 (t, J = 2.3 Hz, 1H), 3.89 (s, 6H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , 55.60; HRMS (APCI) calculated for C 15 H 14 3 (+): ; Found: CMe 3ai, white solid, yield: 76% 1 H MR (400 MHz, CDCl 3 ): δ 8.22 (d, J = 8.4 Hz, 2H), 8.05 (d, J = 8.4 Hz, 2H), 7.93 (d, J = 8.0 Hz, 1H), 7.67 (d, J = 8.4 Hz, 1H), (m, 1H), (m, 1H), 3.97 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , , IR (cm -1 ): 3040, 2960, 1726, 1611, 1491, 1285, 1125, 1109, 739, 700. HRMS (APCI) calculated for C 15 H 12 3 (+): ; Found: Melting Point: S-10
11 o C. Cl 3aj, white solid, yield: 73% 1 H MR (400 MHz, CDCl 3 ): δ 7.91 (m, 3H), 7.67 (d, J = 8.4 Hz, 1H), (m, 1H), (m, 2H), (m, 1H); 13 C MR (100 MHz, CDCl 3 ): δ , , , , , , , , , , HRMS (APCI) calculated for C 13 H 9 Cl(+): ; Found: Melting Point: o C. CF 3 3ak, pale yellow solid, yield: 64% 1 H MR (400 MHz, CDCl 3 ): δ 8.25 (s, 1H), 8.18 (d, J = 7.7 Hz, 1H), 7.93 (d, J = 8.0 Hz, 1H), 7.82 (d, J = 7.9 Hz, 1H), (m, 1H), (m, 1H), (m, 1H), 7.44 (m,1h); 13 C MR (101 MHz, CDCl 3 ): δ , , (J=33.0), , , , , (J=3.3), (J=3.3), , (J=273.7), , , HRMS (APCI) calculated for C 14 H 9 F 3 (+): ; Found: Melting Point: o C. F 3al, yellow solid, yield: 47% 1 H MR (400 MHz, CDCl 3 ): δ (m, 2H), 7.67 (d, J=8.5,1H), (m, 1H), (m, 1H), (m, 2H), (m, 1H). 13 C MR (101 MHz, CDCl 3 ): δ , (J 1F = 253.1), , , (J 3F = 8.4), (J 4F = 3.1), , (J 4F = 3.7), , (J 3F = 7.1), , , (J 2F = 21.6), HRMS (APCI) calculated for C 13 H 9 F(+): ; Found: S-11
12 Cl Cl 3am, white solid, yield: 48% 1 H MR (400 MHz, CDCl 3 ): δ 7.92 (d, J = 8.0 Hz, 1H), 7.88 (d, J = 1.9 Hz, 2H), 7.69 (d, J = 8.4 Hz, 1H), (m, 1H), 7.54 (t, J = 1.9 Hz, 1H), 7.44 (m, 1H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , IR (cm -1 ): 3080, 2924, 1558, 1506, 1362, 750, 683. HRMS (APCI) calculated for C 13 H 8 Cl 2 (+): ; Found: an, yellow solid, yield: 75% 1 H MR (400 MHz, CDCl 3 ): δ 8.48 (s, 1H), 8.10 (dd, J=8.5, 1.6, 1H), 8.07 (d, J=8.0, 1H), 8.03 (d, J=8.5, 1H), 7.99 (dd, J=4.9, 4.3, 1H), (m, 1H), 7.69 (d, J=8.5, 1H), 7.63 (m, 1H), 7.59 (dt, J=6.8, 3.0, 2H), (m, 1H); 13 C MR (126 MHz, CDCl 3 ): δ , , , , , , , , , , , , , , , , IR (cm -1 ): 3055, 2924, 2855, 1609, 1506, 1387, 1236, 835,820, 745, 675. HRMS (APCI) calculated for C 17 H 12 (+): ; Found: Melting Point: o C. 3ao, yellow oil, yield: 63% 1 H MR (400 MHz, CDCl 3 ): δ 8.11 (dt, J = 8.0, 0.9 Hz, 1H), 7.71 (dd, J = 1.8, 0.6 Hz, 1H), (m, 2H), 7.40 (m, 1H), 7.20 (dd, J = 3.5, 0.6 Hz, 1H), 6.64 (dd, J = 3.5, 1.8 Hz, 1H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , HRMS (APCI) calculated for C 11 H 8 2 (+): ; Found: S-12
13 S 3ap, yellow solid, yield: 55% 1 H MR (400 MHz, CDCl 3 ): δ 8.01 (d, J = 8.0 Hz, 1H), 7.83 (dd, J = 3.7, 1.1 Hz, 1H), (m, 1H), (m, 1H), 7.56 (dd, J = 5.1, 1.1 Hz, 1H), 7.41 (m, 1H), 7.25 (dd, J = 5.1, 3.7 Hz, 1H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , IR (cm -1 ): 2924, 2855, 1611, 1543, 1508, 1489, 1456, 1443, 1236, 891, 847, 746, 700. HRMS (APCI) calculated for C 11 H 8 S(+): ; Found: aq, pale yellow oil, yield:63% 1 H MR (500 MHz, CDCl 3 ): δ (m, 1H), (m, 2H), 7.29 (m, 1H), (m, 2H), 1.90 (dd, J=15.0, 7.4, 2H), 1.05 (t, J=7.4, 3H); 13 C MR (126 MHz, CDCl 3 ): δ , , , , , , , 27.22, 21.11, IR (cm -1 ): 2963, 2928, 2874, 1612, 1522, 1439, 1379, 1238, 748. HRMS (APCI) calculated for C 10 H 12 (+): ; Found: ar, pale yellow oil, yield: 40% 1 H MR (500 MHz, CDCl 3 ): δ7.72 (d, J=7.9, 1H), (m, 2H), (m, 1H), 3.43 (dt, J=14.0, 7.0, 1H), (m, 6H); 13 C MR (126 MHz, CDCl 3 ): δ , , , , , , , 26.88, HRMS (APCI) calculated for C 10 H 12 (+): ; Found: as, white solid, yield: 41% S-13
14 1 H MR (500 MHz, CDCl 3 ): δ7.71 (d, J=7.9, 1H), (m, 2H), (m, 1H), 3.09 (m, 1H), (m, 2H), 1.90 (d, J=13.0, 2H), (m, 3H), 1.46 (dd, J=25.3, 12.5, 2H), (m, 1H). 13 C MR (126 MHz, CDCl 3 ): δ , , , , , , , 36.40, 31.41, 26.19, IR (cm -1 ): 2936, 2857, 1558, 1508, 1236, 754. HRMS (APCI) calculated for C 13 H 16 (+): ; Found: Melting Point: o C. 3at, yellow solid, yield: 64% 1 H MR (500 MHz, CDCl 3 ): δ 7.67 (d, J = 7.8 Hz, 1H), 7.52 (t, J = 9.0 Hz, 2H), 7.26 (dd, J = 10.5, 3.8 Hz, 1H), (m, 1H), (m, 2H), 2.01 (dd, J = 12.2, 7.4 Hz, 2H), 1.88 (s, 2H), (m, 2H); 13 C MR (126 MHz, CDCl 3 ): δ , , , , , , , 36.92, 31.34, HRMS (APCI) calculated for C 12 H 14 (+): ; Found: au, pale yellow oil, yield: 51% 1 H MR (400 MHz, CDCl 3 ): δ 7.68 (d, J = 7.9 Hz, 1H), 7.54 (d, J = 2.7 Hz, 2H), (m, 1H), (m, 1H), 1.25 (dd, J = 6.4, 4.3 Hz, 2H), (m, 2H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , 7.63, HRMS (APCI) calculated for C 10 H 10 (+): ; Found: ba, yellow solid, yield: 50% 1 H MR (400 MHz, CDCl 3 ): δ 7.88 (d, J = 8.1 Hz, 2H), 7.74 (d, J = 7.9 Hz, 1H), (m, 3H), 7.27 (dd, J = 8.2, 6.9 Hz, 1H), 2.63 (s, 3H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , 21.48, IR (cm -1 ): 2957, 1940, 1506, S-14
15 1406, 1111, 824, 743. HRMS (APCI) calculated for C 15 H 14 (+): ; Found: Melting Point: o C. 3ca, white solid, yield: 57% 1 H MR (400 MHz, CDCl 3 ): δ 7.86 (d, J = 8.1 Hz, 2H), 7.79 (d, J = 8.2 Hz, 1H), 7.43 (s, 1H), 7.37 (d, J = 7.9 Hz, 2H), 7.20 (d, J = 8.1 Hz, 1H), 2.55 (s, 3H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , 21.91, IR (cm -1 ): 2922, 2853, 1917, 1626, 1609, 1485, 1412, 1260, 1128, 1113, 824, 754, 662. HRMS (APCI) calculated for C 15 H 14 (+) : ; Found: Melting Point: o C. 3da, yellow solid, yield: 50% 1 H MR (400 MHz, CDCl 3 ): δ 7.86 (d, J = 8.1 Hz, 2H), 7.69 (s, 1H), 7.53 (d, J = 8.6 Hz, 1H), 7.41 (dd, J=8.8, 1.2, 1H), 7.37 (d, J = 7.9 Hz, 2H), 2.51 (s, 3H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , 21.48, HRMS (APCI) calculated for C 15 H 14 (+): ; Found: Me 3ea, white solid, yield: 60% 1 H MR (400 MHz, CDCl 3 ): δ 7.84 (d, J = 8.1 Hz, 2H), 7.75 (d, J = 8.8 Hz, 1H), 7.36 (d, J = 7.9 Hz, 2H), 7.06 (d, J = 2.0 Hz, 1H), 6.98 (dd, J = 8.8, 2.1 Hz, 1H), 3.91 (s, 3H), 2.45 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , 92.73, 55.78, HRMS S-15
16 (APCI) calculated for C 15 H 14 2 (+): ; Found: Melting Point: o C. F 3fa, white solid, yield: 55% 1 H MR (400 MHz, CDCl 3 ): δ (m, 1H), (m, 2H), 7.37 (d, J = 8.0 Hz, 2H), 7.32 (dd, J = 8.5, 2.0 Hz, 1H), 7.14 (td, J = 8.9, 2.1 Hz, 1H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ (J 3F = 13.7), (J 1F = 252.0), , , , , , (J 3F = 11.0 ), , (J 2F = 25.5), (J 2F =26.9), HRMS (APCI) calculated for C 14 H 11 F(+): ; Found: Melting Point: o C. Cl 3ga, pale yellow solid, yield: 64% 1 H MR (400 MHz, CDCl 3 ): δ 7.82 (d, J = 8.0 Hz, 3H), 7.64 (d, J = 1.3 Hz, 1H), (m, 3H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , HRMS (APCI) calculated for C 14 H 11 Cl(+): ; Found: Melting Point: o C. Br 3ha, white solid, yield: 76% 1 H MR (400 MHz, CDCl 3 ): δ 7.82 (d, J = 7.7 Hz, 3H), 7.77 (d, J = 8.5 Hz, 1H), 7.49 (dd, J = 8.5, 1.4 Hz, 1H), 7.37 (d, J = 7.9 Hz, 2H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , IR (cm -1 ): 2920, 2849, 1595, 1485, 1402, 826, 754. S-16
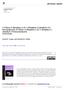
17 HRMS (APCI) calculated for C 14 H 11 Br(+): ; Found: Melting Point: o C. Cl 3ia, white solid, yield: 40% 1 H MR (400 MHz, CDCl 3 ): δ 7.88 (d, J = 1.3 Hz, 1H), 7.81 (d, J = 8.1 Hz, 2H), (m, 2H), 7.37 (d, J = 7.9 Hz, 2H), 2.46 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ , , , , , , , , , , , IR (cm -1 ): 2922, 2855, 1491, 1425, 1358, 1265, 812, 758; HRMS (APCI) calculated for C 14 H 11 Cl(+): ; Found: Melting Point: o C. Br 3ja, white solid, yield: 48% 1 H MR (400 MHz, CDCl 3 ): δ 8.06 (d, J = 1.7 Hz, 1H), 7.82 (d, J = 8.1 Hz, 2H), 7.68 (dd, J = 8.8, 1.9 Hz, 1H), 7.54 (d, J = 8.8 Hz, 1H), 7.38 (d, J = 7.9 Hz, 2H), 2.47 (s, 3H); 13 C MR (101 MHz, CDCl 3 ): δ162.69, , , , , , , , , , , HRMS (APCI) calculated for C 14 H 11 Br(+): ; Found: X-ray Crystallographic Data for 3-(p-tolyl)benzo[d]isoxazole Figure S1. 3-(p-tolyl)benzo[d]isoxazole (3aa) S-17
18 S-18
19 Table S2. Crystal data and structure refinement for a. Identification code a Empirical formula C14 H11 Formula weight Temperature 295(2) K Wavelength Å Crystal system Monoclinic Space group P 21/c Unit cell dimensions a = (18) Å = 90. b = (4) Å = (7). c = (3) Å = 90. Volume (19) Å 3 Z 8 Density (calculated) Mg/m 3 Absorption coefficient mm -1 F(000) 880 Crystal size 0.30 x 0.10 x 0.05 mm 3 Theta range for data collection 6.56 to Index ranges -24<=h<=30, -12<=k<=13, -9<=l<=8 Reflections collected Independent reflections 3881 [R(int) = ] Completeness to theta = % Absorption correction Semi-empirical from equivalents Max. and min. transmission and Refinement method Full-matrix least-squares on F 2 Data / restraints / parameters 3881 / 0 / 291 Goodness-of-fit on F Final R indices [I>2sigma(I)] R1 = , wr2 = R indices (all data) R1 = , wr2 = Largest diff. peak and hole and e.å -3 S-19
20 Table S3 Atomic coordinates ( x 10 4 ) and equivalent isotropic displacement parameters (Å 2 x 10 3 ) for a. U(eq) is defined as one third of the trace of the orthogonalized U ij tensor. x y z U(eq) (1) 4411(1) 9719(1) 4035(2) 84(1) (2) 577(1) 4791(1) 6083(2) 92(1) (1) 3875(1) 9898(1) 4427(2) 76(1) (2) 1121(1) 4960(1) 5772(2) 81(1) C(1) 1428(1) 8582(2) 5887(3) 89(1) C(2) 2008(1) 8656(1) 5559(2) 60(1) C(3) 2324(1) 9600(1) 6157(2) 65(1) C(4) 2853(1) 9660(1) 5847(2) 60(1) C(5) 3093(1) 8780(1) 4909(2) 51(1) C(6) 3656(1) 8861(1) 4525(2) 55(1) C(7) 4028(1) 7953(1) 4195(2) 53(1) C(8) 4490(1) 8547(2) 3896(2) 64(1) C(9) 4959(1) 8004(2) 3510(2) 80(1) C(10) 4947(1) 6812(2) 3436(2) 81(1) C(11) 4030(1) 6729(2) 4149(2) 63(1) C(12) 4492(1) 6181(2) 3764(2) 76(1) C(13) 2779(1) 7836(1) 4325(2) 58(1) C(14) 2250(1) 7782(1) 4657(2) 61(1) C(15) 3581(1) 3404(2) 4758(3) 90(1) C(16) 2996(1) 3561(1) 4961(2) 60(1) C(17) 2632(1) 2729(1) 4327(2) 62(1) C(18) 2096(1) 2848(1) 4545(2) 57(1) C(19) 1903(1) 3810(1) 5409(2) 51(1) C(20) 1333(1) 3920(1) 5680(2) 55(1) C(21) 949(1) 3017(1) 5911(2) 54(1) C(22) 487(1) 3621(2) 6163(2) 67(1) C(23) 9(1) 3088(2) 6484(2) 83(1) C(24) 14(1) 1902(2) 6561(2) 82(1) C(25) 2269(1) 4656(1) 6055(2) 60(1) C(26) 2803(1) 4530(1) 5830(2) 65(1) C(27) 471(1) 1260(2) 6303(2) 78(1) C(28) 942(1) 1795(2) 5983(2) 66(1) S-20
21 Table S4. Bond lengths [Å] and angles [ ] for a. (1)-C(8) 1.359(2) (1)-(1) (17) (2)-C(22) 1.359(2) (2)-(2) (18) (1)-C(6) (18) (2)-C(20) (18) C(1)-C(2) 1.502(2) C(1)-H(1A) C(1)-H(1B) C(1)-H(1C) C(2)-C(14) 1.377(2) C(2)-C(3) 1.393(2) C(3)-C(4) 1.372(2) C(3)-H(3) C(4)-C(5) (19) C(4)-H(4) C(5)-C(13) 1.389(2) C(5)-C(6) (19) C(6)-C(7) 1.431(2) C(7)-C(8) 1.380(2) C(7)-C(11) 1.399(2) C(8)-C(9) 1.384(2) C(9)-C(10) 1.362(3) C(9)-H(9) C(10)-C(12) 1.391(3) C(10)-H(10) C(11)-C(12) 1.371(2) C(11)-H(11) C(12)-H(12) C(13)-C(14) 1.375(2) C(13)-H(13) C(14)-H(14) C(15)-C(16) 1.502(2) C(15)-H(15A) C(15)-H(15B) C(15)-H(15C) S-21
22 C(16)-C(17) 1.380(2) C(16)-C(26) 1.393(2) C(17)-C(18) 1.377(2) C(17)-H(17) C(18)-C(19) (19) C(18)-H(18) C(19)-C(25) 1.396(2) C(19)-C(20) (19) C(20)-C(21) 1.433(2) C(21)-C(22) 1.378(2) C(21)-C(28) 1.397(2) C(22)-C(23) 1.386(2) C(23)-C(24) 1.357(3) C(23)-H(23) C(24)-C(27) 1.389(3) C(24)-H(24) C(25)-C(26) 1.375(2) C(25)-H(25) C(26)-H(26) C(27)-C(28) 1.370(2) C(27)-H(27) C(28)-H(28) C(8)-(1)-(1) (11) C(22)-(2)-(2) (11) C(6)-(1)-(1) (12) C(20)-(2)-(2) (13) C(2)-C(1)-H(1A) C(2)-C(1)-H(1B) H(1A)-C(1)-H(1B) C(2)-C(1)-H(1C) H(1A)-C(1)-H(1C) H(1B)-C(1)-H(1C) C(14)-C(2)-C(3) (14) C(14)-C(2)-C(1) (15) C(3)-C(2)-C(1) (15) C(4)-C(3)-C(2) (14) C(4)-C(3)-H(3) S-22
23 C(2)-C(3)-H(3) C(3)-C(4)-C(5) (14) C(3)-C(4)-H(4) C(5)-C(4)-H(4) C(13)-C(5)-C(4) (13) C(13)-C(5)-C(6) (13) C(4)-C(5)-C(6) (13) (1)-C(6)-C(7) (13) (1)-C(6)-C(5) (13) C(7)-C(6)-C(5) (13) C(8)-C(7)-C(11) (14) C(8)-C(7)-C(6) (14) C(11)-C(7)-C(6) (13) (1)-C(8)-C(7) (14) (1)-C(8)-C(9) (16) C(7)-C(8)-C(9) (18) C(10)-C(9)-C(8) (16) C(10)-C(9)-H(9) C(8)-C(9)-H(9) C(9)-C(10)-C(12) (16) C(9)-C(10)-H(10) C(12)-C(10)-H(10) C(12)-C(11)-C(7) (15) C(12)-C(11)-H(11) C(7)-C(11)-H(11) C(11)-C(12)-C(10) (18) C(11)-C(12)-H(12) C(10)-C(12)-H(12) C(14)-C(13)-C(5) (14) C(14)-C(13)-H(13) C(5)-C(13)-H(13) C(13)-C(14)-C(2) (14) C(13)-C(14)-H(14) C(2)-C(14)-H(14) C(16)-C(15)-H(15A) C(16)-C(15)-H(15B) H(15A)-C(15)-H(15B) C(16)-C(15)-H(15C) S-23
24 H(15A)-C(15)-H(15C) H(15B)-C(15)-H(15C) C(17)-C(16)-C(26) (14) C(17)-C(16)-C(15) (16) C(26)-C(16)-C(15) (15) C(18)-C(17)-C(16) (14) C(18)-C(17)-H(17) C(16)-C(17)-H(17) C(17)-C(18)-C(19) (13) C(17)-C(18)-H(18) C(19)-C(18)-H(18) C(18)-C(19)-C(25) (13) C(18)-C(19)-C(20) (12) C(25)-C(19)-C(20) (13) (2)-C(20)-C(21) (13) (2)-C(20)-C(19) (13) C(21)-C(20)-C(19) (13) C(22)-C(21)-C(28) (14) C(22)-C(21)-C(20) (15) C(28)-C(21)-C(20) (13) (2)-C(22)-C(21) (14) (2)-C(22)-C(23) (16) C(21)-C(22)-C(23) (18) C(24)-C(23)-C(22) (16) C(24)-C(23)-H(23) C(22)-C(23)-H(23) C(23)-C(24)-C(27) (16) C(23)-C(24)-H(24) C(27)-C(24)-H(24) C(26)-C(25)-C(19) (14) C(26)-C(25)-H(25) C(19)-C(25)-H(25) C(25)-C(26)-C(16) (14) C(25)-C(26)-H(26) C(16)-C(26)-H(26) C(28)-C(27)-C(24) (19) C(28)-C(27)-H(27) C(24)-C(27)-H(27) S-24
25 C(27)-C(28)-C(21) (15) C(27)-C(28)-H(28) C(21)-C(28)-H(28) Symmetry transformations used to generate equivalent atoms: S-25
26 Table S5. Anisotropic displacement parameters (Å 2 x 10 3 )for a. The anisotropic displacement factor exponent takes the form: -2π 2 [ h 2 a* 2 U h k a* b* U 12 ] U 11 U 22 U 33 U 23 U 13 U 12 (1) 67(1) 69(1) 118(1) 15(1) 8(1) -13(1) (2) 74(1) 72(1) 132(1) -5(1) 21(1) 16(1) (1) 68(1) 58(1) 102(1) 10(1) 3(1) -6(1) (2) 75(1) 59(1) 112(1) -2(1) 15(1) 8(1) C(1) 64(1) 100(2) 104(2) 2(1) 9(1) 14(1) C(2) 61(1) 59(1) 60(1) 4(1) 0(1) 9(1) C(3) 75(1) 57(1) 63(1) -7(1) 4(1) 15(1) C(4) 74(1) 46(1) 59(1) -4(1) -4(1) 3(1) C(5) 61(1) 45(1) 46(1) 1(1) -1(1) 2(1) C(6) 64(1) 49(1) 50(1) 4(1) -2(1) -5(1) C(7) 57(1) 58(1) 44(1) 0(1) 2(1) 1(1) C(8) 64(1) 67(1) 61(1) 4(1) 4(1) -7(1) C(9) 57(1) 106(2) 78(1) 3(1) 10(1) -6(1) C(10) 63(1) 105(2) 75(1) -14(1) 7(1) 14(1) C(11) 64(1) 61(1) 65(1) -6(1) 4(1) 0(1) C(12) 71(1) 71(1) 86(1) -15(1) 3(1) 11(1) C(13) 65(1) 50(1) 59(1) -8(1) 4(1) 2(1) C(14) 65(1) 53(1) 65(1) -6(1) -1(1) -2(1) C(15) 65(1) 97(2) 109(2) 7(1) 6(1) -1(1) C(16) 63(1) 60(1) 57(1) 6(1) 4(1) -5(1) C(17) 71(1) 54(1) 61(1) -4(1) 11(1) 0(1) C(18) 67(1) 51(1) 53(1) -6(1) 4(1) -8(1) C(19) 63(1) 46(1) 45(1) 3(1) 2(1) -3(1) C(20) 67(1) 48(1) 51(1) 0(1) 4(1) 5(1) C(21) 57(1) 60(1) 45(1) 4(1) 2(1) 1(1) C(22) 68(1) 68(1) 66(1) 4(1) 9(1) 9(1) C(23) 60(1) 110(2) 81(1) 8(1) 13(1) 9(1) C(24) 65(1) 105(2) 78(1) 18(1) 5(1) -14(1) C(25) 78(1) 48(1) 54(1) -3(1) 6(1) -5(1) C(26) 76(1) 59(1) 58(1) -1(1) -1(1) -17(1) C(27) 70(1) 74(1) 89(1) 16(1) 2(1) -10(1) C(28) 65(1) 61(1) 70(1) 11(1) 1(1) -1(1) S-26
27 6. Mechanistic Studies 6.1 KIE Experiment Synthesis of deuterated substrate 1a-d 5 D D D D B(H)2 + D H CuCl Pyridine 4A sieves, 1,2-Dichloroethane rt 1.H 2 H 2, MeH/CHCl 3, rt 2.Ac 2,Et 2, 0 C to rt D D D D H D 1a-d 5 Following the general procedure for the synthesis of substrate 1 1, deuterated substrate 1a-d 5 was obtained from (d 5 -phenyl)boronic acid D D H D D D Intermolecular Kinetic Isotope Effect H H H H HAc H D D + HAc + H D D D 10 mol% Pd(TFA)2 2.5 equiv TBHP t-amh, 60 C, 1h 2 H 4 /D 4 A mixture of 1a-d 5 (31.2 mg, 0.2 mmol), 1a (30.2 mg, 0.2 mmol), Pd(TFA) 2 (6.6mg, 10 mol%), p-tolualdehyde (23.6µl, 0.2 mmol) and TBHP (0.06 ml, 0.5 mmol) in tert-amyl alcohol (1ml) was stirred at 60 for 1h under 2 atmosphere. The solvent was evaporated to dryness in vacuo. The residual was separated on a silica gel column to get the products. S-27
28 The intermolecular KIE was determined by 1 HMR: K H /K D =3.2 CH 3 H 4 /D 4 S-28
29 6.2 HAc 10 mol% Pd(TFA)2 2.5 eq TBHP t-amh, 60 o C, 20h S.M 1 2 -Phenoxyacetamides substrates (1) (0.4 mmol) and Pd(TFA) 2 (10 mol%) were weighed into a 25ml pressure tube, to which was added tert-amyl alcohol (1 ml) and TBHP (1 mmol) in a glove box. The reaction vessel was stirred at 60 for 20 h. The solvent was evaporated and the residue was purified by column chromatography on silica gel. We only obtained the starting material 1. o phenyl acetate was observed, which ruled out the possibility of nitrogen radical initiation a HAc 10 mol% Pd(TFA)2 2.5 eq TBHP D-t-Butanol, 60 o C 2, 6h S.M -Phenoxyacetamides substrates (1) (0.4 mmol) and Pd(TFA) 2 (10 mol%) were weighed into a 25mL pressure tube, to which was added D-tert-Butanol (1 ml) and TBHP (1 mmol) in a glove box. The reaction vessel was stirred at 60 for 6 h. The solvent was evaporated and the residue was purified by column chromatography on silica gel. We only recovered the starting material without any D-compound. 1a HAc 10 mol% Pd(TFA)2 D-t-Butanol, 60 o C 2, 1h S.M 77% recovery Then we carried the experiment without TBHP and reducing the reaction time to 1h, the result was consisted with that above mentioned. So we concluded that the C-H activation was irreversible. 6.4 HAc + H 10 mol% Pd(TFA)2 t-amh, 60 o C, 20h.R. 2 1a 2a -Phenoxyacetamides substrates (1) (0.4 mmol) and Pd(TFA) 2 (10 mol%) were weighed into a 25mL pressure tube, to which was added tert-amyl alcohol (1 ml) and aldehyde (0.8 mmol) in a glove box. The reaction vessel was stirred at 60 for 20 h. The solvent was evaporated and the residue was purified by column chromatography on silica gel. o product was obtained and the aldehyde was recovered which demonstrated that S-29
30 aldehyde could be only transformed when TBHP was present. HAc + H 1 eq Pd(TFA)2 t-amh, 60 ο C, 20h 2.R 1a 2a When the loading of catalyst was increased to 1equivalent, we failed to obtain the desired product, which rejected a catalytic cycle involving Pd(II)/Pd(0)/Pd(II) process. 6.5 Effect of radical scavenger a,b 1a HAc + 2a 2eq H 10 mol% Pd(TFA)2 2.5 equiv TBHP TEMP t-amh, 60 C, 20h 2 3aa Entry TEMP Yield(GC) (mol%) % % R R. a The reactions were carried out in a 0.1 mmol-scale of 1a. b Yield was determined by GC using mesitylene as internal standard. 6.6 F + H H THF K 1k HAc Potassium tert-butoxide (1.24g, 11mmol) was added to a solution of acetohydroxamic acid (0.83g, 11 mmol) in dry THF (15 ml). After stirring for 1 h, 2-fluorobenzophenone (1.7ml, 10 mmol) in THF (15 ml) was added, and the reaction was brought to reflux. After refluxing for overnight, the reaction mixture was cooled and distributed between ether and saturated aqueous ammonium chloride solution, after which the organic layer was separated, dried (MgS 4 ), and evaporated, then purified by flash silica gel column chromatography to give the corresponding product (1k). 2 Ph S-30
31 HAc Ph 1k, white solid, 5% 1 H MR (500 MHz, DMS): δ11.66 (s, 1H), 7.78 (d, J=7.4, 2H), 7.65 (t, J=7.2, 1H), 7.51 (t, J=7.4, 3H), 7.35 (d, J=7.0, 1H), (m, 2H), 1.82 (s, 3H); 13 C MR (126 MHz, DMS): δ195.07, , , , , , , , , , , , IR (cm -1 ): 3117, 2928, 1653, 1508, 928, 756. HRMS (ESI) calculated for C 15 H 13 3 a(+): ; Found: HAc Ph X mol% Pd(TFA)2 Additive, xidant t-amh, 60 C, 20h 1k 2 100% conversion 3ad Ph Entry X Additive xidant Yield(GC) 1 10 mol% Pd(TFA) % 2 10 mol% Pd(TFA) 2 1 eq TFA - 73% 3 10 mol% Pd(TFA) eq TBHP 19% The starting materials were all exclusively converted to the product by GC detection, which indicated that the starting material 1k was the intermediate in the process of the reaction. 6.7 HAc + H 10 mol% Pd(TFA)2 2.5 equiv TBHP t-amh, 60 C, 20h 2 + We observed the generation of tert-pentyl acetate under our standard reaction conditions by GC-MS. 7. DFT Calculations All the calculations were carried out with the Gaussian 09 package. 3 Geometry optimization and energy calculations were performed with the B3LYP method. 4 The 6-31G (d) basis set was used for all atoms except Pd, for which a LAL2DZ basis set with ECP was used. Single point energy calculations were then carried out on the above-obtained geometries at the M06 5 /SDD G (d, p) level and solvent effect (solvent = t BuH) was calculated using the SMD 7 S-31
32 solvation model. Computed structures were illustrated using CYLVIEW 8 drawings. TS1_A TS2_A TS3_A TS4_A Figure S2. The structures of four transition states. Ac HAc R Ph B10 Ph B11 Ac Pd H H Pd(TFA)2 C-H activation 1 CF 3 CH Pd B5 Ac aldehyde insertion nucleophilic attack H 2 R Ac PdH (II) Ph β-h elimination H Ac Pd Ph B9 B7 Figure S3. A catalytic cycle involving direct aldehyde insertion. S-32
33 Ac H Pd CF 3 TS1_B CF 3 H Pd CF 3 TS2_B Ac Ac Pd H Ph TS3_B Ph H TS4_B Pd Ac PdH Ph TS5_B TS3_B 36.8 (35.0) 33.9 (33.2) B (38.2) TS4_B sol_tbuh G ( H ) kcal/mol 0.0 (0.0) -6.2 ( ) B1 TS1_B -1.6 ( ) ( ) B2-9.1 ( -8.0 ) B3 TS2_B 5.0 (4.2) ( -8.5 ) B4-8.2 (4.3) B5 ( ) (6.5) B (6.5) B9 TS5_B 14.6 (12.7) (12.1) B10 B6 Ac H Pd CF 3 B1 CF 3 H CF 3 B2 Ac Pd CF 3 H Pd CF 3 B3 Ac H Pd CF 3 Ac Pd Ac Ph B4 B5 B6 H Pd Ac ( ) B11 Ac Pd H Ph B7 Ph H B8 Pd Ac PdH Ph B9 Ac Ph Pd H B10 Ph B11 H Ac Ph Pro ( ) Pro -H activation C-H activation aldehyde insertion β-h elimination Figure S4. Gibbs free energy profile for the mechanism in Figure S3. ucleophilic attack eacylation The C-H activation step gave palladcycle intermediate B5. In the main text, radical addition of acyl radical generated from aldehyde by TBHP gave intermediate A6. Alternatively, the benzaldehyde could coordinate to intermediate B5 and the direct aldehyde insertion led to the intermediate B7. β-hydrogen elimination of B7 could form intermediate B9 which was similar to A8 in the radical mechanism shown in the main text. The calculation results showed that the aldehyde insertion was the rate-determining step with very high activation energy of 52.3kcal/mol. Therefore, this mechanism was not reasonable in our case. S-33
34 Ac TBHP HAc Ph Pd(TFA) 2 C2 Ph C12 H Ac Ac Pd Ph CF 3 C11 F 3 C Ac Pd C3 CF 3 TBHP H Ac Pd Ph CF 3 C10 reductive elimination Ph Pd Ac CF 3 C7 CF 3 Ac Pd Ph C4 C-H activation CF 3 Figure S5. A mechanism proposed involving nitrogen radical initiation. sol_tbuh G ( H) kcal/mol 0.0 (0.0) + Pd(TFA) 2 HAc C (35.3) C1 C (39.2) Ac H t-bu Ac Pd CF 3 F 3 C 39.8 CF 3 TS1_C 20.4 (20.7) C2 C2 Ac t-buh Ac Pd Ph C4-0.6 (-0.5) CF 3 C3 Ac H t-bu TS1_C (-70.4) C4 Ac H Pd CF 3 Ph CF 3 TS2_C (-70.6) C5 TS2_C Ac H Pd CF 3 Ph CF 3 C (-47.3) (-53.0) C6 Ac Pd Ph CF 3 TS3_C Ac Pd CF 3 H Ph CF 3 C (-74.4) C7 Ac Pd CF 3 Ph C (-63.7) TS3_C (-84.4) C8 Ac Pd Ph CF 3 TS4_C Ac Pd Ph CF 3 C7 Ac Pd Ph CF 3 C11 C (-89.0) C10 TS4_C (-82.1) Pro -H activation Radical addition C-H activation Reductive elimiantion ucleophilic attack eacylation Figure S6. Gibbs free energy profile for the mechanism in Figure S (-72.0) Ac Pd Ph CF 3 C Ac Ph H C (-87.7) C11 Ac Pd Ph CF 3 C9 Ph Pro (-76.6) C (-92.6) As shown in Figure S5, a mechanism involving nitrogen radical initiation was proposed which was similar to the mechanism reported by Kang Zhao and coworkers. 9 The radical activation of -phenoxyacetamides gave the amide nitrogen radical that underwent radical addition to form a Pd III radical intermediate C3. This step costed activation energy of 39.8 kcal/mol (Figure S6) and is the S-34
35 rate-determining step. Therefore, we excluded this mechanism in our case. The following steps, C-H activation, reductive elimination, nucleophilic attack and deacylation were similar to the steps in the mechanism proposed in the main text. sol_tbuh Ac Pd Ph A9 CF 3 Pd Ph TS5_A_1 CF 3 G ( H) ( -87.7) ( -57.4) sol_tbuh G ( H) Ph Ph H H A10 TS5_A_ ( -76.6) ( -34.6) Figure S7. Gibbs free energy for elimination reaction. In our computational study, we considered the pathway from A9 to TS5_A_1, in which the attack of the Ac group took place before hydrolysis. This pathway had a very high barrier. We further calculated the pathway of intramolecular to acetyl transfer. The calculation suggested that this pathway also had a very high barrier (43.5kcal/mol) and was unlikely to occur. Table S6. Energies of structures on pathway A (single point energy were calculated at the M06/SDD, G(d,p) level of theory in solvent). Structure E H G E H G PdTFA Sub PhCH tbuh tbuh TFA tamh tamylac H A TS1_A A A TS2_A S-35
36 A A A TS3_A A A TS4_A A A Pro Table S7. Energies of structures on pathway B (single point energy were calculated at the M06/SDD, G(d,p) level of theory in solvent). Structure E H G E H G B1(A1) TS1_B(TS1_A) B2(A2) B3(A3) TS2_B(TS2_A) B4(A4) B5(A5) B TS3_B B B TS4_B B TS5_B B B11(A10) Pro Table S8. Energies of structures on pathway C (single point energy were calculated at the M06/SDD, G(d,p) level of theory in solvent). Structure E H G E H G C TS1_C C C C C TS2_C S-36
37 C C7(A6) C TS3_C(TS3_A) C9(A7) C10(A8) TS4_C(TS4_A) C11(A9) C12(A10) Pro Cartesian coordinates (in Å) of related structures which were calculated at the B3LYP/Lanl2dz G(d) level of theory. TS1_A TS2_A Cartesian coordinates Cartesian coordinates ATM X Y Z ATM X Y Z H C C C C C C C C C C C H C C C H C C C H C H H H H H C H C H H H H C C Pd H F S-37
38 H F H F Pd F F F F F F TS3_A TS4_A Cartesian coordinates Cartesian coordinates ATM X Y Z ATM X Y Z C C C C C C C C C C H H C C H H H H H H C C C C H H H H H H C C C C F F F F F F Pd Pd C C C C C C S-38
39 C C C C H H C C H H C C H H H H H H A1 A2 Cartesian coordinates Cartesian coordinates ATM X Y Z ATM X Y Z H H C C C C C C C C C C C C C C C C C C C C H H H H H H H H H H C C C C H H H H H H Pd Pd F F F F S-39
Redox-Innocent Metal-Assisted Cleavage of S-S. Bond in a Disulfide-Containing Ligand.
 Supplementary Information for Redox-Innocent Metal-Assisted Cleavage of S-S Bond in a Disulfide-Containing Ligand. Charlène Esmieu, Maylis Orio, Laurent Le Pape, Colette Lebrun, Jacques Pécaut, Stéphane
Supplementary Information for Redox-Innocent Metal-Assisted Cleavage of S-S Bond in a Disulfide-Containing Ligand. Charlène Esmieu, Maylis Orio, Laurent Le Pape, Colette Lebrun, Jacques Pécaut, Stéphane
Synthesis and spectroscopic properties of β meso directly linked porphyrin corrole hybrid compounds
 Supporting Information for Synthesis and spectroscopic properties of β meso directly linked porphyrin corrole hybrid compounds Baris Temelli * and Hilal Kalkan Address: Hacettepe University, Department
Supporting Information for Synthesis and spectroscopic properties of β meso directly linked porphyrin corrole hybrid compounds Baris Temelli * and Hilal Kalkan Address: Hacettepe University, Department
Supporting Information
 Highly diastereoselective cyclopropanation of -methylstyrene catalyzed by a C 2 -symmetrical chiral iron porphyrin complex Daniela Intrieri, Stéphane Le Gac, Alessandro Caselli, Eric Rose, Bernard Boitrel,
Highly diastereoselective cyclopropanation of -methylstyrene catalyzed by a C 2 -symmetrical chiral iron porphyrin complex Daniela Intrieri, Stéphane Le Gac, Alessandro Caselli, Eric Rose, Bernard Boitrel,
Supporting Information
 Momiyama, Kanan, Liu page S1 Synthesis of Acyclic!,"-Unsaturated Ketones via Pd(II)-Catalyzed Intermolecular Reaction of Alkynamides and Alkenes Norie Momiyama, Matthew W. Kanan and David R. Liu* Department
Momiyama, Kanan, Liu page S1 Synthesis of Acyclic!,"-Unsaturated Ketones via Pd(II)-Catalyzed Intermolecular Reaction of Alkynamides and Alkenes Norie Momiyama, Matthew W. Kanan and David R. Liu* Department
Supporting Information
 Supporting Information rganocatalytic Mitsunobu Reactions Tracy Yuen Sze But and Patrick H. Toy * Department of Chemistry, The University of Hong Kong, Pokfulam Road, Hong Kong, People s Republic of China
Supporting Information rganocatalytic Mitsunobu Reactions Tracy Yuen Sze But and Patrick H. Toy * Department of Chemistry, The University of Hong Kong, Pokfulam Road, Hong Kong, People s Republic of China
2,4 and 2,5-bis(benzooxazol-2 -yl)hydroquinone (DHBO) and their borate complexes: Synthesis and Optical properties
 Electronic Supplementary Material (ESI) for ew Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre ational de la Recherche Scientifique 2016 Supplementary Material 2,4 and
Electronic Supplementary Material (ESI) for ew Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre ational de la Recherche Scientifique 2016 Supplementary Material 2,4 and
Supporting Information
 Supporting Information for Simple two-step synthesis of 2,4-disubstituted pyrroles and 3,5-disubstituted pyrrole-2-carbonitriles from enones Murat Kucukdisli 1, Dorota Ferenc 1, Marcel Heinz 2, Christine
Supporting Information for Simple two-step synthesis of 2,4-disubstituted pyrroles and 3,5-disubstituted pyrrole-2-carbonitriles from enones Murat Kucukdisli 1, Dorota Ferenc 1, Marcel Heinz 2, Christine
Macrocyclic Scaffolds Derived from para-aminobenzoic acid. Electronic Supplementary material
 Macrocyclic Scaffolds Derived from para-aminobenzoic acid Electronic Supplementary material Fred Campbell a, Jeffrey Plante, a Christopher Carruthers, a Michaele J. Hardie, a Timothy Prior b and Andrew
Macrocyclic Scaffolds Derived from para-aminobenzoic acid Electronic Supplementary material Fred Campbell a, Jeffrey Plante, a Christopher Carruthers, a Michaele J. Hardie, a Timothy Prior b and Andrew
Supporting Information
 Supporting Information A New Generation of Radiofluorinated Pyrimidine-2,4,6-triones as MMP-targeted Radiotracers for Positron Emission Tomography Daniela Schrigten,, Hans-Jörg Breyholz, Stefan Wagner,
Supporting Information A New Generation of Radiofluorinated Pyrimidine-2,4,6-triones as MMP-targeted Radiotracers for Positron Emission Tomography Daniela Schrigten,, Hans-Jörg Breyholz, Stefan Wagner,
DPO and POPOP Carboxylate-Analogs Sensors by Sequential Palladium-Catalysed Direct Arylation of Oxazole-4-Carboxylates
 Electronic Supplementary Information DP and PPP Carboxylate-Analogs Sensors by Sequential Palladium-Catalysed Direct Arylation of xazole-4-carboxylates Cécile Verrier, Catherine Fiol-Petit, Christophe
Electronic Supplementary Information DP and PPP Carboxylate-Analogs Sensors by Sequential Palladium-Catalysed Direct Arylation of xazole-4-carboxylates Cécile Verrier, Catherine Fiol-Petit, Christophe
Supporting information for. Base-Mediated Cascade Cyclization: Stereoselective Synthesis of Benzooxazocinone
 Supporting information for Base-Mediated Cascade Cyclization: Stereoselective Synthesis of Benzooxazocinone Chiranan Pramthaisong, Rattana Worayuthakarn, Vannapha Pharikronburee, Tanwawan Duangthongyou,,
Supporting information for Base-Mediated Cascade Cyclization: Stereoselective Synthesis of Benzooxazocinone Chiranan Pramthaisong, Rattana Worayuthakarn, Vannapha Pharikronburee, Tanwawan Duangthongyou,,
Supporting Information. Metalated Ir(III) complexes based on the luminescent diimine ligands: synthesis and photophysical study.
 Supporting Information Metalated Ir(III) complexes based on the luminescent diimine ligands: synthesis and photophysical study. Julia R. Shakirova, Olesya A. Tomashenko, Ekaterina E. Galenko, Alexander
Supporting Information Metalated Ir(III) complexes based on the luminescent diimine ligands: synthesis and photophysical study. Julia R. Shakirova, Olesya A. Tomashenko, Ekaterina E. Galenko, Alexander
Design, Synthesis and Antitumor Activity of Novel link-bridge and. B-Ring Modified Combretastatin A-4 (CA-4) Analogues as Potent. Antitubulin Agents
 Design, Synthesis and Antitumor Activity of Novel link-bridge and B-Ring Modified Combretastatin A-4 (CA-4) Analogues as Potent Antitubulin Agents Yong-Tao Duan 1, Ruo-Jun Man 1, Dan-Jie Tang 1, Yong-Fang
Design, Synthesis and Antitumor Activity of Novel link-bridge and B-Ring Modified Combretastatin A-4 (CA-4) Analogues as Potent Antitubulin Agents Yong-Tao Duan 1, Ruo-Jun Man 1, Dan-Jie Tang 1, Yong-Fang
Synthetic Procedure for aminolink-na dimer used for Immobilization. H N O C 6 F 5
 Supplementary Methods Synthetic Procedure for aminolink-a dimer used for Immobilization. -Boc-aminolink-A (3) Synthetic Scheme of aminolink-a-dimer (8) A (1) 2 ab 3 C, Me, 68% Cl 92% 3: = Boc 4: = C 6
Supplementary Methods Synthetic Procedure for aminolink-a dimer used for Immobilization. -Boc-aminolink-A (3) Synthetic Scheme of aminolink-a-dimer (8) A (1) 2 ab 3 C, Me, 68% Cl 92% 3: = Boc 4: = C 6
Reactions of 1,5-Diaryl-3-Trifluoromethyl Pent-1-en-4-yn-3-yl Cations with Benzene in TfOH. Synthesis of CF 3 - Helicopter -Like Molecules
 Supporting Information Reactions of 1,5-Diaryl-3-Trifluoromethyl Pent-1-en-4-yn-3-yl Cations with Benzene in TfOH. Synthesis of CF 3 - Helicopter -Like Molecules Aleksey V. Zerov, Galina L. Starova, Vitalii
Supporting Information Reactions of 1,5-Diaryl-3-Trifluoromethyl Pent-1-en-4-yn-3-yl Cations with Benzene in TfOH. Synthesis of CF 3 - Helicopter -Like Molecules Aleksey V. Zerov, Galina L. Starova, Vitalii
Synthesis of Esters of Substituted 6-Aminohexanoic Acid as Potential Transdermal Penetration Enhancers
 Synthesis of Esters of Substituted 6-Aminohexanoic Acid as Potential Transdermal Penetration Enhancers Katerina Brychtova, ldrich Farsa, Jozef Csollei Department of Chemical Drugs, Faculty of Pharmacy,
Synthesis of Esters of Substituted 6-Aminohexanoic Acid as Potential Transdermal Penetration Enhancers Katerina Brychtova, ldrich Farsa, Jozef Csollei Department of Chemical Drugs, Faculty of Pharmacy,
Supporting Information
 Supporting Information Experimental General procedures The product distribution for the reaction of PCl 3 for the synthesis of phosphorodiamidites/ phosphoramidite was examined in situ by 31 P NMR and
Supporting Information Experimental General procedures The product distribution for the reaction of PCl 3 for the synthesis of phosphorodiamidites/ phosphoramidite was examined in situ by 31 P NMR and
Supporting Information for. First Practical Cross-Alkylation of Primary Alcohols with a New and Recyclable Impregnated. Iridium on Magnetite Catalyst
 Supporting Information for First Practical Cross-Alkylation of Primary Alcohols with a New and Recyclable Impregnated Iridium on Magnetite Catalyst Rafael Cano,Miguel Yus and Diego J. Ramón* Instituto
Supporting Information for First Practical Cross-Alkylation of Primary Alcohols with a New and Recyclable Impregnated Iridium on Magnetite Catalyst Rafael Cano,Miguel Yus and Diego J. Ramón* Instituto
Towards Metal Complexes that can Directionally Walk Along Tracks: Controlled Stepping of a Molecular Biped with a Palladium(II) Foot
 S1 - Supporting Information Towards Metal Complexes that can Directionally Walk Along Tracks: Controlled Stepping of a Molecular Biped with a Palladium(II) Foot Jonathon E. Beves, Victor Blanco, Barry
S1 - Supporting Information Towards Metal Complexes that can Directionally Walk Along Tracks: Controlled Stepping of a Molecular Biped with a Palladium(II) Foot Jonathon E. Beves, Victor Blanco, Barry
Efficient Palladium-catalyzed Coupling Reactions of Aryl Bromides and Chlorides with Phenols
 Efficient Palladium-catalyzed Coupling Reactions of Aryl Bromides and Chlorides with Phenols Tongjie Hu, a Thomas Schulz, b Christian Torborg, b Xiaorong Chen, a Jun Wang, a Matthias Beller b* and Jun
Efficient Palladium-catalyzed Coupling Reactions of Aryl Bromides and Chlorides with Phenols Tongjie Hu, a Thomas Schulz, b Christian Torborg, b Xiaorong Chen, a Jun Wang, a Matthias Beller b* and Jun
Supporting Information
 upporting Information Unexpected ynthesis of ovel 3-allyl-5-(arylidene)-2-thioxo-thiazolidin-4- ones in Reactions of 3-Allylrhodanine with 2-Arylidene-4-methyl-5- oxopyrazolidinium ylides Rahhal El Ajlaoui
upporting Information Unexpected ynthesis of ovel 3-allyl-5-(arylidene)-2-thioxo-thiazolidin-4- ones in Reactions of 3-Allylrhodanine with 2-Arylidene-4-methyl-5- oxopyrazolidinium ylides Rahhal El Ajlaoui
Supporting Information
 Supporting Information C 2 fixation employing an Iridium(I)- hydroxide complex Byron J. Truscott, David J. elson, Alexandra M. Z. Slawin and Steven P. olan * EaStCHEM School of Chemistry, University of
Supporting Information C 2 fixation employing an Iridium(I)- hydroxide complex Byron J. Truscott, David J. elson, Alexandra M. Z. Slawin and Steven P. olan * EaStCHEM School of Chemistry, University of
structurally reduced cadpr analogue with calciummobilizing
 Supporting Information for Synthesis of cyclic N 1 -pentylinosine phosphate, a new structurally reduced cadpr analogue with calciummobilizing activity on PC12 cells Ahmed Mahal,1, Stefano D Errico,1, Nicola
Supporting Information for Synthesis of cyclic N 1 -pentylinosine phosphate, a new structurally reduced cadpr analogue with calciummobilizing activity on PC12 cells Ahmed Mahal,1, Stefano D Errico,1, Nicola
Supporting Information
 Supporting Information Development of Photostable Near-Infrared Cyanine Dyes Animesh Samanta, Marc Vendrell, Rajkumar Das and Young-Tae Chang. List of contents: 1. Synthetic procedures and characterization
Supporting Information Development of Photostable Near-Infrared Cyanine Dyes Animesh Samanta, Marc Vendrell, Rajkumar Das and Young-Tae Chang. List of contents: 1. Synthetic procedures and characterization
Synthesis of Novel Peptide Linkers: Simultaneous Cyclization and Labeling
 UPPRTING INFRMATIN ynthesis of Novel Peptide Linkers: imultaneous Cyclization and Labeling Gajanan K. Dewkar, Pedro B. Carneiro, Matthew C. T. Hartman* Department of Chemistry and Massey Cancer Center,
UPPRTING INFRMATIN ynthesis of Novel Peptide Linkers: imultaneous Cyclization and Labeling Gajanan K. Dewkar, Pedro B. Carneiro, Matthew C. T. Hartman* Department of Chemistry and Massey Cancer Center,
Convenient photooxidation of alcohols using dye sensitised zinc oxide in combination with silver nitrate and TEMPO
 Convenient photooxidation of alcohols using dye sensitised zinc oxide in combination with silver nitrate and TEMP Vineet Jeena and Ross S. Robinson* Department of Chemistry, University of KwaZulu-Natal,
Convenient photooxidation of alcohols using dye sensitised zinc oxide in combination with silver nitrate and TEMP Vineet Jeena and Ross S. Robinson* Department of Chemistry, University of KwaZulu-Natal,
Supporting Information for Copper(I)-NHC complexes as efficient catalysts for the synthesis of 1,4-disubstituted 1,2,3-sulfonyltriazoles in air
 Supporting Information for Copper(I)-NHC complexes as efficient catalysts for the synthesis of 1,4-disubstituted 1,2,3-sulfonyltriazoles in air Faϊma Lazreg a and Catherine S. J. Cazin a,b * a EastCHEM
Supporting Information for Copper(I)-NHC complexes as efficient catalysts for the synthesis of 1,4-disubstituted 1,2,3-sulfonyltriazoles in air Faϊma Lazreg a and Catherine S. J. Cazin a,b * a EastCHEM
Supporting Information
 Supporting Information Novel, efficient and bio-based synthesis of secondary arylamines from (-)-shikimic acid Wei Wu, a,b Yong Zou, *,a Yu Chen, a,b Jun Li, c Zeliang Lv, a,b Wen Wei, a Tongkun Huang,
Supporting Information Novel, efficient and bio-based synthesis of secondary arylamines from (-)-shikimic acid Wei Wu, a,b Yong Zou, *,a Yu Chen, a,b Jun Li, c Zeliang Lv, a,b Wen Wei, a Tongkun Huang,
To a slurry of 2,2 -dilithiobiphenyl bis TMEDA adduct (16) (27.0 g, 67.8 mmol) in diethyl
 Page S1 Contents of the supporting information:?? Experimental procedure for 19.?? Characterization of 27 (including 1 H-, 13 C-, DEPT, 1 H- 1 H COSY, 1 H- 13 C correlation spectra) and X-Ray data for
Page S1 Contents of the supporting information:?? Experimental procedure for 19.?? Characterization of 27 (including 1 H-, 13 C-, DEPT, 1 H- 1 H COSY, 1 H- 13 C correlation spectra) and X-Ray data for
Selective isomerization of terminal alkenes to Z-2-alkenes catalyzed by an airstable molybdenum(0) complex
 Selective isomerization of terminal alkenes to Z-2-alkenes catalyzed by an airstable molybdenum(0) complex Joseph Becica, Owen D. Glaze, Derek I. Wozniak, Graham E. Dobereiner* Department of Chemistry,
Selective isomerization of terminal alkenes to Z-2-alkenes catalyzed by an airstable molybdenum(0) complex Joseph Becica, Owen D. Glaze, Derek I. Wozniak, Graham E. Dobereiner* Department of Chemistry,
Supporting Information for:
 Supporting Information for: [Ir(N^N^N)(C^N)L] + : A New Family of Luminophores Combining Tunability and Enhanced Photostability Danielle N. Chirdon, Wesley J. Transue, Husain N. Kagalwala, Aman Kaur, Andrew
Supporting Information for: [Ir(N^N^N)(C^N)L] + : A New Family of Luminophores Combining Tunability and Enhanced Photostability Danielle N. Chirdon, Wesley J. Transue, Husain N. Kagalwala, Aman Kaur, Andrew
Dimethoxide-Catalyzed Condensation of Aldehydes with Alkenyl Trichloroacetates
 S1 Supporting information: Selective Synthesis of a,b-unsaturated Ketones by Dibutyltin Dimethoxide-Catalyzed Condensation of Aldehydes with Alkenyl Trichloroacetates Akira Yanagisawa, * Riku Goudu, and
S1 Supporting information: Selective Synthesis of a,b-unsaturated Ketones by Dibutyltin Dimethoxide-Catalyzed Condensation of Aldehydes with Alkenyl Trichloroacetates Akira Yanagisawa, * Riku Goudu, and
Development of a Practical Buchwald-Hartwig Amine Arylation Protocol using a Conveniently Prepared (NHC)Pd(R-allyl)Cl Catalyst
 Development of a Practical Buchwald-Hartwig Amine Arylation Protocol using a Conveniently Prepared (HC)Pd(R-allyl)Cl Catalyst Mark J. Cawley, a F. Geoffrey.. Cloke, b Stuart E. Pearson, c James S. Scott
Development of a Practical Buchwald-Hartwig Amine Arylation Protocol using a Conveniently Prepared (HC)Pd(R-allyl)Cl Catalyst Mark J. Cawley, a F. Geoffrey.. Cloke, b Stuart E. Pearson, c James S. Scott
Supporting Information for: Ruthenium Alkylidenes: Fast Initiators for Olefin Metathesis. Organometallics
 Supporting Information for: Ruthenium Alkylidenes: Fast Initiators for Olefin Metathesis Organometallics Joseph E. Williams, Mary J. Harner, and Michael B. Sponsler* Department of Chemistry Syracuse University
Supporting Information for: Ruthenium Alkylidenes: Fast Initiators for Olefin Metathesis Organometallics Joseph E. Williams, Mary J. Harner, and Michael B. Sponsler* Department of Chemistry Syracuse University
Supplemental Information. Synthesis, Characterization, and Solid State Elucidation of Unusual Pyridine Donor Uranyl Complexes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Supplemental Information Synthesis, Characterization, and Solid State Elucidation of Unusual Pyridine
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Supplemental Information Synthesis, Characterization, and Solid State Elucidation of Unusual Pyridine
Supporting Information
 Oxidative Furan-to-Indole Rearrangement. Synthesis of 2-(2-Acylvinyl)indoles and Flinderole С Analogues Anton S. Makarov, Anton A. Merkushev, Maxim G. Uchuskin, * Igor V. Trushkov Supporting Information
Oxidative Furan-to-Indole Rearrangement. Synthesis of 2-(2-Acylvinyl)indoles and Flinderole С Analogues Anton S. Makarov, Anton A. Merkushev, Maxim G. Uchuskin, * Igor V. Trushkov Supporting Information
organic compounds Phenyl N-(4-fluorophenyl)carbamate o1036 Yang and Wang doi: /s Acta Cryst. (2009).
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Phenyl N-(4-fluorophenyl)carbamate Zhao Yang and Zhi-Xiang Wang* Department of Pharmaceutical Engineering, China
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Phenyl N-(4-fluorophenyl)carbamate Zhao Yang and Zhi-Xiang Wang* Department of Pharmaceutical Engineering, China
Experimental. Crystal data. C 30 H 32 N 2 O 7 CH 4 O M r = Monoclinic, P2 1 a = (4) Å b = (3) Å c = (5) Å = 105.
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 9-{[4-(Dimethylamino)benzyl]amino}- 5-(4-hydroxy-3,5-dimethoxyphenyl)- 5,5a,8a,9-tetrahydrofuro[3 0,4 0 :6,7]-
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 9-{[4-(Dimethylamino)benzyl]amino}- 5-(4-hydroxy-3,5-dimethoxyphenyl)- 5,5a,8a,9-tetrahydrofuro[3 0,4 0 :6,7]-
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 [6,13-Bis(2,4-dichlorobenzoyl)- 5,7,12,14-tetramethyldibenzo[b,i]- [1,4,8,11]tetraazacyclotetradecinatoj
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 [6,13-Bis(2,4-dichlorobenzoyl)- 5,7,12,14-tetramethyldibenzo[b,i]- [1,4,8,11]tetraazacyclotetradecinatoj
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Bis[l-1,2-bis(1H-imidazol-1-ylmethyl)- benzene-j 2 N 3 :N 30 ]disilver(i) bis(4- amino-2,5-dichlorobenzenesulfonate)
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Bis[l-1,2-bis(1H-imidazol-1-ylmethyl)- benzene-j 2 N 3 :N 30 ]disilver(i) bis(4- amino-2,5-dichlorobenzenesulfonate)
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Silylium-Arene Adducts: An Experimental And Theoretical Study Muhammad Farooq Ibad, Peter Langer, Axel Schulz* and Alexander Villinger* This file includes: 1. Experimental p. 2 2.
SUPPORTING INFORMATION Silylium-Arene Adducts: An Experimental And Theoretical Study Muhammad Farooq Ibad, Peter Langer, Axel Schulz* and Alexander Villinger* This file includes: 1. Experimental p. 2 2.
Preparation and evaluation of demulsifiers agents for Basra crude oil
 Appl Petrochem Res (212) 1:29 33 DOI 1.7/s1323-11-3-1 ORIGINAL ARTICLE Preparation and evaluation of demulsifiers agents for Basra crude oil Hikmeat Abd Al-Raheem Ali Received: 2 July 211 / Accepted: 23
Appl Petrochem Res (212) 1:29 33 DOI 1.7/s1323-11-3-1 ORIGINAL ARTICLE Preparation and evaluation of demulsifiers agents for Basra crude oil Hikmeat Abd Al-Raheem Ali Received: 2 July 211 / Accepted: 23
data reports l-chlorido-bis[(dimethyl sulfoxide-jo)bis(triphenylphosphane-jp)silver(i)] Structure description Synthesis and crystallization
![data reports l-chlorido-bis[(dimethyl sulfoxide-jo)bis(triphenylphosphane-jp)silver(i)] Structure description Synthesis and crystallization data reports l-chlorido-bis[(dimethyl sulfoxide-jo)bis(triphenylphosphane-jp)silver(i)] Structure description Synthesis and crystallization](/thumbs/96/127067570.jpg) l-chlorido-bis[(dimethyl sulfoxide-jo)bis(triphenylphosphane-jp)silver(i)] nitrate ISSN 2414-3146 Yun-Hua Li* and Yong-Liang Shao College of Chemistry and Chemical Engineering, Lanzhou University Lanzhou
l-chlorido-bis[(dimethyl sulfoxide-jo)bis(triphenylphosphane-jp)silver(i)] nitrate ISSN 2414-3146 Yun-Hua Li* and Yong-Liang Shao College of Chemistry and Chemical Engineering, Lanzhou University Lanzhou
Department of Chemistry, College of Science, Tianjin University, Tianjin , P. R. China.
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2019 Supporting Information for Synthesis, Catalysis, and DFT Study of A Ruthenium Carbene
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2019 Supporting Information for Synthesis, Catalysis, and DFT Study of A Ruthenium Carbene
Supplementary Information
 Supplementary Information SYNTHESIS AND EVALUATION OF COUMARIN-RESVERATOL HYBRIDS AS SOYBEAN 15-LIPOXYGENAZE INHIBITORS Samira Rahmani-Nezhad, Leila Khosravani, Mina Saeedi, Kouros Divsalar, Loghman Firoozpour,
Supplementary Information SYNTHESIS AND EVALUATION OF COUMARIN-RESVERATOL HYBRIDS AS SOYBEAN 15-LIPOXYGENAZE INHIBITORS Samira Rahmani-Nezhad, Leila Khosravani, Mina Saeedi, Kouros Divsalar, Loghman Firoozpour,
Fingerprinting the oxidation state of U(IV) by
 Fingerprinting the oxidation state of U(IV) by emission spectroscopy Emtithal Hashem, 1 Giulia Lorusso 2 Marco Evangelisti, 2 Thomas McCabe, 1 Carola Schulzke, 3 James A. Platts 4 and Robert J. Baker 1*
Fingerprinting the oxidation state of U(IV) by emission spectroscopy Emtithal Hashem, 1 Giulia Lorusso 2 Marco Evangelisti, 2 Thomas McCabe, 1 Carola Schulzke, 3 James A. Platts 4 and Robert J. Baker 1*
Supporting Information. Dinuclear Aluminum Poly(phenolate) Complexes as Efficient Catalysts for Cyclic Carbonate Synthesis
 Supporting Information Dinuclear Aluminum Poly(phenolate) Complexes as Efficient Catalysts for Cyclic Carbonate Synthesis Pengfei Gao, Zhiwen Zhao, Lijuan Chen, Dan Yuan* and Yingming Yao* Key Laboratory
Supporting Information Dinuclear Aluminum Poly(phenolate) Complexes as Efficient Catalysts for Cyclic Carbonate Synthesis Pengfei Gao, Zhiwen Zhao, Lijuan Chen, Dan Yuan* and Yingming Yao* Key Laboratory
Experimental. Crystal data
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 N-(Diphenylcarbamoyl)-N,N 0,N 0,N 00,N 00 - pentamethylguanidinium tetraphenylborate Ioannis Tiritiris Fakultät
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 N-(Diphenylcarbamoyl)-N,N 0,N 0,N 00,N 00 - pentamethylguanidinium tetraphenylborate Ioannis Tiritiris Fakultät
Experimental. Crystal data. C 22 H 28 N 2 O 3 CH 4 O M r = Monoclinic, C2=c a = (4) Å b = (2) Å c = (5) Å = 96.
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 N 0 -(3,5-Di-tert-butyl-4-hydroxybenzylidene)-2-hydroxybenzohydrazide methanol solvate Wagee A. Yehye, Azhar Ariffin*
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 N 0 -(3,5-Di-tert-butyl-4-hydroxybenzylidene)-2-hydroxybenzohydrazide methanol solvate Wagee A. Yehye, Azhar Ariffin*
Mitoxantrone and Analogues Bind and Stabilise i-motif Forming DNA Sequences
 Mitoxantrone and Analogues Bind and Stabilise i-motif Forming DA Sequences Elisé P. Wright, Henry A. Day, Ali M. Ibrahim, Jeethendra Kumar, Leo J. E. Boswell, Camille Huguin, Clare E. M. Stevenson, Klaus
Mitoxantrone and Analogues Bind and Stabilise i-motif Forming DA Sequences Elisé P. Wright, Henry A. Day, Ali M. Ibrahim, Jeethendra Kumar, Leo J. E. Boswell, Camille Huguin, Clare E. M. Stevenson, Klaus
SUPPORTING INFORMATION
 Photoassisted Synthesis of Enantiopure Alkaloid Mimics N.N. Bhuvan Kumar, O. A. Mukhina, A. G. Kutateladze S1 Photoassisted Synthesis of Enantiopure Alkaloid Mimics Possessing Unprecedented Polyheterocyclic
Photoassisted Synthesis of Enantiopure Alkaloid Mimics N.N. Bhuvan Kumar, O. A. Mukhina, A. G. Kutateladze S1 Photoassisted Synthesis of Enantiopure Alkaloid Mimics Possessing Unprecedented Polyheterocyclic
2. Experimental Crystal data
 data reports ISSN 1600-5368 Crystal structure of bis{2,4-di-tert-butyl- 6-[(isopropylimino)methyl]phenolatoj 2 N,O}zinc dichloromethane monosolvate Yuan-Zeng Hao Guangzhou Super-Dragon Engineering Plastics
data reports ISSN 1600-5368 Crystal structure of bis{2,4-di-tert-butyl- 6-[(isopropylimino)methyl]phenolatoj 2 N,O}zinc dichloromethane monosolvate Yuan-Zeng Hao Guangzhou Super-Dragon Engineering Plastics
organic papers 2-[4-(Dimethylamino)phenyl]-4,5-diphenyl- 1H-imidazole isopropanol solvate Comment
![organic papers 2-[4-(Dimethylamino)phenyl]-4,5-diphenyl- 1H-imidazole isopropanol solvate Comment organic papers 2-[4-(Dimethylamino)phenyl]-4,5-diphenyl- 1H-imidazole isopropanol solvate Comment](/thumbs/94/120853572.jpg) organic papers Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 2-[4-(Dimethylamino)phenyl]-4,5-diphenyl- 1H-imidazole isopropanol solvate Hassan Martinez, a Horst Puschmann, b
organic papers Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 2-[4-(Dimethylamino)phenyl]-4,5-diphenyl- 1H-imidazole isopropanol solvate Hassan Martinez, a Horst Puschmann, b
Experimental. Crystal data. C 30 H 27 O 8 P M r = Triclinic, P1 a = (6) Å b = (4) Å c = (4) Å = (3) = 72.
 Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Tetramethyl 1,1,2-triphenyl-2H-1k 5 - phosphole-2,3,4,5-tetracarboxylate Krzysztof K. Krawczyk, a Krystyna Wojtasiewicz, a Jan K.
Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Tetramethyl 1,1,2-triphenyl-2H-1k 5 - phosphole-2,3,4,5-tetracarboxylate Krzysztof K. Krawczyk, a Krystyna Wojtasiewicz, a Jan K.
Supplementary Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supplementary Information Hoveyda-Grubbs catalyst analogues bearing derivatives of N-phenylpyrrol
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Supplementary Information Hoveyda-Grubbs catalyst analogues bearing derivatives of N-phenylpyrrol
Reactivity of a Series of Isostructural Cobalt Pincer Complexes with CO 2, CO, and H +
 Supporting Information Reactivity of a Series of Isostructural Cobalt Pincer Complexes with CO 2, CO, and H + David W. Shaffer, Samantha I. Johnson,, Arnold L. Rheingold, Joseph W. Ziller, William A. Goddard,
Supporting Information Reactivity of a Series of Isostructural Cobalt Pincer Complexes with CO 2, CO, and H + David W. Shaffer, Samantha I. Johnson,, Arnold L. Rheingold, Joseph W. Ziller, William A. Goddard,
Experimental. Crystal data
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Ethyl N-[4-(3-methyl-4,5-dihydrobenzo- [g]indazol-1-yl)phenylsulfonyl]thiocarbamate ethanol monosolvate Abdullah
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Ethyl N-[4-(3-methyl-4,5-dihydrobenzo- [g]indazol-1-yl)phenylsulfonyl]thiocarbamate ethanol monosolvate Abdullah
Supporting Information
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 Supporting Information Mesogenic
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 Supporting Information Mesogenic
data reports Bis{2-[({[3-(dimethylazaniumyl)propyl]imino}- methyl)phenyl]sulfanido}nickel(ii) tetraphenylborate Structure description
![data reports Bis{2-[({[3-(dimethylazaniumyl)propyl]imino}- methyl)phenyl]sulfanido}nickel(ii) tetraphenylborate Structure description data reports Bis{2-[({[3-(dimethylazaniumyl)propyl]imino}- methyl)phenyl]sulfanido}nickel(ii) tetraphenylborate Structure description](/thumbs/95/125071125.jpg) ISSN 2414-3146 Bis{2-[({[3-(dimethylazaniumyl)propyl]imino}- methyl)phenyl]sulfanido}nickel(ii) tetraphenylborate Alexandra Meloccaro, Joshua R. Zimmerman and David M. Eichhorn* Received 25 June 2017 Accepted
ISSN 2414-3146 Bis{2-[({[3-(dimethylazaniumyl)propyl]imino}- methyl)phenyl]sulfanido}nickel(ii) tetraphenylborate Alexandra Meloccaro, Joshua R. Zimmerman and David M. Eichhorn* Received 25 June 2017 Accepted
Supporting Information
 Supporting Information Design and synthesis of new Transient Receptor Potential Vanilloid Type-1 (TRPV1) channel modulators: identification and pharmacological characterization of the N-(4-hydroxy-3-methoxybenzyl)-4-(thiophen-2-
Supporting Information Design and synthesis of new Transient Receptor Potential Vanilloid Type-1 (TRPV1) channel modulators: identification and pharmacological characterization of the N-(4-hydroxy-3-methoxybenzyl)-4-(thiophen-2-
Bis(4,6-di-tert-butyl-2-{N-[4-(diethylamino)phenyl]- carboximidoyl}phenolato)cobalt(ii)
![Bis(4,6-di-tert-butyl-2-{N-[4-(diethylamino)phenyl]- carboximidoyl}phenolato)cobalt(ii) Bis(4,6-di-tert-butyl-2-{N-[4-(diethylamino)phenyl]- carboximidoyl}phenolato)cobalt(ii)](/thumbs/94/119443393.jpg) Bis(4,6-di-tert-butyl-2-{N-[4-(diethylamino)phenyl]- carboximidoyl}phenolato)cobalt(ii) ISSN 2414-3146 C.Vidya Rani, a L. Mitu, b G. Chakkaravarthi c * and G. Rajagopal d * Received 23 May 2017 Accepted
Bis(4,6-di-tert-butyl-2-{N-[4-(diethylamino)phenyl]- carboximidoyl}phenolato)cobalt(ii) ISSN 2414-3146 C.Vidya Rani, a L. Mitu, b G. Chakkaravarthi c * and G. Rajagopal d * Received 23 May 2017 Accepted
Fluorinated triazapentadienyl ligand supported ethyl zinc(ii) complexes: reaction with dioxygen and catalytic applications in the Tishchenko reaction
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Fluorinated triazapentadienyl ligand supported ethyl zinc(ii) complexes: reaction with
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Fluorinated triazapentadienyl ligand supported ethyl zinc(ii) complexes: reaction with
Bromomethyllithium-Mediated Chemoselective Homologation of Disulfides to Dithioacetals
 Electronic upplementary Material (EI) for ChemComm. This journal is The Royal ociety of Chemistry 2016 upporting Information for Bromomethyllithium-Mediated Chemoselective Homologation of Disulfides to
Electronic upplementary Material (EI) for ChemComm. This journal is The Royal ociety of Chemistry 2016 upporting Information for Bromomethyllithium-Mediated Chemoselective Homologation of Disulfides to
Supplemental Information for: Imidazole-appended Macrocyclic Complexes of Fe(II), Co(II) and Ni(II) as paracest Agents
 Supplemental Information for: Imidazole-appended Macrocyclic Complexes of Fe(II), Co(II) and Ni(II) as paracest Agents Patrick J. Burns, Jordan M. Cox, and Janet R. Morrow Department of Chemistry, University
Supplemental Information for: Imidazole-appended Macrocyclic Complexes of Fe(II), Co(II) and Ni(II) as paracest Agents Patrick J. Burns, Jordan M. Cox, and Janet R. Morrow Department of Chemistry, University
Experimental. Crystal data
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Calixarene-based molecular capsule from olefin metathesis Shimelis T. Hailu, Ray J. Butcher,* Paul F. Hudrlik
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Calixarene-based molecular capsule from olefin metathesis Shimelis T. Hailu, Ray J. Butcher,* Paul F. Hudrlik
5-Chloro-2-(thiophen-2-yl)-1-(thiophen-2-ylmethyl)-1H- benzimidazole 6-Chloro-2-(thiophen-2-yl)-1-(thiophen-2- ylmethyl)-1h-benzimidazole (0.94/0.
 Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 5-Chloro-2-(thiophen-2-yl)-1-(thiophen-2-ylmethyl)-1H- benzimidazole 6-Chloro-2-(thiophen-2-yl)-1-(thiophen-2- ylmethyl)-1h-benzimidazole
Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 5-Chloro-2-(thiophen-2-yl)-1-(thiophen-2-ylmethyl)-1H- benzimidazole 6-Chloro-2-(thiophen-2-yl)-1-(thiophen-2- ylmethyl)-1h-benzimidazole
Sydnone anions and abnormal N-heterocyclic carbenes of O- ethylsydnones. Characterizations, calculations and catalyses
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Sydnone anions and abnormal N-heterocyclic carbenes of O- ethylsydnones. Characterizations, calculations
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Sydnone anions and abnormal N-heterocyclic carbenes of O- ethylsydnones. Characterizations, calculations
Supporting Information
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Structural and insights into the coordination chemistry and reactivity
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Structural and insights into the coordination chemistry and reactivity
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 n-butyldichlorido{4-cyclohexyl-1-[1- (pyridin-2-yl-jn)ethylidene]thiosemicarbazidato-j 2 N 1,S}tin(IV) Md.
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 n-butyldichlorido{4-cyclohexyl-1-[1- (pyridin-2-yl-jn)ethylidene]thiosemicarbazidato-j 2 N 1,S}tin(IV) Md.
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Di-l-hydroxido-j 4 O:O-l-trifluoromethanesulfonato-j 2 O:O 0 -bis[(5,5 0 - dimethyl-2,2-bipyridine-j 2 N,N
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Di-l-hydroxido-j 4 O:O-l-trifluoromethanesulfonato-j 2 O:O 0 -bis[(5,5 0 - dimethyl-2,2-bipyridine-j 2 N,N
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Monoclinic, C2=c a = 14.519 (3) Å b = 14.303 (3) Å c = 12.461 (3) Å = 101.94 (3) V = 2531.6 (9) Å 3 Z =4
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Monoclinic, C2=c a = 14.519 (3) Å b = 14.303 (3) Å c = 12.461 (3) Å = 101.94 (3) V = 2531.6 (9) Å 3 Z =4
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 a = 8.8035 (11) Å b = 18.138 (2) Å c = 20.966 (3) Å = 95.512 (2) V = 3332.4 (7) Å 3 Z =4 Mo K radiation
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 a = 8.8035 (11) Å b = 18.138 (2) Å c = 20.966 (3) Å = 95.512 (2) V = 3332.4 (7) Å 3 Z =4 Mo K radiation
Experimental. Crystal data. C 33 H 48 N 4 O M r = Monoclinic, P2 1 a = (2) Å b = (2) Å c = (2) Å = 94.
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 2-(2H-Benzotriazol-2-yl)-6-[(dicyclohexylamino)methyl]-4-(2,4,4-trimethylpentan-2-yl)phenol Ming-Jen Chen, a Ban-Hsin
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 2-(2H-Benzotriazol-2-yl)-6-[(dicyclohexylamino)methyl]-4-(2,4,4-trimethylpentan-2-yl)phenol Ming-Jen Chen, a Ban-Hsin
metal-organic papers l 3 -Oxo-hexa-l 2 -pivalato-(pivalic acid-jo)- bis(pyridine-jn)trimanganese(iii,iii,ii) Comment Musa Sarı, a * Mehmet Poyraz, b
 metal-organic papers Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 l 3 -Oxo-hexa-l 2 -pivalato-(pivalic acid-jo)- bis(pyridine-jn)trimanganese(iii,iii,ii) Musa Sarı, a * Mehmet
metal-organic papers Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 l 3 -Oxo-hexa-l 2 -pivalato-(pivalic acid-jo)- bis(pyridine-jn)trimanganese(iii,iii,ii) Musa Sarı, a * Mehmet
Experimental. Crystal data. C 60 H 88 O 4 4CHCl 3 2H 2 O M r = Orthorhombic, Pbcn a = (6) Å b = (6) Å c = 25.
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 25,26,27,28-Tetrabutoxy-5,11,17,23- tetra-tert-butylcalix[4]arene chloroform tetrasolvate dihydrate Zhengyi Li,
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 25,26,27,28-Tetrabutoxy-5,11,17,23- tetra-tert-butylcalix[4]arene chloroform tetrasolvate dihydrate Zhengyi Li,
CHAPTER - 2 SYNTHESIS OF SUBSTITUTED-2,4-DIHYDRO [1,2,4]TRIAZOL-3-ONE.
![CHAPTER - 2 SYNTHESIS OF SUBSTITUTED-2,4-DIHYDRO [1,2,4]TRIAZOL-3-ONE. CHAPTER - 2 SYNTHESIS OF SUBSTITUTED-2,4-DIHYDRO [1,2,4]TRIAZOL-3-ONE.](/thumbs/96/128066464.jpg) 37 CHAPTER - 2 SYNTHESIS OF SUBSTITUTED-2,4-DIHYDRO [1,2,4]TRIAZOL-3-ONE. 2.1 INTRODUCTION: 1,2,4-Triazol-3-ones and their derivatives show a broad spectrum of biological activities [78] such as antivirals
37 CHAPTER - 2 SYNTHESIS OF SUBSTITUTED-2,4-DIHYDRO [1,2,4]TRIAZOL-3-ONE. 2.1 INTRODUCTION: 1,2,4-Triazol-3-ones and their derivatives show a broad spectrum of biological activities [78] such as antivirals
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Hexaaquacadmium(II) bis{[n-(2- oxidobenzylidene)glycyl-l-leucinato]- cuprate(ii)} dihydrate Guolin Zhang,
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Hexaaquacadmium(II) bis{[n-(2- oxidobenzylidene)glycyl-l-leucinato]- cuprate(ii)} dihydrate Guolin Zhang,
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Monoclinic, P2 1 =n a = 13.5565 (1) Å b = 15.7136 (2) Å c = 18.2264 (3) Å = 109.978 (1) V = 3648.97 (8)
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Monoclinic, P2 1 =n a = 13.5565 (1) Å b = 15.7136 (2) Å c = 18.2264 (3) Å = 109.978 (1) V = 3648.97 (8)
Data collection. Refinement. R[F 2 >2(F 2 )] = wr(f 2 ) = S = reflections 1118 parameters
![Data collection. Refinement. R[F 2 >2(F 2 )] = wr(f 2 ) = S = reflections 1118 parameters Data collection. Refinement. R[F 2 >2(F 2 )] = wr(f 2 ) = S = reflections 1118 parameters](/thumbs/90/102605898.jpg) organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Non-merohedrally twinned hexamethylenetetramine 4-nitrophenol water (1/2/1), triclinic modification Seik Weng
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Non-merohedrally twinned hexamethylenetetramine 4-nitrophenol water (1/2/1), triclinic modification Seik Weng
Synthesis and Application of Stereoretentive Ruthenium Catalysts on the Basis of the M7 and the Ru-Benzylidene- Oxazinone Designs
 ynthesis and Application of tereoretentive Ruthenium Catalysts on the Basis of the M7 and the Ru-Benzylidene- xazinone Designs Adrien Dumas,, Daniel. Müller, Idriss Curbet, Loïc Toupet, Matthieu Rouen,
ynthesis and Application of tereoretentive Ruthenium Catalysts on the Basis of the M7 and the Ru-Benzylidene- xazinone Designs Adrien Dumas,, Daniel. Müller, Idriss Curbet, Loïc Toupet, Matthieu Rouen,
Experimental. Crystal data
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 5,11,17,23-Tetra-tert-butyl-25,27-bis[2- (4-nitrophenoxy)ethoxy]calix[4]arene- 26,28-diol acetonitrile tetrasolvate
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 5,11,17,23-Tetra-tert-butyl-25,27-bis[2- (4-nitrophenoxy)ethoxy]calix[4]arene- 26,28-diol acetonitrile tetrasolvate
Bis{3,5-di-tert-butyl-N-[(4-dimethylamino)phenyl]- salicylaldiminato}cobalt(ii)
![Bis{3,5-di-tert-butyl-N-[(4-dimethylamino)phenyl]- salicylaldiminato}cobalt(ii) Bis{3,5-di-tert-butyl-N-[(4-dimethylamino)phenyl]- salicylaldiminato}cobalt(ii)](/thumbs/94/119443391.jpg) Bis{3,5-di-tert-butyl-N-[(4-dimethylamino)phenyl]- salicylaldiminato}cobalt(ii) ISSN 2414-3146 C. Vidya Rani, a L. Mitu, b G. Chakkaravarthi c * and G. Rajagopal d * Received 28 March 2017 Accepted 6 April
Bis{3,5-di-tert-butyl-N-[(4-dimethylamino)phenyl]- salicylaldiminato}cobalt(ii) ISSN 2414-3146 C. Vidya Rani, a L. Mitu, b G. Chakkaravarthi c * and G. Rajagopal d * Received 28 March 2017 Accepted 6 April
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Carbonyl{3,3 0 -di-tert-butyl-5,5 0 -dimethoxy-2,2 0 -bis[(4,4,5,5-tetramethyl-1,3,2- dioxaphospholan-2-yl)oxy]biphenylj
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Carbonyl{3,3 0 -di-tert-butyl-5,5 0 -dimethoxy-2,2 0 -bis[(4,4,5,5-tetramethyl-1,3,2- dioxaphospholan-2-yl)oxy]biphenylj
Experimental. Crystal data. C 42 H 63 O 4 P M r = Monoclinic, P2 1 =n a = (4) Å c = (4) Å = (6) Data collection
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Tris(2,4-di-tert-butylphenyl) phosphate T. Vinuchakkaravarthy, C. K. Sangeetha and D. Velmurugan* Centre of Advanced
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Tris(2,4-di-tert-butylphenyl) phosphate T. Vinuchakkaravarthy, C. K. Sangeetha and D. Velmurugan* Centre of Advanced
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 catena-poly[[[bis(o,o 0 -diisobutyl dithiophosphato-j 2 S,S 0 )nickel(ii)]-l-1,2-bis(4- pyridylmethylene)hydrazine-j
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 catena-poly[[[bis(o,o 0 -diisobutyl dithiophosphato-j 2 S,S 0 )nickel(ii)]-l-1,2-bis(4- pyridylmethylene)hydrazine-j
Jian-Wei Liu, Jing Zheng, Jin-Long Wang, Jie Xu, Hui-Hui Li, Shu-Hong Yu*
 Supporting Information Ultrathin 18 O 49 Nanowire Assemblies for Electrochromic Devices Jian-ei Liu, Jing Zheng, Jin-Long ang, Jie Xu, Hui-Hui Li, Shu-Hong Yu* Experimental Section Synthesis and Assembly
Supporting Information Ultrathin 18 O 49 Nanowire Assemblies for Electrochromic Devices Jian-ei Liu, Jing Zheng, Jin-Long ang, Jie Xu, Hui-Hui Li, Shu-Hong Yu* Experimental Section Synthesis and Assembly
data reports Bis(4-cobaltoceniumyl-1-ferrocenyl-3-methyltriazolylidene)gold(I) hexafluoridophosphate trifluoromethanesulfonate Structure description
 ISSN 2414-3146 Bis(4-cobaltoceniumyl-1-ferrocenyl-3-methyl-1,2,3- triazolylidene)gold(i) hexafluoridophosphate trifluoromethanesulfonate (1.2/1.8) Stefan Vanicek, Klaus Wurst, Holger Kopacka and Benno
ISSN 2414-3146 Bis(4-cobaltoceniumyl-1-ferrocenyl-3-methyl-1,2,3- triazolylidene)gold(i) hexafluoridophosphate trifluoromethanesulfonate (1.2/1.8) Stefan Vanicek, Klaus Wurst, Holger Kopacka and Benno
Synthesis of Silver Nanowires with Reduced Diameters Using Benzoin-Derived Radicals to Make Transparent Conductors with High Transparency and Low Haze
 Supporting Information Synthesis of Silver Nanowires with Reduced Diameters Using Benzoin-Derived Radicals to Make Transparent Conductors with High Transparency and Low Haze Zhiqiang Niu,, Fan Cui,, Elisabeth
Supporting Information Synthesis of Silver Nanowires with Reduced Diameters Using Benzoin-Derived Radicals to Make Transparent Conductors with High Transparency and Low Haze Zhiqiang Niu,, Fan Cui,, Elisabeth
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Three-dimensional TiO 2 /CeO 2 Nanowire composite for Efficient Formaldehyde
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Three-dimensional TiO 2 /CeO 2 Nanowire composite for Efficient Formaldehyde
Experimental. Crystal data. C 15 H 21 FN 3 O 4 P M r = Monoclinic, P2 1 =n a = (6) Å b = (4) Å c = (9) Å = 106.
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 A second monoclinic polymorph of N-[bis(morpholin-4-yl)phosphinoyl]- 4-fluorobenzamide with the P2 1 /n space
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 A second monoclinic polymorph of N-[bis(morpholin-4-yl)phosphinoyl]- 4-fluorobenzamide with the P2 1 /n space
Experimental. Crystal data. C 30 H 48 O 3 M r = Orthorhombic, P a = (11) Å b = (13) Å c = 15.
 Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 14-Hydroxy-8,14-secogammacera-7-ene- 3,21-dione from the bark of Lansium domesticum Corr. Unang Supratman, a Tri Mayanti, a Khalijah
Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 14-Hydroxy-8,14-secogammacera-7-ene- 3,21-dione from the bark of Lansium domesticum Corr. Unang Supratman, a Tri Mayanti, a Khalijah
A General Strategy for the Preparation of C-Terminal Peptide α-ketoacids by Solid Phase Peptide Synthesis. Lei Ju and Jeffrey W.
 A General Strategy for the Preparation of C-Terminal Peptide α-ketoacids by Solid Phase Peptide Synthesis Lei Ju and Jeffrey W. Bode* Roy and Diana Vagelos Laboratories, Department of Chemistry, University
A General Strategy for the Preparation of C-Terminal Peptide α-ketoacids by Solid Phase Peptide Synthesis Lei Ju and Jeffrey W. Bode* Roy and Diana Vagelos Laboratories, Department of Chemistry, University
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 cis-dichloridobis(1,10-phenanthrolinej 2 N,N 0 )manganese(ii) 2,6-dihydroxybenzoic acid water (2/1/4) Qian
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 cis-dichloridobis(1,10-phenanthrolinej 2 N,N 0 )manganese(ii) 2,6-dihydroxybenzoic acid water (2/1/4) Qian
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Highly Potent Extranuclear-targeted Luminiscent Iridium(III) Antitumor Agents
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Highly Potent Extranuclear-targeted Luminiscent Iridium(III) Antitumor Agents
Three Carboxyphenyl Groups Possessing Zinc Porphyrins: Efficient, Stable, and Cost-effective Sensitizers for Dye-Sensitized Solar Cells
 Supporting information Three Carboxyphenyl Groups Possessing Zinc Porphyrins: Efficient, Stable, and Cost-effective Sensitizers for Dye-Sensitized Solar Cells Ram B. Ambre, Gao-Fong Chang, and Chen-Hsiung
Supporting information Three Carboxyphenyl Groups Possessing Zinc Porphyrins: Efficient, Stable, and Cost-effective Sensitizers for Dye-Sensitized Solar Cells Ram B. Ambre, Gao-Fong Chang, and Chen-Hsiung
metal-organic compounds
 metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Dichlorido(3-phenylindenylidene)bis- (triphenylphosphane)ruthenium(ii) tetrahydrofuran disolvate Jan W.
metal-organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 Dichlorido(3-phenylindenylidene)bis- (triphenylphosphane)ruthenium(ii) tetrahydrofuran disolvate Jan W.
Experimental. Crystal data
 organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 The cocrystal 10-(2-methyl-4-phenyl-1H- inden-6-yl)-10h-phenothiazine 10-(2- methyl-7-phenyl-1h-inden-5-yl)-10hphenothiazine
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 The cocrystal 10-(2-methyl-4-phenyl-1H- inden-6-yl)-10h-phenothiazine 10-(2- methyl-7-phenyl-1h-inden-5-yl)-10hphenothiazine
Table of Contents. Synthetic procedures for 1-substituted indenes. Synthetic procedures and characterizing data for new compounds S4
 Supporting Information: Design of a Versatile and Improved Precatalyst Scaffold for Palladium Catalyzed Cross-Coupling: (η 3-1- t Bu-indenyl) 2 (µ- Cl) 2 Pd 2 Patrick R. Melvin, a Ainara Nova, b, * David
Supporting Information: Design of a Versatile and Improved Precatalyst Scaffold for Palladium Catalyzed Cross-Coupling: (η 3-1- t Bu-indenyl) 2 (µ- Cl) 2 Pd 2 Patrick R. Melvin, a Ainara Nova, b, * David
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information Aerobic oxidation at benzylic positions catalyzed by a simple Pd(OAc)
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information Aerobic oxidation at benzylic positions catalyzed by a simple Pd(OAc)
Non-Amphiphilic Assembly in Water: Polymorphic Nature, Thread Structure and Thermodynamic Incompatibility
 Supporting Information Non-Amphiphilic Assembly in Water: Polymorphic Nature, Thread Structure and Thermodynamic Incompatibility Lei Wu, Jyotsana Lal, Karen A. Simon, Erik A. Burton, Yan-Yeung Luk *,,
Supporting Information Non-Amphiphilic Assembly in Water: Polymorphic Nature, Thread Structure and Thermodynamic Incompatibility Lei Wu, Jyotsana Lal, Karen A. Simon, Erik A. Burton, Yan-Yeung Luk *,,
